Abstract
Purpose
To compare the outcomes of PN to those of RN in very elderly patients treated for clinically localized renal tumor.
Patients and methods
A purpose-built multi-institutional international database (RESURGE project) was used for this retrospective analysis. Patients over 75 years old and surgically treated for a suspicious of localized renal with either PN or RN were included in this database. Surgical, renal function and oncological outcomes were analyzed. Propensity scores for the predicted probability to receive PN in each patient were estimated by logistic regression models. Cox proportional hazard models were estimated to determine the relative change in hazard associated with PN vs RN on overall mortality (OM), cancer-specific mortality (CSM) and other-cause mortality (OCM).
Results
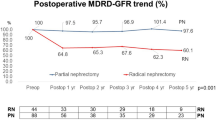
A total of 613 patients who underwent RN were successfully matched with 613 controls who underwent PN. Higher overall complication rate was recorded in the PN group (33% vs 25%; p = 0.01). Median follow-up for the entire cohort was 35 months (interquartile range [IQR] 13–63 months). There was a significant difference between RN and PN in median decline of eGFR (39% vs 17%; p < 0.01). PN was not correlated with OM (HR = 0.71; p = 0.56), OCM (HR = 0.74; p = 0.5), and showed a protective trend for CSM (HR = 0.19; p = 0.05). PN was found to be a protective factor for surgical CKD (HR = 0.28; p < 0.01) and worsening of eGFR in patients with baseline CKD. Retrospective design represents a limitation of this analysis.
Conclusions
Adoption of PN in very elderly patients with localized renal tumor does not compromise oncological outcomes, and it allows better functional preservation at mid-term (3-year) follow-up, relative to RN. Whether this functional benefit translates into a survival benefit remains to be determined.

Similar content being viewed by others
References
Campbell S, Uzzo RG, Allaf ME et al (2017) Renal mass and localized renal cancer: AUA Guideline. J Urol 198(3):520–529
Van Poppel H, Da Pozzo L, Albrecht W et al (2011) A prospective, randomised EORTC intergroup phase 3 study comparing the oncologic outcome of elective nephron-sparing surgery and radical nephrectomy for low-stage renal cell carcinoma. Eur Urol 59(4):543–552
Kane CJ, Mallin K, Ritchey J, Cooperberg MR, Carroll PR (2008) Renal cell cancer stage migration: analysis of the National Cancer Data Base. Cancer 113(1):78–83
Bianchi M, Gandaglia G, Trinh QD et al (2014) A population-based competing-risks analysis of survival after nephrectomy for renal cell carcinoma. Urol Oncol 32(1):46.e1–46.e7. https://doi.org/10.1016/j.urolonc.2013.06.010
Levey AS, Stevens LA (2010) Estimating GFR using the CKD epidemiology collaboration (CKD-EPI) creatinine equation: more accurate GFR estimates, lower CKD prevalence estimates, and better risk predictions. Am J Kidney Dis 55(4):622–627
Sekhon J (2011) Multivariate and propensity score matching. J Stat Softw 42(7):52
Grambsch PM, Therneau TM (1994) Proportional hazards tests and diagnostics based on weighted residuals. Biometrika 81(3):515–526
Kim SP, Gross CP, Meropol N et al (2017) National treatment trends among older patients with T1-localized renal cell carcinoma. Urol Oncol 35(3):113.e15–113.e21
Sun M, Becker A, Tian Z et al (2014) Management of localized kidney cancer: calculating cancer-specific mortality and competing risks of death for surgery and nonsurgical management. Eur Urol 65(1):235–241
Patel HD, Kates M, Pierorazio PM et al (2014) Survival after diagnosis of localized T1a kidney cancer: current population-based practice of surgery and nonsurgical management. Urology 83(1):126–132
Tan H-J, Daskivich TJ, Shirk JD, Filson CP, Litwin MS, Hu JC (2017) Health status and use of partial nephrectomy in older adults with early-stage kidney cancer. Urol Oncol 35(4):153.e7–153.e14
Lane BR, Abouassaly R, Gao T et al (2010) Active treatment of localized renal tumors may not impact overall survival in patients aged 75 years or older. Cancer 116(13):3119–3126
Mashni JW, Assel M, Maschino A et al (2015) New chronic kidney disease and overall survival after nephrectomy for small renal cortical tumors. Urology 86(6):1137–1143
Chung JS, Son NH, Lee SE et al (2015) Overall survival and renal function after partial and radical nephrectomy among older patients with localised renal cell carcinoma: a propensity-matched multicentre study. Eur J Cancer 51(4):489–497
Smaldone MC, Egleston B, Uzzo RG, Kutikov A (2012) Does partial nephrectomy result in a durable overall survival benefit in the medicare population? J Urol 188(6):2089–2094
Scosyrev E, Messing EM, Sylvester R, Campbell S, Van Poppel H (2014) Renal function after nephron-sparing surgery versus radical nephrectomy: results from EORTC randomized trial 30904. Eur Urol 65(2):372–377
Go AS, Chertow GM, Fan D, McCulloch CE, Hsu C (2004) Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med 351(13):1296–1305
Huang WC, Elkin EB, Levey AS, Jang TL, Russo P (2009) Partial nephrectomy versus radical nephrectomy in patients with small renal tumors-is there a difference in mortality and cardiovascular outcomes? J Urol 181(1):55–62
Capitanio U, Terrone C, Antonelli A et al (2015) Nephron-sparing techniques independently decrease the risk of cardiovascular events relative to radical nephrectomy in patients with a T1a-T1b renal mass and normal preoperative renal function. Eur Urol 67(4):683–689
Tan H-J, Norton EC, Ye Z, Hafez KS, Gore JL, Miller DC (2012) Long-term survival following partial vs radical nephrectomy among older patients with early-stage kidney cancer. JAMA 307(15):1629–1635
Shuch B, Hanley J, Lai J et al (2013) Overall survival advantage with partial nephrectomy: a bias of observational data? Cancer 119(16):2981–2989
Schiavina R, Borghesi M, Dababneh H et al (2015) Small renal masses managed with active surveillance: predictors of tumor growth rate after long-term follow-up. Clin Genitourin Cancer 13(2):e87–e92
May M, Cindolo L, Zigeuner R et al (2014) Results of a comparative study analyzing octogenarians with renal cell carcinoma in a competing risk analysis with patients in the seventh decade of life. Urol Oncol 32(8):1252–1258
Author information
Authors and Affiliations
Contributions
Protocol/project development: MCM, NP, UC, AA, ID, FP, and RA. Data collection or management: MCM, NP, OR-F, EL, TT, CF, CZ, PU, GF, CDN, ATT, MF, LB, JT, RC, RD, BDC, TS, AL, CP, MF, AB, ZH, AB, AR, and TA. Data analysis: GB, CG, MCM, and AC. Manuscript writing: RA, MCM, NP. Manuscript editing: UC, AA, ID, KHR, TM, CZ, AM, J-AL, AT, ODC, SM, LS, PB, AC, CT, FM, AB, JP, AA, KT, BY, EL, SC, SP, and ID.
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declare that they have no competing interests.
Research involving human participants
The RESURGE database is an IRB approved study at participating Centers.
Informed consent
Patients included in this retrospective analysis had signed an informed consent at the time of the surgery.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mir, M.C., Pavan, N., Capitanio, U. et al. Partial versus radical nephrectomy in very elderly patients: a propensity score analysis of surgical, functional and oncologic outcomes (RESURGE project). World J Urol 38, 151–158 (2020). https://doi.org/10.1007/s00345-019-02665-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-019-02665-2