Abstract
Introduction
High-quality evidence regarding questions of diagnostic accuracy relies on transparent reporting of study results. The quality of reporting for such studies in the urologic literature is unknown.
Methods
In accordance with an a priori protocol, we systematically searched for all articles on diagnostic accuracy studies published in four major urologic journals in 2015. Using the 2015 STAndards for Reporting Diagnostic accuracy studies (STARD) checklist, two of us independently abstracted data. For each article, we calculated STARD summary scores (scale of 0–30, with higher scores reflecting higher-quality reporting). We compared scores by journal, topic, and sample size.
Results
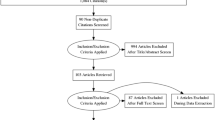
We screened 819 references of which 61 met inclusion criteria. Nearly two-thirds of studies (39/61%; 63.9%) addressed prostate cancer diagnosis or staging; less than one in ten (6/61%; 9.8%) was conducted in non-oncological disease settings. The major focus for the investigation of new index tests lay in imaging modalities (33/61%; 54.1%); over half of these imaging studies addressed magnetic resonance imaging (18/61%; 29.5%). The average STARD score was 18.9 ± 2.4 (range 12–24). Six criteria had poor reporting compliance and were met by less than 20% of studies. We found no association between reporting quality and topic, journal or study size.
Conclusions
The reporting quality of studies of diagnostic accuracy appears modest and independent of topic, journal or study size. There is an urgent need for greater awareness for the reporting quality of these studies among readers, editors, and investigators to raise evidentiary standards on issues of diagnosis.


Similar content being viewed by others
References
Simera I, Altman DG (2009) ACP journal club. editorial: writing a research article that is “fit for purpose”: EQUATOR network and reporting guidelines. Ann Intern Med 151:JC2-2–JC2-3
Schulz KF, Altman DG, Moher D et al (2010) CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials. PLoS Med 7:e1000251
Narayan VM, Cone EB, Smith D et al (2016) Improved reporting of randomized controlled trials in the urologic literature. Eur Urol 70:1044–1049
Bossuyt PM, Reitsma JB, Bruns DE et al (2003) The STARD statement for reporting studies of diagnostic accuracy: explanation and elaboration. Ann Intern Med 138:W1–12
Scales CD Jr, Dahm P, Sultan S et al (2008) How to use an article about a diagnostic test. J Urol 180:469–476
Bossuyt PM, Reitsma JB, Bruns DE et al (2015) STARD 2015: an updated list of essential items for reporting diagnostic accuracy studies. BMJ 351:h5527
Han JL, Gandhi S, Bockoven CG et al (2017) The landscape of systematic reviews in urology (1998–2015): an assessment of methodological quality. BJU Int 119:638–649
Tseng TY, Breau RH, Fesperman SF et al (2008) Evaluating the evidence: the methodological and reporting quality of comparative observational studies of surgical interventions in urological publications. BJU Int 103:1026–1031
Lokker C, Haynes RB, Wilczynski NL et al (2011) Retrieval of diagnostic and treatment studies for clinical use through PubMed and PubMed’s Clinical Queries filters. JAMIA 18:652–659
Covidence systematic review software, Melbourne, Australia, Veritas Health Innovation. www.covidence.org. Accessed 20 Aug 2018
Research registry. www.researchregistry.com. Accessed 20 Aug 2018
McGinn T, Wyer PC, Newman TB et al (2004) Tips for learners of evidence-based medicine: 3. measures of observer variability (kappa statistic). CMAJ 171:1369–1373
Ahmed HU, El-Shater Bosaily A, Brown LC et al (2017) Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet 389:815–822
Korevaar DA, Wang J, van Enst WA et al (2015) Reporting diagnostic accuracy studies: some improvements after 10 years of STARD. Radiology 274:781–789
Gallo L, Hua N, Mercuri M et al (2017) Adherence to standards for reporting diagnostic accuracy in emergency medicine research. Acad Emerg Med 24:914–919
Toews I, Binder N, Wolff RF et al (2017) Guidance in author instructions of hematology and oncology journals: a cross sectional and longitudinal study. PLoS One 12:e0176489
Esserman LJ, Thompson IM, Reid B (2013) Overdiagnosis and overtreatment in cancer: an opportunity for improvement. JAMA 310:797–798
Macleod MR, Michie S, Roberts I et al (2014) Biomedical research: increasing value, reducing waste. Lancet 383:101–104
Korevaar DA, Hooft L, Askie LM et al (2017) Facilitating prospective registration of diagnostic accuracy studies: a STARD initiative. Clin Chem 63:1331–1341
Kunath F, Grobe HR, Rucker G et al (2012) Do journals publishing in the field of urology endorse reporting guidelines? A survey of author instructions. Urol Int 88:54–59
Acknowledgement
We thank Mary Knaterrud, Ph.D. for her careful editorial review of the grammar and wording of this manuscript.
Author information
Authors and Affiliations
Contributions
DWS: protocol/project development, data collection and management, manuscript writing and editing SG: data collecting and management, manuscript editing PD: protocol/project development, data analysis, manuscript writing and editing
Corresponding author
Ethics declarations
Conflict of interest
Nothing to disclose.
Research involving human participants and/or animals
For this type of study formal consent is not required. This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Smith, D.W., Gandhi, S. & Dahm, P. The reporting quality of studies of diagnostic accuracy in the urologic literature. World J Urol 37, 969–974 (2019). https://doi.org/10.1007/s00345-018-2446-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-018-2446-9