Abstract
Introduction
The 2005 international society of urological pathology consensus statement on Gleason grading in prostate cancer revised Gleason scoring in clinical practice. The potential for grade migration with this refinement poses difficulties in interpreting historical series. We report the characteristics of a recent cohort of consecutive Gleason score 9 or 10 prostate cancers in our institution. The purpose of this study was to define the clinicopathologic variables and staging information for this high-risk population, and to identify whether traditional prostate staging techniques are adequate for this subcohort of men.
Materials and methods
A computational review of our pathology database was performed. Between May 2010 and September 2012, 1,295 consecutive biopsies were undertaken, 168 of which were high-grade tumours (12.97 %). This group were divided into two cohorts of which 84 (12.05 %) had a highest reported Gleason score of 9 (N = 79) or 10 (N = 5) and 84 were reported as Gleason 8. All biopsies were double-reported by pathologists with a special interest in uropathology.
Results
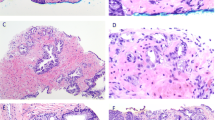
Men diagnosed with a Gleason pattern 5 tumour were statistically far more likely to have advanced disease on direct rectal examination of the prostate compared with Gleason sum 8 tumours (p < 0.001) and a positive first-degree family history of prostate cancer (p < 0.001). Overall, Gleason sum 9/10 prostate cancers were also found to be statistically more aggressive than Gleason sum 8 tumours on TRUS core biopsy analysis with significantly higher levels of perineural invasion (p < 0.0001) and extracapsular extension (p = 0.001) as well as a higher levels of tumour found within the core biopsy sample. Those men diagnosed with Gleason pattern 5 prostate cancer also had radiological indicators of increased tumour aggressiveness compared with Gleason sum 8 cancer with respect to bone (p = 0.0002) and visceral (p = 0.044) metastases at presentation.
Conclusions
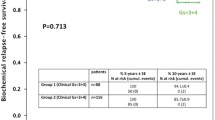
This series of Gleason score 9/10 prostate cancers serves to highlight the large disease burden, adverse pathologic features, and locally advanced nature of this aggressive subtype, which has previously been under-described in the literature, and differs from historical series in having a large high-grade cohort demonstrating high rates of metastatic disease. A history of prostate cancer amongst first-degree relatives was particularly prevalent in this population raising the issue of screening in a high-risk population. The high incidence of visceral metastatic disease at presentation supports upfront staging with CT thorax, abdomen, and pelvis in patients with Gleason 9 or 10 prostate cancers.

Similar content being viewed by others
Abbreviations
- CaP:
-
Prostate cancer
- CT:
-
Computed tomography
- EBRT:
-
External beam radiation therapy
- GP:
-
General practitioner
- ISUP:
-
International society for urological pathology
- MRI:
-
Magnetic resonance imaging
- PSA:
-
Prostate-specific antigen
- TNM:
-
Tumour, node, metastasis
- TRUS:
-
Transrectal ultrasound
References
Epstein JI (2010) An update of the Gleason grading system. J Urol 183:443
Nanda A, Chen MH, Renshaw AA et al (2009) Gleason pattern 5 prostate cancer: further stratification of patients with high-risk disease and implications for future randomized trials. Int J Radiat Oncol Biol Phy 74:1419
Pierorazio PM, Guzzo TJ, Han M et al (2010) Long-term survival after radical prostatectomy for men with high Gleason sum in pathologic specimen. Urology 76:715
Chan SM, Garcia FJ, Chin JL et al (2011) The clinical significance of in-depth pathological assessment of extraprostatic extension and margin status in radical prostatectomies for prostate cancer. Prostate Cancer Prostatic Dis 14(4):307–312
Sebo TJ, Bock BJ, Cheville JC et al (2000) The percent of cores positive for cancer in prostate needle biopsy specimens is strongly predictive of tumor stage and volume at radical prostatectomy. J Urol 163:174–178
D’Amico AV, Whittington R, Malkowicz SB et al (2000) Clinical utility of the percentage of positive prostate biopsies in defining biochemical outcome after radical prostatectomy for patients with clinically localized prostate cancer. J Clin Oncol 18:1164–1172
Freedland SJ, Aronson WJ, Terris MK et al (2003) Percent of prostate needle biopsy cores with cancer is significant independent predictor of prostate specific antigen recurrence following radical prostatectomy: results from SEARCH database. J Urol 169:2136–2141
Chaux A, Fajardo DA, Gonzalez-Roibon N et al (2012) High grade prostatic adenocarcinoma present in a single biopsy core is associated with increased extraprostatic extension, seminal vesicle invasion, and positive surgical margins at prostatectomy. Urology 79(4):863–868
Eifler JB, Feng Z, Lin BM et al (2012) An updated prostate cancer staging nomogram (Partin Tables) based on cases from 2006–2011. BJU Int 111:22–29
Epstein JI, Allsbrook WC Jr, Amin MB et al (2005) The 2005 international society of urological pathology (ISUP) consensus conference on Gleason grading of prostatic carcinoma. Am J Surg Pathol 29:1228–1242
Uemura H, Hoshino K, Sasaki T et al (2009) Usefulness of the 2005 International Society of Urologic Pathology Gleason grading system in prostate biopsy and radical prostatectomy specimens. BJU Int 103:1190–1194
Stark JR, Perner S, Stampfer MJ et al (2009) Gleason score and lethal prostate cancer: does 3 + 4 = 4 + 3? J Clin Oncol 27:3459–3464
Stenmark MH, Blas K, Halverson S et al (2011) Continued benefit to androgen deprivation therapy for prostate cancer patients treated with dose-escalated radiation therapy across multiple definitions of high-risk disease. Int J Radiat Oncol Biol Phys 81:e335–e344
Patel AA, Chen MH, Renshaw AA, D’Amico AV (2007) PSA failure following definitive treatment of prostate cancer having biopsy Gleason score 7 with tertiary grade 5. JAMA 298:1533–1538
Sabolch A, Feng FY, Daignault-Newton S et al (2011) Gleason pattern 5 is the greatest risk factor for clinical failure and death from prostate cancer after dose-escalated radiation therapy and hormonal ablation. Int J Radiat Oncol Biol Phys 81:e351–e360
Eifler JB, Feng Z, Lin BM et al. An updated prostate cancer staging nomogram (Partin tables) based on cases from 2006 to 2011. BJU Int. (in press). doi:10.1111/j.1464-410X.2012.11324.x
Epstein JI, Feng Z, Trock BJ et al (2012) Upgrading and downgrading of prostate cancer from biopsy to radical prostatectomy: incidence and predictive factors using the modified Gleason grading system and factoring in tertiary grades. Eur Urol 61(5):1019–1024
Moussa AS, Li J, Soriano M et al (2009) Prostate biopsy and clinical and pathological variables that predict significant grading changes in patients with intermediate and high grade prostate cancer. BJU Int 103(1):43–48
DeLancey JO, Wood DP Jr, He C et al (2013) Evidence of perineural invasion on prostate biopsy specimen and survival after radical prostatectomy. Urology 81(2):354–357
Feng FY, Qian Y, Stenmark MH et al (2011) Perineural invasion predicts increased recurrence, metastasis and death from prostate cancer following treatment with dose-escalated radiation therapy. Int J Radiat Oncol Biol Phys 81(4):e361–e367
Nanda A, Chen MH, Renshaw AA et al (2009) Gleason pattern 5 prostate cancer: further stratification of patients with high-risk disease and implications for future randomized trials. Int J Radiat Oncol Biol Phys 74(5):1419–1423
Moreira DM, Banez LL, Presti JC et al (2010) Predictors of secondary treatment following biochemical recurrence after radical prostatectomy: results from the shared equal access regional cancer hospital database. BJU Int 105(1):28–33
Elshafei A, Moussa AS, Hatem A et al (2013) Does positive family history of prostate cancer increase the risk of prostate cancer on initial prostate biopsy. Urology 81(4):826–830
Roudgari H, Hemminki K, Brandt A et al (2012) Prostate cancer risk assessment model: a scoring model based on the Swedish family-cancer database. J Med Genet 49(5):345–352
Jansson KF, Akre O, Garmo H et al (2012) Concordance of tumour differentiation among brothers with prostate cancer. Eur Urol 62(4):656–661
Cancer Research UK (2011) European age-standardized rates of prostate cancer. GLOBOCAN 2008 v1.2. IARC. http://www.cancerresearchuk.org/cancer-info/cancerstats/types/prostate/incidence/uk-prostate-cancer-incidence-statistics
Holmstrom B, Holmberg E, Egevad L, Adolfsson J, Johansson JE, Stattin P, National Prostate Cancer Registry of Sweden (2010) Outcome of primary versus deferred radical prostatectomy in the National Prostate Cancer Register of Sweden Follow-Up Study. J Urol 184:1322–1327
O’Brien D, Loeb S, Carvalhal GF et al (2011) Delay of surgery in men with low risk prostate cancer. J Urol 185:2143–2147
Lee N, Newhouse JH, Olsson CA et al (1999) Which patients with newly diagnosed prostate cancer need a computed tomography scan of the abdomen and pelvis? An analysis based on 588 patients. Urology 54(3):490–494
Masterson TA, Touijer K (2008) The role of endorectal coil MRI in preoperative staging and decision-making for the treatment of clinically localized prostate cancer. MAGMA 21(6):371–377
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
O’Kelly, F., Elamin, S., Cahill, A. et al. Characteristics of modern Gleason 9/10 prostate adenocarcinoma: a single tertiary centre experience within the Republic of Ireland. World J Urol 32, 1067–1074 (2014). https://doi.org/10.1007/s00345-013-1184-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-013-1184-2