Abstract
Purpose
Conventional techniques used to harvest and culture bladder smooth muscle cells (SMCs) have been thought to yield homogeneous populations of SMCs. In order to delineate the cellular composition of tissue derived bladder cells, this study was conducted to determine whether current culturing techniques result in a uniform population of bladder SMCs that may be utilized for bladder tissue engineering.
Methods
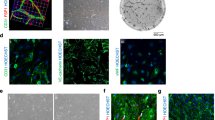
Patient derived bladder muscle was isolated and manually minced followed by enzymatic digestion. Cells were cultured in d-valine α-MEM with decreasing levels of fetal bovine serum then fixed and permeabilized for flow cytometric and immunofluorescent analyses. Antibody staining of cultured cells consisted of α-SMA, von Willebrand factor, pan-cytokeratin, CD31, and CD90. Cells were visualized using directly conjugated fluorescein isothiocyanate primary or IgG-Alexa-555 conjugated secondary antibodies.
Results
Flow cytometric analyses revealed mixed populations of cells expressing non-SMC epitopes as corroborated by immunofluorescent studies. High density oligonucleotide array analysis revealed expression levels of known bladder SMC genes and the expression of endothelial and fibroblast related markers (P < 0.005).
Conclusions
Phenotypic analyses demonstrate cell heterogeneity when SMCs are acquired and cultured through conventional methods. Standardized criteria based upon objective experimentation need to be established in order to better characterize bladder SMCs that are to be utilized for bladder tissue engineering.



Similar content being viewed by others
References
Samson G, Cardenas DD (2007) Neurogenic bladder in spinal cord injury. Phys Med Rehabil Clin 18:255–274
Proesmans W (2008) The neurogenic bladder: introducing four contributions. Pediatr Nephrol 23:537–540. doi:10.1007/s00467-008-0768-3
Mingin GC, Nguyen HT, Mathias RS et al (2002) Growth and metabolic consequences of bladder augmentation in children with myelomeningocele and bladder exstrophy. Pediatrics 110:1193–1198. doi:10.1542/peds.110.6.1193
Atala A, Bauer SB, Soker S et al (2006) Tissue-engineered autologous bladders for patients needing cystoplasty. Lancet 367:1241–1246. doi:10.1016/S0140-6736(06)68438-9
Magnan M, Berthod F, Champigny MF et al (2006) In vitro reconstruction of a tissue-engineered endothelialized bladder from a single porcine biopsy. J Pediatr Urol 2:261–270. doi:10.1016/j.jpurol.2005.11.019
Au P, Tam J, Fukumura D, Jain RK (2007) Small blood vessel engineering. Methods Mol Med 140:183–195. doi:10.1007/978-1-59745-443-8_11
Jayo MJ, Jain D, Wagner BJ et al (2008) Early cellular and stromal responses in regeneration versus repair of a mammalian bladder using autologous cell and biodegradable scaffold technologies. J Urol 180:392–397. doi:10.1016/j.juro.2008.02.039
Eichler MJ, Carlson MA (2005) Modeling dermal granulation tissue with the linear fibroblast-populated collagen matrix: a comparison with the round matrix model. J Dermatol Sci 41:97–108. doi:10.1016/j.jdermsci.2005.09.002
Baskin LS, Howard PS, Duckett JW et al (1993) Bladder smooth muscle cells in culture: I. Identification and characterization. J Urol 149:190–197
Baskin LS, Macarak EJ, Duckett JW et al (1993) Culture of urethral fibroblasts: cell morphology, proliferation and extracellular matrix synthesis. J Urol 150:1260–1266
Sutherland RS, Baskin LS, Hayward SW et al (1996) Regeneration of bladder urothelium, smooth muscle, blood vessels and nerves into an acellular tissue matrix. J Urol 156:571–577. doi:10.1016/S0022-5347(01)65755-0
Zhang Y, Kropp BP, Moore P et al (2000) Coculture of bladder urothelial and smooth muscle cells on small intestinal submucosa: potential applications for tissue engineering technology. J Urol 164:928–934. doi:10.1016/S0022-5347(05)67220-5
Zhang Y, Kropp BP, Lin HK et al (2004) Bladder regeneration with cell-seeded small intestinal submucosa. Tissue Eng 10:181–187. doi:10.1089/107632704322791835
Howard PS, Renfrow D, Schechter NM et al (2004) Mast cell chymase is a possible mediator of neurogenic bladder fibrosis. Neurourol Urodyn 23:374–382. doi:10.1002/nau.20032
Mothersill C, O’Brien A, Seymour CB (1990) The effect of radiation in combination with carcinogens on the growth of normal urothelium in explant culture. Radiat Environ Biophys 29:213–223. doi:10.1007/BF01210524
Mothersill C, Seymour CB, Mulvin D et al (1992) Endothelial cell proliferation is induced by radiation in cultured explants of human urothelium and oesophageal mucosa. EXS 61:407–410
Frauli M, Ludwig H (1987) Inhibition of fibroblast proliferation in a culture of human endometrial stromal cells using a medium containing d-valine. Arch Gynecol Obstet 241:87–96. doi:10.1007/BF00931229
Vardhanabhuti S, Blakemore SJ, Clark SM et al (2006) A comparison of statistical tests for detecting differential expression using Affymetrix oligonucleotide microarrays. OMICS 10:555–556. doi:10.1089/omi.2006.10.555
Wettenhall JM, Simpson KM, Satterley K et al (2006) affylmGUI: a graphical user interface for linear modeling of single channel microarray data. Bioinformatics 22:897–899. doi:10.1093/bioinformatics/btl025
Hagood JS, Lasky JA, Nesbitt JE et al (2001) Differential expression, surface binding, and response to connective tissue growth factor in lung fibroblast subpopulations. Chest 120:64S–66S. doi:10.1378/chest.120.1_suppl.S64
RayChaudhury A, Elkins M, Kozien D et al (2001) Regulation of PECAM-1 in endothelial cells during cell growth and migration. Exp Biol Med 226:686–691
Redick SD, Bautch VL (1999) Developmental platelet endothelial cell adhesion molecule expression suggests multiple roles for a vascular adhesion molecule. Am J Pathol 154:1137–1147
Jaffe EA, Hoyer LW, Nachman RL (1974) Synthesis of von Willebrand factor by cultured human endothelial cells. Proc Natl Acad Sci USA 71:906–1909. doi:10.1073/pnas.71.5.1906
Lazzaro VA, Walker RJ, Duggin GG et al (1992) Inhibition of fibroblast proliferation in l-valine reduced selective media. Res Commun Chem Pathol Pharmacol 75:39–48
Sabatini F, Petecchia L, Tavian M et al (2005) Human bronchial fibroblasts exhibit a mesenchymal stem cell phenotype and multilineage differentiating potentialities. Lab Invest 85:962–971. doi:10.1038/labinvest.3700300
Wu HY, Baskin LS, Liu W et al (1999) Understanding bladder regeneration: smooth muscle ontogeny. J Urol 162:1101–1105. doi:10.1016/S0022-5347(01)68082-0
Terada N, Hamazaki T, Oka M et al (2002) Bone marrow cells adopt the phenotype of other cells by spontaneous cell fusion. Nature 416:542–545. doi:10.1038/nature730
Jankowski RJ, Deasy BM, Cao B et al (2002) The role of CD34 expression and cellular fusion in the regeneration capacity of myogenic progenitor cells. J Cell Sci 115:4361–4374. doi:10.1242/jcs.00110
Hipp JA, Hipp JD, Yoo JJ et al (2008) Microarray analysis of bladder smooth muscle from patients with myelomeningocele. BJU Int 102:741–746. doi:10.1111/j.1464-410X.2008.07606.x
Kropp BP, Zhang Y, Tomasek JJ et al (1999) Characterization of cultured bladder smooth muscle cells: assessment of in vitro contractility. J Urol 162:1779–1784. doi:10.1016/S0022-5347(05)68237-7
Chern J, Kamm KE, Rosenfeld CR (1995) Smooth muscle myosin heavy chain isoforms are developmentally regulated in male fetal and neonatal sheep. Pediatr Res 38:697–703. doi:10.1203/00006450-199511000-00011
Deng M, Mohanan S, Polyak E et al (2007) Caldesmon is necessary for maintaining the actin and intermediate filaments in cultured bladder smooth muscle cells. Cell Motil Cytoskeleton 64:951–965. doi:10.1002/cm.20236
Conflict of interest statement
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sharma, A.K., Donovan, J.L., Hagerty, J.A. et al. Do current bladder smooth muscle cell isolation procedures result in a homogeneous cell population? Implications for bladder tissue engineering. World J Urol 27, 687–694 (2009). https://doi.org/10.1007/s00345-009-0391-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-009-0391-3