Abstract
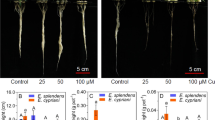
To understand the adaptation mechanism of vetiver under copper (Cu) stress, the effects of 5 concentrations of Cu (0, 40, 60, 120, and 150 mg/L CuSO4) on the morphological and physiological responses of vetiver roots were studied. The differential protein expression in vetiver roots treated with Cu-free (CK) and 60 mg/L CuSO4 was analyzed by 4D label-free quantitative proteomics technology. The results showed that the root length, root volume, and root tip number of vetiver initially increased and then decreased with increasing Cu concentration. The maximum values of root length and root volume appeared at 60 mg/L, which increased by 13.91% and 85.16%, respectively, compared with CK; copper ions (Cu2+) and malondialdehyde (MDA) contents increased with increasing Cu concentration and increased by 1626.40% ~ 2197.46% and 3.61% ~ 76.07% compared with CK, respectively; superoxide dismutase (SOD) increased first and then decreased and increased by 126.52% at 60 mg/L compared with CK; soluble protein reduced by 11.76% at 60 mg/L compared with CK (P < 0.05). A total of 1676 differentially expressed proteins were identified in vetiver roots after Cu stress, of which 765 were upregulated and 911 were downregulated. The differentially expressed proteins mainly occur in subcellular structures such as the cytoplasm, nucleus, and tonoplast; are enriched in amino sugar and nucleotide sugar metabolism, phenylpropanoid biosynthesis, cyanoamino acid metabolism, galactose metabolism, steroid biosynthesis, and other pathways; are involved in hydrolase, internal transferases, pyruvate carboxykinase, etc.; and identified the c-terminal domains of cupin, serine carboxypeptidase, peroxidase, and α-l-arabinofuranosidase. The key proteins, including USP, FRK2, HXK3, RFS6, EXPB, OMT2, AS, SODA3, Pod, ZB8, and PRX7, were significantly expressed and are involved in vetiver's regulation of energy metabolism, root morphological growth, and oxidative stress to increase the tolerance of plants to Cu stress. This study further elaborated on the adaptive mechanisms of vetiver under Cu stress and provided a theoretical basis for the ecological restoration of tailings vegetation.







Similar content being viewed by others
References
Aina R, Labra M, Fumagalli P et al (2007) Thiol-peptide level and proteomic changes in response to cadmium toxicity in Oryza sativa L. roots. Environ Exp Bot 59(3):381–392. https://doi.org/10.1016/j.envexpbot.2006.04.010
Atkinson NJ, Urwin PE (2012) The interaction of plant biotic and abiotic stresses: from genes to the field. J Exp Bot 63(10):3523–3543. https://doi.org/10.1093/jxb/ers100
Azeem S, Li Z, Zheng H et al (2016) Quantitative proteomics study on Lsi1 in regulation of rice (Oryza sativa L.) cold resistance. Plant Growth Regul 78:307–323. https://doi.org/10.1007/s10725-015-0094-2
Biyiklioglu S, Alptekin B, Akpinar BA et al (2018) A large-scale multiomics analysis of wheat stem solidness and the wheat stem sawfly feeding response, and syntenic associations in barley, Brachypodium, and rice. Funct Integr Genom 18:241–259. https://doi.org/10.1007/s10142-017-0585-5
Bordoloi K, Dihingia P, Krishnatreya D, Agarwala N (2021) Genome-wide identification, characterization and expression analysis of the expansin gene family under drought stress in tea (Camellia sinensis L.). Plant Science Today 8(1):32–44
Borges KLR, Salvato F, Loziuk PL, Muddiman DC, Azevedo RA (2019) Quantitative proteomic analysis of tomato genotypes with differential cadmium tolerance. Environ Sci Pollut Res 26:26039–26051. https://doi.org/10.1007/s11356-019-05766-y
Chao N, Qi Q, Li S, Ruan B, Jiang X, Gai Y (2021) Characterization and functional analysis of the Hydroxycinnamoyl-CoA: shikimate hydroxycinnamoyl transferase (HCT) gene family in poplar. PeerJ 9:e10741. https://doi.org/10.7717/peerj.10741
Chen Z, Yan W, Sun L, Tian J, Liao H (2016) Proteomic analysis reveals growth inhibition of soybean roots by manganese toxicity is associated with alteration of cell wall structure and lignification. J Proteom 143:151–160. https://doi.org/10.1016/j.jprot.2016.03.037
Chunquan Z, Wenjun H, Xiaochuang C et al (2021) Physiological and proteomic analyses reveal effects of putrescine-alleviated aluminum toxicity in rice roots. Rice Sci 28(6):579–593. https://doi.org/10.1016/j.rsci.2021.03.002
Dai X, Jia C, Lu J, Yu Z (2024) Metabolism of phenolic compounds and antioxidant activity in different tissue parts of post-harvest chive (Allium schoenoprasum L.). Antioxidants 13(3):279. https://doi.org/10.3390/antiox13030279
Danh LT, Truong P, Mammucari R, Tran T, Foster N (2009) Vetiver grass, Vetiveria zizanioides: a choice plant for phytoremediation of heavy metals and organic wastes. Int J Phytorem 11(8):664–691. https://doi.org/10.1080/15226510902787302
Dorafshan MM, Abedi-Koupai J, Eslamian S, Amiri MJ (2023) Vetiver grass (Chrysopogon zizanoides L.): A hyper-accumulator crop for bioremediation of unconventional water. Sustainability 15(4):3529. https://doi.org/10.3390/su15043529
El-Beltagi HS, Sofy MR, Aldaej MI, Mohamed HI (2020) Silicon alleviates copper toxicity in flax plants by up-regulating antioxidant defense and secondary metabolites and decreasing oxidative damage. Sustainability 12(11):4732. https://doi.org/10.3390/su12114732
Feil SB, Pii Y, Valentinuzzi F, Tiziani R, Mimmo T, Cesco S (2020) Copper toxicity affects phosphorus uptake mechanisms at molecular and physiological levels in Cucumis sativus plants. Plant Phys Biochem 157:138–147. https://doi.org/10.1016/j.plaphy.2020.10.023
Ganie SA, Molla KA, Henry RJ, Bhat KV, Mondal TK (2019) Advances in understanding salt tolerance in rice. Theor Appl Genet 132:851–870. https://doi.org/10.1007/s00122-019-03301-8
Giannakoula A, Therios I, Chatzissavvidis C (2021) Effect of lead and copper on photosynthetic apparatus in citrus (Citrus aurantium L.) plants. The role of antioxidants in oxidative damage as a response to heavy metal stress. Plants 10(1):155. https://doi.org/10.3390/plants10010155
Halim N S A, Hamzah N, Baharudin F et al. (2023) Improving iron and copper uptake by changing the ratios in root of vetiver grass. IOP Conference Series: Earth and Environmental Science 1216(1): 012041. https://doi.org/10.1088/1755-1315/1216/1/012041
Hossain MA, Li ZG, Hoque TS, Burritt DJ, Fujita M, Munné-Bosch S (2018) Heat or cold priming-induced cross-tolerance to abiotic stresses in plants: key regulators and possible mechanisms. Protoplasma 255:399–412. https://doi.org/10.1007/s00709-017-1150-8
Hu Z, Zhao C, Li Q, Feng Y, Zhang X, Lu Y, Ying R, Yin A, Ji W (2023) Heavy metals can affect plant morphology and limit plant growth and photosynthesis processes. Agronomy 13(10):2601. https://doi.org/10.3390/agronomy13102601
Huang G, Guo G, Yao S, Zhang N, Hu H (2016) Organic acids, amino acids compositions in the root exudates and Cu-accumulation in castor (Ricinus communis L.) under Cu stress. Int J Phytorem 18(1):33–40. https://doi.org/10.1080/15226514.2015.1058333
Ingram PA, Malamy JE (2010) Root system architecture. In: Jean-Claude K, Michel D (eds) Advances in Botanical Research. Academic Press, Cambridge. https://doi.org/10.1016/B978-0-12-380868-4.00002-8
Jaswanthi N, Krishna MSR, Sahitya UL, Suneetha P (2019) Apoplast proteomic analysis reveals drought stress-responsive protein datasets in chilli (Capsicum annuum L.). Data Brief 25:104041. https://doi.org/10.1016/j.dib.2019.104041
Jin H, Xu M, Chen H, Zhang S, Han X, Tang Z, Sun R (2016) Comparative proteomic analysis of differentially expressed proteins in Amaranthus hybridus L. roots under cadmium stress. Water, Air, Soil Pollut 227:1–12. https://doi.org/10.1007/s11270-016-2914-z
Kohli SK, Khanna K, Bhardwaj R, Abd-Allah EF, Ahmad P, Corpas FJ (2019) Assessment of subcellular ROS and NO metabolism in higher plants: multifunctional signaling molecules. Antioxidants 8(12):641. https://doi.org/10.3390/antiox8120641
Koobaz P, Ghaffari MR, Heidari M et al (2020) Proteomic and metabolomic analysis of desiccation tolerance in wheat young seedlings. Plant Phys Biochem 146:349–362. https://doi.org/10.1016/j.plaphy.2019.11.017
Kováč J, Lux A, Vaculík M (2018) Formation of a subero-lignified apical deposit in root tip of radish (Raphanus sativus) as a response to copper stress. Ann Bot 122(5):823–831. https://doi.org/10.1093/aob/mcy013
Li F, Shi J, Shen C, Chen G, Hu S, Chen Y (2009) Proteomic characterization of copper stress response in Elsholtzia splendens roots and leaves. Plant Mol Biol 71:251–263. https://doi.org/10.1007/s11103-009-9521-y
Li Z, Cheng B, Zeng W, Zhang X, Peng Y (2020a) Proteomic and metabolomic profilings reveal crucial functions of γ-aminobutyric acid in regulating ionic, water, and metabolic homeostasis in creeping bentgrass under salt stress. J Proteome Res 19(2):769–780. https://doi.org/10.1021/acs.jproteome.9b00627
Li Z, Zeng W, Cheng B, Huang T, Peng Y, Zhang X (2020b) γ-Aminobutyric acid enhances heat tolerance associated with the change of proteomic profiling in creeping bentgrass. Molecules 25(18):4270. https://doi.org/10.3390/molecules25184270
Li XY, Lin ML, Lu F, Zhou X, Xiong X, Chen LS, Huang ZR (2023) Physiological and ultrastructural responses to excessive-copper-induced toxicity in two differentially copper tolerant Citrus species. Plants 12(2):351. https://doi.org/10.3390/plants12020351
Liang K, Liang S, Zhu H (2020) Comparative proteomics analysis of the effect of selenium treatment on the quality of foxtail millet. Lwt 131:109691. https://doi.org/10.1016/j.lwt.2020.109691
Litterer LA, Plaisance KL, Schnurr JA, Storey KK, Jung HJG, Gronwald JW, Somers DA (2006) Biosynthesis of UDP-glucuronic acid in developing soybean embryos: possible role of UDP-sugar pyrophosphorylase. Phys Plant 128(2):200–211. https://doi.org/10.1111/j.1399-3054.2006.00728.x
Liu SJ, Xu HH, Wang WQ et al (2016) Identification of embryo proteins associated with seed germination and seedling establishment in germinating rice seeds. J Plant Phys 196:79–92. https://doi.org/10.1016/j.jplph.2016.02.021
Maes D, Finke A, Smallwood C et al (2022) Cellular photosynthetic pigment and structural change in Festuca arundinacea (Tall Fescue) after exposure to acute and chronic chromium and copper stress. Adv Environ Eng Res 3(2):1–28. https://doi.org/10.21926/aeer.2202025
Mansoor S, Ali Wani O, Lone JK, Manhas S, Kour N, Alam P, Ahmad A, Ahmad P (2022) Reactive oxygen species in plants: from source to sink. Antioxidants 11(2):225. https://doi.org/10.3390/antiox11020225
Minen RI, Martínez MP, Iglesias AA, Figueroa CM (2020) Biochemical characterization of recombinant UDP-sugar pyrophosphorylase and galactinol synthase from Brachypodium distachyon. Plant Phys Biochem 155:780–788. https://doi.org/10.1016/j.plaphy.2020.08.030
Nayak AK, Panda SS, Basu A, Dhal NK (2018) Enhancement of toxic Cr (VI), Fe, and other heavy metals phytoremediation by the synergistic combination of native Bacillus cereus strain and Vetiveria zizanioides L. Int J Phytorem 20(7):682–691. https://doi.org/10.1080/15226514.2017.1413332
Neumann D, Zur Nieden U, Lichtenberger O, Leopold I (1995) How does Armeria maritima tolerate high heavy metal concentrations? J Plant Phys 146(5–6):704–717. https://doi.org/10.1016/S0176-1617(11)81937-1
Pawlak-Sprada S, Arasimowicz-Jelonek M, Podgórska M, Deckert J (2011) Activation of phenylpropanoid pathway in legume plants exposed to heavy metals. Part I. Effects of cadmium and lead on phenylalanine ammonia-lyase gene expression, enzyme activity and lignin content. Acta Biochim Polon 58(2):211–216. https://doi.org/10.18388/abp.2011_2267
Peng W, Ming QL, Zhai X et al (2019) Polysaccharide fraction extracted from endophytic fungus Trichoderma atroviride D16 has an influence on the proteomics profile of the Salvia miltiorrhiza hairy roots. Biomolecules 9(9):415. https://doi.org/10.3390/biom9090415
Qiao F, Kong LA, Peng H et al (2019) Transcriptional profiling of wheat (Triticum aestivum L.) during a compatible interaction with the cereal cyst nematode Heterodera avenae. Sci Rep 9(1):2184. https://doi.org/10.1038/s41598-018-37824-9
Rafique S (2019) Differential expression of leaf proteome of tolerant and susceptible maize (Zea mays L.) genotypes in response to multiple abiotic stresses. Biochem Cell Biol 97(5):581–588. https://doi.org/10.1139/bcb-2018-0338
Sham A, Moustafa K, Al-Shamisi S, Alyan S, Iratni R, AbuQamar S (2017) Microarray analysis of Arabidopsis WRKY33 mutants in response to the necrotrophic fungus Botrytis cinerea. PLoS ONE 12(2):e0172343. https://doi.org/10.1371/journal.pone.0172343
Sharma R, Bhardwaj R, Thukral AK, Al-Huqail AA, Siddiqui MH, Ahmad P (2019) Oxidative stress mitigation and initiation of antioxidant and osmoprotectant responses mediated by ascorbic acid in Brassica juncea L. subjected to copper (II) stress. Ecotoxicol Environ Safety 182:109436. https://doi.org/10.1016/j.ecoenv.2019.109436
Šimon M, Shen ZJ, Ghoto K et al (2021) Proteomic investigation of Zn-challenged rice roots reveals adverse effects and root physiological adaptation. Plant Soil 460:69–88. https://doi.org/10.1007/s11104-020-04772-1
Singhania RR, Patel AK, Sukumaran RK, Larroche C, Pandey A (2013) Role and significance of beta-glucosidases in the hydrolysis of cellulose for bioethanol production. Bioresour Technol 127:500–507. https://doi.org/10.1016/j.biortech.2012.09.012
Souza VL, de Almeida AAF, de Souza JS et al (2014) Altered physiology, cell structure, and gene expression of Theobroma cacao seedlings subjected to Cu toxicity. Environ Sci Pollut Res 21:1217–1230. https://doi.org/10.1007/s11356-013-1983-4
Thounaojam TC, Panda P, Mazumdar P, Kumar D, Sharma GD, Sahoo L, Sanjib P (2012) Excess copper induced oxidative stress and response of antioxidants in rice. Plant Physiol Biochem 53:33–39. https://doi.org/10.1016/j.plaphy.2012.01.006
Wu S, Cao G, Adil MF et al (2020) Changes in water loss and cell wall metabolism during postharvest withering of tobacco (Nicotiana tabacum L.) leaves using tandem mass tag-based quantitative proteomics approach. Plant Physiol Biochem 150:121–132. https://doi.org/10.1016/j.plaphy.2020.02.040
Yang Y, Saand MA, Abdelaal WB et al (2020) iTRAQ-based comparative proteomic analysis of two coconut varieties reveals aromatic coconut cold-sensitive in response to low temperature. J Proteom 220:103766. https://doi.org/10.1016/j.jprot.2020.103766
Zhang L, Pan Y, Lv W, Xiong ZT (2014) Physiological responses of biomass allocation, root architecture, and invertase activity to copper stress in young seedlings from two populations of Kummerowia stipulacea (maxim.) Makino. Ecotoxicol Environ Safety 104:278–284. https://doi.org/10.1016/j.ecoenv.2014.03.013
Zhang Z, Ke M, Qu Q et al (2018) Impact of copper nanoparticles and ionic copper exposure on wheat (Triticum aestivum L.) root morphology and antioxidant response. Environ Pollut 239:689–697. https://doi.org/10.1016/j.envpol.2018.04.066
Zhou X, Fu L, Xia Y, Zheng L, Chen C, Shen Z, Chen Y (2017) Arbuscular mycorrhizal fungi enhance the copper tolerance of Tagetes patula through the sorption and barrier mechanisms of intraradical hyphae. Metallomics 9(7):936–948. https://doi.org/10.1039/c7mt90028g
Zhu LC, Jing SU, Jin YR, Zhao HY, Tian XC, Zhang C, Li MJ (2021) Genome-wide identification, molecular evolution, and expression divergence of the hexokinase gene family in apple. J Integr Agric 20(8):2112–2125. https://doi.org/10.1016/S2095-3119(20)63562-6
Funding
This work was supported by the National Natural Science Foundation of China (31500451) and the Science and Technology Cooperation Support Project of Guizhou Province ([2020]1Y046).
Author information
Authors and Affiliations
Contributions
DL and HW conceived and designed the experiments; DL, HW, and TY performed the experiments; DL, HW, and WX collected and analyzed the data; SL and WC projected the management; DL wrote the manuscript. WC revised the manuscript. All authors contributed to the final draft of the manuscript. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Handling Editor: Puneet Singh Chauhan.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, D., Wang, H., Gu, T. et al. Effects of Cu Stress on Root Growth and Proteomics of Vetiver (Vetiveria zizanioides L.). J Plant Growth Regul (2024). https://doi.org/10.1007/s00344-024-11324-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00344-024-11324-9