Abstract
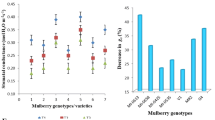
Identification of tolerant mulberry genotypes and elucidation of stress-tolerant mechanisms are two priority areas need to be addressed for sustainable mulberry leaf production. In the present study, 16 mulberry genotypes were evaluated under at low (75% FC), medium (50% FC), and high moisture stress (25% FC) conditions along with well-irrigated plants (100% FC) and measured the photosynthetic efficiency and antioxidant potential. In well-irrigated plants, photosynthetic rate was high in S13 (16.45 µmol/m2/s), Mysore Local, RC2, and AGB8 (13.62–16 µmol/m2/s). All genotypes showed a gradual decline in photosynthetic rate during the progression of moisture stress (low to high stress). MSG2, AGB8, RC1, RC2, and Nagalur Estate showed high photosynthetic rate (7.45–10.15 µmol/m2/s) under high moisture stress, whereas low photosynthetic rate was recorded in Mysore Local (3.46 µmol/m2/s) and K2 (3.50 µmol/m2/s). All tolerant genotypes showed higher activities of SOD (104.25–122.44 Units/g) and POX (25.61–33.73 Units/g) activities under high moisture stress. Bundi-3, S34, RC2, and AGB8 showed higher SOD activity and low enzyme activity was observed in K2. Non-enzymatic antioxidants and osmolytes were high in moisture stress-tolerant genotypes. This study identified that high photosynthetic rate is one of the important physiological indicator for moisture stress tolerance in mulberry. Also higher activities of antioxidant enzymes (SOD and POX), presence of high quantity of non-enzymatic antioxidants (ascorbic acids, reduced glutathione, and phenols), and accumulation of osmolytes (proline and glycine betaine) are identified as stress tolerance biochemical markers in mulberry genotypes. Evaluation Index (EI) value was calculated and genotypes suitable for well-irrigated condition as well as for low, medium, and high moisture stress conditions were identified based on EI values.










Similar content being viewed by others
Abbreviations
- FC:
-
Field capacity
- SOD:
-
Superoxide dismutase
- POX:
-
Peroxidase
- EI:
-
Evaluation Index
References
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59:206–216
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free praline for water-stress studies. Plant Soil 39:205–207
Bohnert HJ, Nelson DE, Jensen RG (1995) Adaptations to environmental stresses. Plant Cell 7(7):1099–1111
Bray EA, Bailey-Serres J, Weretilnyk E (2000) Responses to abiotic stresses. In: Gruissem W, Buchannan B, Jones R (eds) Biochemistry and molecular biology of plants. American Society of Plant Physiologists, Rockville, pp 1158–1249
Burg MB, Ferraris JD (2008) Intracellular organic osmolytes: function and regulation. J Biol Chem 283:7309–7313
Cattivelli L, Rizza F, Badeck FW, Mazzucotelli E, Mastrangelo EM, Francia E, Mare C, Tondelli A, Stanca AM (2008) Drought tolerance improvement in crop plants: an integrated view from breeding to genomics. Field Crop Res 105:1–14
Chaves MM, Oliveira MM (2004) Mechanisms underlying plant resilience to water deficits: prospects for water saving agriculture. J Exp Bot 55:2365–2384
Chinnusamy V, Zhu J, Zhu JK (2007) Cold stress regulation of gene expression in plants. Trends Plant Sci 12:444–451
Dhindsa RA, Plumb-Dhindsa P, Thorpe TA (1981) Leaf senescence: correlated with increased permeability and lipid peroxidation and decreased levels of superoxide dismutase and catalase. J Exp Bot 126:93–101
Diamant S, Eliahu N, Rosenthal D, Goloubinoff P (2001) Chemical chaperones regulate molecular chaperones in vitro and in cells under combined salt and heat stresses. J Biol Chem 276:39586–39591
Dubois MK, Giller KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 26:350–356
Fang Y, Xiong L (2015) General mechanisms of drought response and their application in drought resistance improvement in plants. Cell Mol Life Sci 72(4):673–689
Farooq M, Aziz T, Basra SMA, Cheema MA, Rehamn H (2008) Chilling tolerance in hybrid maize induced by seed priming with salicylic acid. J Agron Crop Sci 194:161–168
Fazeli F, Ghorbanli M, Niknam V (2007) Effect of drought on biomass, protein content, lipid peroxidation and antioxidant enzymes in two sesame cultivars. Biol Plant 51:98–103
Flagella Z, Giuliani MM, Rotunno T, DiCaterina R, DeCaro A (2004) Effect of saline water on oil yield and quality of a high oleic sunflower (Helianthus annuus L.) hybrid. Eur J Agron 21:267–272
Foyer CH, Noctor G (2005) Oxidant and antioxidant signaling in plants: a re-evaluation of the concept of oxidative stress in a physiological context. Plant Cell Environ 28:1056–1071
Giri J (2011) Glycine betaine and abiotic stress tolerance in plants. Plant Signal Behav 6:1746–1751
Grieve CM, Grattan SR (1983) Rapid assay for determination of water soluble quarternary ammonium compounds. Plant Soil 70:303–307
Guha A, Rasineni GK, Reddy AR (2010) Drought tolerance in mulberry (Morus spp.): a physiological approach with insights into growth dynamics and leaf yield production. Exp Agric 46(4):471–488
Guha A, Sengupta D, Rasineni GK, Reddy AR (2011) Non-enzymatic antioxidative defence in drought-stressed mulberry (Morus indica L.) genotypes. Trees. https://doi.org/10.1007/s00468-011-0665-4
Hare RD, Cress WA (1997) Metabolic implications of stress induced proline accumulation in plants. Plant Growth Regul 21:79–102
Hare PD, Cress WA, Van Staden J (1998) Dissecting the roles of osmolyte accumulation during stress. Plant Cell Environ 21:535–553
Hasanuzzaman M, Nahar K, Hossain MS, Al Mahmud J, Rahman A, Inafuku M, Oku H, Fujita M (2017) Coordinated actions of glyoxalase and antioxidant defense systems in conferring abiotic stress tolerance in plants. Int J Mol Sci 18:200. https://doi.org/10.3390/ijms18010200
Hong-Bo S, Li-Ye C, Ming-An S, Jaleel CA, Hong-Mei M (2008) Higher plant antioxidants and redox signaling under environmental stresses. C R Biol 331:433–441
Jaleel CA, Manivannan P, Wahidi A, Ferooq M, Jasim Al-Juburi H, Somasundaram R, Panneerselvam R (2009) Drought stress in plants: a review on morphological characteristics and pigments composition. Int J Agric Biol 11:100–105
Koul A, Sing D, Gupta SP (1996) Seasonal fluctuations in leaf moisture characters and dry matter in mulberry in subtropics. J Seric Sci 4:23–27
Mahajan S, Tuteja N (2005) Cold, salinity and drought stresses: an overview. Arch Biochem Biophys 444:139–158
Mahimasanthi A, Vedavyasa V, Morrison MN, Sivaprasasd V (2018) Study on drought management practices in mulberry sericulture in dry agro climatic zones of Karnataka State, India. Int Res J Biol Sci 7(3):24–28
Malick CP, Singh MB (1980) Plant enzymology and histo-enzymology. Kalyani, New Delhi
Mano Y, Nirmal Kumar S, Basavaraj HK, MalReddy N, Datta RK (1993) A new method to select promising silkworm breeds/combinations. Indian Silk 31:53
McNeil SD, Nuccio ML, Hanson AD (1999) Betaines and related osmoprotectants. Targets for metabolic engineering of stress resistance. Plant Physiol 123:371–380
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Moran JF, Becana M, Iturbe-Ormaetxe I, Frechilla S, Klucas RV, Aparicio-Tejo P (1994) Drought induces oxidative stress in pea plants. Planta 194:346–352
Moron MS, Depierre JW, Mannervik B (1979) Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. Biochim Biophys Acta 582:67–78
Mukharjee SP, Choudhari MA (1983) Implication of water stress-induced changes in the levels of endogenous ascorbic acid and hydrogen peroxide in Vigna seedlings. Physiol Plant 58:166–170
Putter J (1974). In: Bergmeyer HU (ed) Methods in enzymatic analysis, vol 2. Academic, New York, p 685
Rajput VD, Harish SRK, Verma KK, Sharma L, Quiroz-Figueroa FR, Meena M, Gour VS, Minkina T, Sushkova S, Mandzhieva S (2021) Recent developments in enzymatic antioxidant defence mechanism in plants with special reference to abiotic stress. Biology. https://doi.org/10.3390/biology10040267
Rao AA (2002) Conservation status of mulberry genetic resources in India. In: Paper contributed to expert consultation on promotion of global exchange of sericultural germplasm resources, satellite session of 19th ISC Congress, 2002, Bangkok, Thailand, pp 21–25
Shao H-B, Chu L-Y, Lu Z-H, Kang C-M (2008) Primary antioxidant free radical scavenging and redox signalling pathways in higher plant cells. Int J Biol Sci 4:8–14
Shvaleva AL, Silva FCE, Breia E, Jouve J, Hausman JF, Ameida MH, Maroco JP, Rodrigues ML, Pereira JS, Chaves MM (2006) Metabolic responses to water deficit in two Eucalyptus globulus clones with contrasting drought sensitivity. Tree Physiol 26(2):239–248
Sofo A, Dichio B, Xiloyannis C, Masia A (2004) Antioxidant defenses in olive trees during drought stress: changes in activity of some antioxidant enzymes. Funct Plant Biol 32(1):45–53
Xiang C, Werner BL, Christensen EM, Oliver DJ (2001) The biological functions of glutathione revisited in Arabidopsis transgenic plants with altered glutathione levels. Plant Physiol 126:564–574
Yadav S, Irfan M, Ahmad A, Hayat S (2011) Causes of salinity and plant manifestations to salt stress: a review. J Environ Biol 32(5):667–685
Yancey PH (2005) Organic osmolytes as compatible, metabolic and counteracting cytoprotectants in high osmolarity and other stresses. J Exp Biol 208:2819–2830
Yildiz-Aktas L, Dagnon S, Gurel A, Gesheva E, Edreva A (2009) Drought tolerance in cotton: involvement of non-enzymatic ROS-scavenging compounds. J Agron Crop Sci 195(4):247–253
Yin C, Peng Y, Zang R, Zhu Y, Li C (2005) Adaptive responses of Populus kangdingensis to drought stress. Physiol Plant 123(4):445–451
Zhao W, Liu L, Shen Q, Yang J, Han X, Tian F, Wu J (2020) Effects of water stress on photosynthesis, yield and water use efficiency in winter wheat. Water 12:1–19
Acknowledgements
The authors are thankful to Central Silk Board, Bengaluru, India for proving facilities and fund for carrying out the experiments under the Project PIP 3592.
Author information
Authors and Affiliations
Contributions
Thulasy Gayathri: raised saplings of short-listed genotypes in nursery, maintained the experimental plants under varied moisture stress conditions in pots, carried out physio-biochemical analysis, data analysis, and wrote the research paper for publication. Rajashekar Krishnan: involved in short-listing of genotypes for the experiments and provided useful suggestions. Subrahmaniam Gandhi Doss: involved in the designing of experiments and provided suggestions. Tanmoy Sarkar: involved in the experiments. Babu Lal: provided suggestions/information for the research paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflict of interest regarding the publication of this article.
Additional information
Handling Editor: Vijay Pratap Singh.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gayathri, T., Krishnan, R., Doss, S.G. et al. Evaluation of Mulberry Genotypes Under Moisture Stress for the Identification of Physio-biochemical Markers of Stress Tolerance. J Plant Growth Regul (2024). https://doi.org/10.1007/s00344-024-11241-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00344-024-11241-x