Abstract
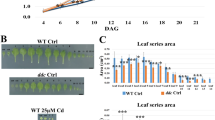
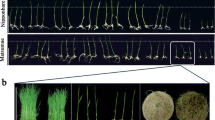
DNA methylation is a kind of epigenetic modification regulated by DNA methyltransferases and DNA demethylases. Here, the influences of DNA methylation inhibitor 5-aza-2'-deoxycytidine (5-azadC) combined with plant hormones [abscisic acid (ABA), methyl jasmonate (MeJA), and gibberellic acid (GA3)] and small gaseous signal molecules [(nitric oxide (NO), hydrogen sulfide (H2S), and hydrogen gas (H2)] on the tomato (Solanum lycopersicum) seedlings growth were explored. Moreover, the expression levels of DNA methyltransferases and demethylases under plant hormones and small gaseous molecules were detected. We found that ABA and MeJA restrained tomato seedling growth, and GA3, NO, H2S, and H2 promoted seedling growth. Nevertheless, 5-azadC treatment retarded the effects of these plant growth regulators mentioned above. Moreover, ABA boosted SlDRM5 expression, whereas GA3 and MeJA treatments inhibited the expression. Sodium hydrosulfide and hydrogen-rich water treatments affected SlDRM5 expression. Moreover, MeJA increased the quantity of SlDML1 transcripts from 3 to 24 h, while other therapies decreased the quantity. It is interesting to note that GA3 expressively induced SlMET1 and SlDML2 expression. Hence, DNA methylation may be essential for plant growth and is controlled by phytohormones and gaseous signal channels. The findings above offer fresh perspectives on how DNA methylation inhibitor reacts to different plant growth regulators.











Similar content being viewed by others
References
Ageeva-Kieferle A, Rudolf EE, Lindermayr C (2019) Redox-dependent chromatin remodeling: a new function of nitric oxide as architect of chromatin structure in plants. Front Plant Sci 10:625. https://doi.org/10.3389/fpls.2019.00625
Ai Y, Jing S, Cheng Z, Song B, Xie C, Liu J, Zhou J (2021) DNA methylation affects photoperiodic tuberization in potato (Solanum tuberosum L.) by mediating the expression of genes related to the photoperiod and GA pathways. Hortic Res 8(1):181. https://doi.org/10.1038/s41438-021-00619-7
Behera J, Kelly KE, Tyagi N (2021) Hydrogen sulfide prevents ethanol-induced ZO-1 CpG promoter hypermethylation-dependent vascular permeability via miR-218/DNMT3a axis. J Cell Physiol 236(10):6852–6867. https://doi.org/10.1002/jcp.30382
Bennett M, Cleaves K, Hewezi T (2021) Expression patterns of DNA methylation and demethylation genes during plant development and in response to phytohormones. Int J Mol Sci 22(18):9681. https://doi.org/10.3390/ijms22189681
Bertini L, Proietti S, Focaracci F, Sabatini B, Caruso C (2018) Epigenetic control of defense genes following MeJA-induced priming in rice (O. sativa). J Plant Physiol. https://doi.org/10.1016/j.jplph.2018.06.007
Browne L, Mead A, Horn C, Chang K, Celikkol AZ, Henriquez LC, Ma F, Beraut E, Meyer SR, Sork VL (2020) Experimental DNA demethylation associates with changes in growth and gene expression of oak tree seedlings. G3 10(3):1019–1028. https://doi.org/10.1534/g3.119.400770
Cao D, Ju Z, Gao C, Mei X, Fu D, Zhu H, Luo Y, Zhu B (2014) Genome-wide identification of cytosine-5 DNA methyltransferases and demethylases in Solanum lycopersicum. Gene 550(2):230–237. https://doi.org/10.1016/j.gene.2014.08.034
Chen K, Li GJ, Bressan RA, Song CP, Zhu JK, Zhao Y (2020) Abscisic acid dynamics, signaling, and functions in plants. J Integr Plant Biol 62(1):25–54. https://doi.org/10.1111/jipb.12899
Chowdhury S, Mukherjee A, Basak S, Das R, Mandal A, Kundu P (2022) Disruption of tomato TGS machinery by ToLCNDV causes reprogramming of vascular tissue-specific TORNADO1 gene expression. Planta 256(4):78. https://doi.org/10.1007/s00425-022-03985-1
Conde D, Moreno-Cortés A, Dervinis C, Ramos-Sánchez JM, Kirst M, Perales M, González-Melendi P, Allona I (2017) Overexpression of DEMETER, a DNA demethylase, promotes early apical bud maturation in poplar. Plant Cell Environ 40(11):2806–2819. https://doi.org/10.1111/pce.13056
Fenn MA, Giovannoni JJ (2021) Phytohormones in fruit development and maturation. Plant J 105(2):446–458. https://doi.org/10.1111/tpj.15112
Ghorbel M, Brini F, Sharma A, Landi M (2021) Role of jasmonic acid in plants: the molecular point of view. Plant Cell Rep 40(8):1471–1494. https://doi.org/10.1007/s00299-021-02687-4
Gong Z, Morales-Ruiz T, Ariza RR, Roldán-Arjona T, David L, Zhu JK (2002) ROS1, a repressor of transcriptional gene silencing in Arabidopsis, encodes a DNA glycosylase/lyase. Cell 111(6):803–814. https://doi.org/10.1016/S0092-8674(02)01133-9
Gu T, Ren S, Wang Y, Han Y, Li Y (2016) Characterization of DNA methyltransferase and demethylase genes in Fragaria vesca. Mol Genet Genom 291(3):1333–1345. https://doi.org/10.1007/s00438-016-1187-y
Guo Q, Feng X, Xue H, Teng X, Jin S, Duan X, Xiao L, Wu Y (2017) Maternal renovascular hypertensive rats treatment with hydrogen sulfide increased the methylation of AT1b Gene in offspring. Am J Hypertens 30(12):1220–1227. https://doi.org/10.1093/ajh/hpx124
Guo X, Xie Q, Li B, Su H (2020) Molecular characterization and transcription analysis of DNA methyltransferase genes in tomato (Solanum lycopersicum). Genet Mol Biol 43(1):e20180295. https://doi.org/10.1590/1678-4685-GMB-2018-0295
Gupta A, Hisano H, Hojo Y, Matsuura T, Ikeda Y, Mori IC, Senthil-Kumar M (2017) Global profiling of phytohormone dynamics during combined drought and pathogen stress in Arabidopsis thaliana reveals ABA and JA as major regulators. Sci Rep 7(1):4017. https://doi.org/10.1038/s41598-017-03907-2
Han ML, Yin J, Zhao YH, Sun XW, Meng JX, Zhou J, Shen T, Li HH, Zhang F (2020) How the color fades from Malus halliana flowers: transcriptome sequencing and DNA methylation analysis. Front Plant Sci 11:576054. https://doi.org/10.3389/fpls.2020.576054
He L, Huang H, Bradai M, Zhao C, You Y, Ma J, Zhao L, Lozano-Durán R, Zhu JK (2022) DNA methylation-free Arabidopsis reveals crucial roles of DNA methylation in regulating gene expression and development. Nat Commun 13(1):1335. https://doi.org/10.1038/s41467-022-28940-2
Huang D, Huo J, Liao W (2021) Hydrogen sulfide: roles in plant abiotic stress response and crosstalk with other signals. Plant Sci 302:110733. https://doi.org/10.1016/j.plantsci.2020.110733
Huo J, Huang D, Zhang J, Fang H, Wang B, Wang C, Liao W (2018) Hydrogen sulfide: a gaseous molecule in postharvest freshness. Front Plant Sci 9:1172. https://doi.org/10.3389/fpls.2018.01172
Kim JS, Lim JY, Shin H, Kim BG, Yoo SD, Kim WT, Huh JH (2019) ROS1-dependent DNA demethylation is required for ABA-inducible NIC3 expression. Plant Physiol 179(4):1810–1821. https://doi.org/10.1104/pp.18.01471
Klosinska M, Picard CL, Gehring M (2016) Conserved imprinting associated with unique epigenetic signatures in the Arabidopsis genus. Nat Plants 2:16145. https://doi.org/10.1038/nplants.2016.145
Köhler C, Wolff P, Spillane C (2012) Epigenetic mechanisms underlying genomic imprinting in plants. Annu Rev Plant Biol 63:331–352. https://doi.org/10.1146/annurev-arplant-042811-105514
Lang Z, Wang Y, Tang K, Tang D, Datsenka T, Cheng J, Zhang Y, Handa AK, Zhu JK (2017) Critical roles of DNA demethylation in the activation of ripening-induced genes and inhibition of ripening-repressed genes in tomato fruit. Proc Natl Acad Sci U S A 114(22):E4511–E4519. https://doi.org/10.1073/pnas.1705233114
Law JA, Jacobsen SE (2010) Establishing, maintaining and modifying DNA methylation patterns in plants and animals. Nat Rev Genet 11(3):204–220. https://doi.org/10.1038/nrg2719
Li S, Yang G (2015) Hydrogen sulfide maintains mitochondrial DNA replication via demethylation of TFAM. Antioxid Redox Signal 23(7):630–642. https://doi.org/10.1089/ars.2014.6186
Li W, Yong Y, Zhang Y, Lyu Y (2019) Transcriptional regulatory network of GA floral induction pathway in LA hybrid lily. Int J Mol Sci 20(11):2694. https://doi.org/10.3390/ijms20112694
Li Z, Hu Y, Chang M, Kashif MH, Tang M, Luo D, Cao S, Lu H, Zhang W, Huang Z, Yue J, Chen P (2021) 5-azacytidine pre-treatment alters DNA methylation levels and induces genes responsive to salt stress in kenaf (Hibiscus cannabinus L.). Chemosphere 271:129562. https://doi.org/10.1016/j.chemosphere.2021.129562
Liu H, Wang C, Li C, Zhao Z, Wei L, Liu Z, Hu D, Liao W (2022) Nitric oxide is involved in hydrogen sulfide-induced adventitious rooting in tomato (Solanum lycopersicum). Funct Plant Biol 49(3):245–258. https://doi.org/10.1071/FP21288
Liu R, How-Kit A, Stammitti L, Teyssier E, Rolin D, Mortain-Bertrand A, Halle S, Liu M, Kong J, Wu C, Degraeve-Guibault C, Chapman NH, Maucourt M, Hodgman TC, Tost J, Bouzayen M, Hong Y, Seymour GB, Giovannoni JJ, Gallusci P (2015) A DEMETER-like DNA demethylase governs tomato fruit ripening. Proc Natl Acad Sci U S A 112(34):10804–10809. https://doi.org/10.1073/pnas.1503362112
Liu R, Lang Z (2020) The mechanism and function of active DNA demethylation in plants. J Integr Plant Biol 62(1):148–159. https://doi.org/10.1111/jipb.12879
Madzima TF, Vendramin S, Lynn JS, Lemert P, Lu KC, McGinnis KM (2021) Direct and indirect transcriptional effects of abiotic stress in Zea mays plants defective in RNA-directed DNA methylation. Front Plant Sci 12:694289. https://doi.org/10.3389/fpls.2021.694289
Mageroy MH, Wilkinson SW, Tengs T, Cross H, Almvik M, Pétriacq P, Vivian-Smith A, Zhao T, Fossdal CG, Krokene P (2020) Molecular underpinnings of methyl jasmonate-induced resistance in Norway spruce. Plant Cell Environ 43(8):1827–1843. https://doi.org/10.1111/pce.13774
Massoumi M, Krens FA, Visser RG, De Klerk GM (2017) Azacytidine and miR156 promote rooting in adult but not in juvenile Arabidopsis tissues. J Plant Physiol 208:52–60. https://doi.org/10.1016/j.jplph.2016.10.010
Matzke MA, Mosher RA (2014) RNA-directed DNA methylation: an epigenetic pathway of increasing complexity. Nat Rev Genet 15(6):394–408. https://doi.org/10.1038/nrg3683
Miljkovic D, Cvetkovic I, Sajic M, Vuckovic O, Harhaji L, Markovic M, Trajkovic V (2004) 5-Aza-2’-deoxycytidine and paclitaxel inhibit inducible nitric oxide synthase activation in fibrosarcoma cells. Eur J Pharmacol 485(1–3):81–88. https://doi.org/10.1016/j.ejphar.2003.11.057
Ou XF, Zhuang TT, Yin WC, Miao YL, Wang B, Zhang YH, Lin XY, Xu CM, Wettstein D, Rustgi S, Liu B (2015) DNA methylation changes induced in rice by exposure to high concentrations of the nitric oxide modulator, sodium nitroprusside. Plant Mol Biol Rep 33(5):1428–1440. https://doi.org/10.1007/s11105-014-0843-9
Pavlopoulou A, Kossida S (2007) Plant cytosine-5 DNA methyltransferases: structure, function, and molecular evolution. Genomics 90(4):530–541. https://doi.org/10.1016/j.ygeno.2007.06.011
Rai KK, Rai N, Rai SP (2018) Salicylic acid and nitric oxide alleviate high temperature induced oxidative damage in Lablab purpureus L plants by regulating bio-physical processes and DNA methylation. Plant Physiol Biochem 128:72–88. https://doi.org/10.1016/j.plaphy.2018.04.023
Read A, Weiss T, Crisp PA, Liang Z, Noshay J, Menard CC, Wang C, Song M, Hirsch CN, Springer NM, Zhang F (2022) Genome-wide loss of CHH methylation with limited transcriptome changes in Setaria viridis DOMAINS REARRANGED METHYLTRANSFERASE (DRM) mutants. Plant J 111(1):103–116. https://doi.org/10.1111/tpj.15781
Ruan J, Zhou Y, Zhou M, Yan J, Khurshid M, Weng W, Cheng J, Zhang K (2019) Jasmonic acid signaling pathway in plants. Int J Mol Sci 20(10):2479. https://doi.org/10.3390/ijms20102479
Sallam N, Moussa M (2021) DNA methylation changes stimulated by drought stress in ABA-deficient maize mutant vp10. Plant Physiol Biochem 160:218–224. https://doi.org/10.1016/j.plaphy.2021.01.024
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 3(6):1101–1108. https://doi.org/10.1038/nprot.2008.73
Shi M, Wang C, Wang P, Zhang M, Liao W (2022) Methylation in DNA, histone, and RNA during flowering under stress condition: a review. Plant Sci 324:111431. https://doi.org/10.1016/j.plantsci.2022.111431
Sun M, Yang Z, Liu L, Duan L (2022) DNA methylation in plant responses and adaption to abiotic stresses. Int J Mol Sci 23(13):6910. https://doi.org/10.3390/ijms23136910
Sun S, Zhu J, Guo R, Whelan J, Shou H (2021) DNA methylation is involved in acclimation to iron deficiency in rice (Oryza sativa). Plant J 107(3):727–739. https://doi.org/10.1111/tpj.15318
Waadt R, Seller CA, Hsu PK, Takahashi Y, Munemasa S, Schroeder JI (2022) Plant hormone regulation of abiotic stress responses. Nat Rev Mol Cell Biol 23(10):680–694. https://doi.org/10.1038/s41580-022-00479-6
Wang C, Deng Y, Liu Z, Liao W (2021) Hydrogen sulfide in plants: crosstalk with other signal molecules in response to abiotic stresses. Int J Mol Sci 22(21):12068. https://doi.org/10.3390/ijms222112068
Wang C, Fang H, Gong T, Zhang J, Niu L, Huang D, Huo J, Liao W (2020) Hydrogen gas alleviates postharvest senescence of cut rose “Movie star” by antagonizing ethylene. Plant Mol Biol 102(3):271–285. https://doi.org/10.1007/s11103-019-00946-3
Wang C, Hou X, Qi N, Li C, Luo Y, Hu D, Li Y, Liao W (2022) An optimized method to obtain high-quality RNA from different tissues in Lilium davidii var. unicolor. Sci Rep 12(1):2825. https://doi.org/10.1038/s41598-022-06810-7
Wang L, Tan YJ, Wang M, Chen YF, Li XY (2019) DNA Methylation inhibitor 5-Aza-2’-deoxycytidine modulates endometrial receptivity through upregulating HOXA10 expression. Reprod Sc 26(6):839–846. https://doi.org/10.1177/1933719118815575
Wang P, Gao C, Bian X, Zhao S, Zhao C, Xia H, Song H, Hou L, Wan S, Wang X (2016) Genome-wide identification and comparative analysis of cytosine-5 DNA methyltransferase and demethylase families in wild and cultivated peanut. Front Plant Sci 7:7. https://doi.org/10.3389/fpls.2016.00007
Wei J, Shen Y, Dong X, Zhu Y, Cui J, Li H, Zheng G, Tian H, Wang Y, Liu Z (2022) DNA methylation affects freezing tolerance in winter rapeseed by mediating the expression of genes related to JA and CK pathways. Front Genet 13:968494. https://doi.org/10.3389/fgene.2022.968494
Wu K, Xu H, Gao X, Fu X (2021) New insights into gibberellin signaling in regulating plant growth-metabolic coordination. Curr Opin Plant Biol 63:102074. https://doi.org/10.1016/j.pbi.2021.102074
Xiao K, Chen J, He Q, Wang Y, Shen H, Sun L (2020a) DNA methylation is involved in regulation of pepper fruit ripening and interacts with phytohormones. J Exp Bot 71(6):1928–1942
Xiao K, Chen J, He Q, Wang Y, Shen H, Sun L (2020b) DNA methylation is involved in the regulation of pepper fruit ripening and interacts with phytohormones. J Exp Bot 71(6):1928–1942. https://doi.org/10.1093/jxb/eraa003
Yang X, Kundariya H, Xu YZ, Sandhu A, Yu J, Hutton SF, Zhang M, Mackenzie SA (2015) MutS HOMOLOG1-derived epigenetic breeding potential in tomato. Plant Physiol 168(1):222–232. https://doi.org/10.1104/pp.15.00075
Yang Y, Tang K, Datsenka TU, Liu W, Lv S, Lang Z, Wang X, Gao J, Wang W, Nie W, Chu Z, Zhang H, Handa AK, Zhu JK, Zhang H (2019) Critical function of DNA methyltransferase 1 in tomato development and regulation of the DNA methylome and transcriptome. J Integr Plant Biol 61(12):1224–1242. https://doi.org/10.1111/jipb.12778
Yao M, Chen W, Kong J, Zhang X, Shi N, Zhong S, Ma P, Gallusci P, Jackson S, Liu Y, Hong Y (2020) METHYLTRANSFERASE1 and ripening modulate vivipary during tomato fruit development. Plant Physiol 183(4):1883–1897. https://doi.org/10.1104/pp.20.00499
Yu M, Qin C, Li P, Zhang Y, Wang Y, Zhang J, Li D, Wang H, Lu Y, Xie K, Yu Y, Yu Y (2021a) Hydrogen gas alleviates sepsis-induced neuroinflammation and cognitive impairment through regulation of DNMT1 and DNMT3a-mediated BDNF promoter IV methylation in mice. Int Immunopharmacol 95:107583. https://doi.org/10.1016/j.intimp.2021.107583
Yu Z, Zhang G, Teixeira da Silva JA, Li M, Zhao C, He C, Si C, Zhang M, Duan J (2021b) Genome-wide identification and analysis of DNA methyltransferase and demethylase gene families in Dendrobium officinale reveal their potential functions in polysaccharide accumulation. BMC Plant Biol 21(1):21. https://doi.org/10.1186/s12870-020-02811-8
Yuan X, Ma K, Zhang M, Wang J, Zhang Q (2021) Integration of transcriptome and methylome analyses provides insight into the pathway of floral scent biosynthesis in Prunus mume. Front Genet 12:779557. https://doi.org/10.3389/fgene.2021.779557
Zanini D, Todorovic N, Korovljev D, Stajer V, Ostojic J, Purac J, Kojic D, Vukasinovic E, Djordjievski S, Sopic M, Guzonjic A, Ninic A, Erceg S, Ostojic SM (2021) The effects of 6-month hydrogen-rich water intake on molecular and phenotypic biomarkers of aging in older adults aged 70 years and over: a randomized controlled pilot trial. Exp Gerontol 155:111574. https://doi.org/10.1016/j.exger.2021.111574
Zeng W, Huang H, Lin X, Zhu C, Kosami KI, Huang C, Zhang H, Duan CG, Zhu JK, Miki D (2021) Roles of DEMETER in regulating DNA methylation in vegetative tissues and pathogen resistance. J Integr Plant Biol 63(4):691–706. https://doi.org/10.1111/jipb.13037
Zhang H, Lang Z, Zhu JK (2018) Dynamics and function of DNA methylation in plants. Nat Rev Mol Cell Biol 19(8):489–506. https://doi.org/10.1038/s41580-018-0016-z
Zhang Y, He X, Zhao H, Xu W, Deng H, Wang H, Wang S, Su D, Zheng Z, Yang B, Grierson D, Wu J, Liu M (2020) Genome-wide identification of DNA methylases and demethylases in kiwifruit (Actinidia chinensis). Front Plant Sci 11:514993. https://doi.org/10.3389/fpls.2020.514993
Zhong X, Du J, Hale CJ, Gallego-Bartolome J, Feng S, Vashisht AA, Chory J, Wohlschlegel JA, Patel DJ, Jacobsen SE (2014) Molecular mechanism of action of plant DRM de novo DNA methyltransferases. Cell 157(5):1050–1060. https://doi.org/10.1016/j.cell.2014.03.056
Zhu C, Zhang S, Zhou C, Chen L, Fu H, Li X, Lin Y, Lai Z, Guo Y (2020) Genome-wide investigation and transcriptional analysis of cytosine-5 DNA methyltransferase and DNA demethylase gene families in tea plant (Camellia sinensis) under abiotic stress and withering processing. PeerJ 8:e8432. https://doi.org/10.7717/peerj.8432
Funding
This study was supported by the National Key Research and Development Program of China (Grant No. 2018YFD1000800); the National Natural Science Foundation of China (Nos. 32072559, 31860568, 31560563, 31160398, and 32102370); the Natural Science Foundation of Gansu Province, China (Nos. 20JR5RA027, 1606RJZA073, 1606RJZA077, and 1606RJYA252); and the Fuxi Young Talents Fund of Gansu Agricultural University (No. Gaufx-03Y07).
Author information
Authors and Affiliations
Contributions
Conceptualization, WL; Formal analysis, ZL, CL, XP, and HZ; Funding acquisition, WL and CW; Investigation, CW, ZL, YY, and DH; Methodology, CW and ZL; Project administration, ZL and CW; Resources, ZL; Supervision, WL Writing of the original draft, ZL; Writing, reviewing, & editing of the manuscript, ZL, CW, and WL.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Handling editor: Natasa Bauer.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Z., Liu, C., Wang, C. et al. The Involvement of DNA Methylation in Plant Growth Regulators-Mediated Growth in Tomato (Solanum lycopersicum) Seedlings. J Plant Growth Regul 43, 1287–1303 (2024). https://doi.org/10.1007/s00344-023-11183-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-023-11183-w