Abstract
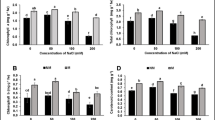
Water logging leads to reduction in soil oxygen, compromising the development and growth of the plant and thus results in serious yield losses. Enzymes involved in nitrogen metabolism play an important role during water logging. Therefore, this investigation was undertaken to evaluate nitrogen metabolism in maize and its wild progenitor (teosinte) under water-logging stress. Accessions of teosinte (Acc.1 and Acc.2) from two geographical locations and maize genotypes PMH-1 (relatively susceptible) and I-172 (relatively tolerant) were taken for the study. Teosinte accessions exhibited unique stress tolerance behavior by maintaining unaltered nitrate, nitrite, and nitric oxide (NO) contents in their shoots. Acc.2 revealed unaltered nitrate and NO contents in its roots during water logging. Acc.1 recorded highest nitrate, nitrite, and NO contents in its roots under stress conditions. I-172 seedlings also maintained unaltered nitrate and high NO contents under water-logging stress. The unaffected glutamate dehydrogenase (GDH) activity in hypoxic roots of I-172 suggested that it could maintain its glutamate pools under stress conditions. The seedlings of Acc.1 and Acc.2 remained unaffected with respect to their glutamine oxoglutarate amino transferase (GOGAT) activity during water logging. GOGAT activity was significantly reduced in water-logged maize genotypes but the values remained higher in the tolerant (I-172) as compared to the susceptible (PMH-1) genotype. The reduction in both GDH and GOGAT activities in PMH-1 roots might be responsible for the increased susceptibility of the genotype toward stress. Biplots of principal component analysis for the biochemical parameters showed that contents of nitrate, nitrite, and total free amino acids and activities of nitrate reductase (NR) and nitrite reductase (NiR) in hypoxic roots contributed significantly toward water-logging response of the plants. Acc.1, Acc.2, I-172 and PMH-1 seedlings showed differential behavior toward stress tolerance by occupying distinct positions on the graph. NR activity and content of crude proteins in shoots contributed significantly toward the variation in stress tolerance response of the genotypes.







Similar content being viewed by others
References
Aggarwal PK, Kalra N, Chander S, Pathak H (2006) InfoCrop: a dynamic simulation model for the assessment of crop yields, losses due to pests, and environmental impact of agroecosystems in tropical environments I. Model description. Agric Syst 89:1–25. https://doi.org/10.1016/j.agsy.2005.08.001
Ahsan N, Lee DG, Lee SH, Kang KY, Bahk JD, Choi MS, Lee IJ, Renaut J, Lee BH (2007) A comparative proteomic analysis of tomato leaves in response to waterlogging stress. Physiol Plant 131:555–570. https://doi.org/10.1111/j.1399-3054.2007.00980.x
AOAC (2005) Official methods of analysis. American Association of Official Agricultural Chemists, Washington DC
Bailey-Serres J, Voesenek LA (2008) Flooding stress: acclimations and genetic diversity. Annu Rev Plant Biol 59:313–339. https://doi.org/10.1146/annurev.arplant.59.032607.092752
Borrego-Benjumea A, Carter A, Tucker JR, Yao Z, Xu W, Badea A (2020) Genome-wide analysis of gene expression provides new insights into waterlogging responses in barley (Hordeum vulgare L.). Plants 9(2):240. https://doi.org/10.3390/plants9020240
Bulen WA (1956) The isolation and characterization of glutamic dehydrogenase from corn leaves. Arch Biochem Biophys 62:173–183. https://doi.org/10.1016/0003-9861(56)90100-x
Caravaca F, Alguacil MdM, Díaz G, Roldán A (2003) Use of nitrate reductase activity for assessing effectiveness of mycorrhizal symbiosis in Dorycnium pentaphyllum under induced water deficit. Commun Soil Sci Plant Anal 34:2291–2302. https://doi.org/10.1081/CSS-120024064
Cataldo DA, Maroon M, Schrader LE, Youngs VL (1975) Rapid colorimetric determination of nitrate in plant tissue by nitration of salicylic acid. Commun Soil Sci Plant Anal 6:71–80. https://doi.org/10.1080/00103627509366547
Colmer TD, Greenway H (2011) Ion transport in seminal and adventitious roots of cereals during O2 deficiency. J Exp Bot 62:39–57. https://doi.org/10.1093/jxb/erq271
Diab H, Cukier C (2013) Impact of waterlogging-induced hypoxia on nitrogen metabolism in the legume Medicago truncatula. Int J Life Sci Bt Pharm Res 2:2250–3137
Gupta KJ, Mur LA, Wany A, Kumari A, Fernie AR, Ratcliffe RG (2020) The role of nitrite and nitric oxide under low oxygen conditions in plants. New Phytol 225:1143–1151. https://doi.org/10.1111/nph.15969
Hageman RHG, Hucklesby DP (1971) Nitrate reductase from higher plants. Methods Enzymol 17:491–503. https://doi.org/10.1016/S0076-6879(71)23121-9
Hecht U, Oelmueller R, Schmidt S, Mohr H (1988) Action of light, nitrate and ammonium on the levels of NADH and ferredoxin dependent glutamate synthases in the cotyledon of mustard seedlings. Planta 175:130–138. https://doi.org/10.1007/bf00402890
Igamberdiev AU, Hill R (2004) Nitrate, NO and haemoglobin in plant adaptation to hypoxia: an alternative to classic fermentation pathways. J Exp Bot 55:2473–2482. https://doi.org/10.1093/jxb/erh272
Jaworski EG (1971) Nitrate reductase assay in intact plant tissues. Biochem Biophys Res Commun 43:1274–1279. https://doi.org/10.1016/S0006-291X(71)80010-4
Kanamori T, Matsumoto H (1972) Glutamine synthetase from rice roots. Arch Biochem Biophys 125:404–412. https://doi.org/10.1016/0003-9861(72)90230-5
Lee YP, Takahashi T (1966) An improved calorimetric determination of amino acids with the use of ninhydrin. Anal Biochem 14:71–77. https://doi.org/10.1016/0003-2697(66)90057-1
Limami AM, Diab H, Lothier J (2014) Nitrogen metabolism in plants under low oxygen stress. Planta 239:531–541. https://doi.org/10.1007/s00425-013-2015-9
Mammadov J, Buyyarapu R, Guttikonda SK, Parliament K, Abdurakhmonov IY, Kumpatla SP (2018) Wild relatives of maize, rice, cotton, and soybean: treasure troves for tolerance to biotic and abiotic stresses. Frontiers Plant Sci 9:886. https://doi.org/10.3389/fpls.2018.00886
Mano Y, Omori F (2007) Breeding for flooding tolerant maize using “teosinte” as a germplasm resource. Plant Root 1:17–21. https://doi.org/10.3117/plantroot.1.17
Oliveira HC, Freschi L, Sodek L (2013) Nitrogen metabolism and translocation in soybean plants subjected to root oxygen deficiency. Plant Physiol Biochem 66:141–149. https://doi.org/10.1016/j.plaphy.2013.02.015
Panozzo A, Cortivo CD, Ferrari M, Vicelli B, Varotto S, Vamerali T (2019) Morphological changes and expressions of AOX1A, CYP81D8, and putative PFP genes in a large set of commercial maize hybrids under extreme waterlogging. Front Plant Sci 10:62. https://doi.org/10.3389/fpls.2019.00062
Peng Y, Zhou Z, Zhang Z, Yu X, Zhang X, Du K (2018) Molecular and physiological responses in roots of two full-sib poplars uncover mechanisms that contribute to differences in partial submergence tolerance. Sci Rep 8:1–15. https://doi.org/10.1038/s41598-018-30821-y
Planchet E, Jagadis Gupta K, Sonoda M, Kaiser WM (2005) Nitric oxide emission from tobacco leaves and cell suspensions: rate limiting factors and evidence for the involvement of mitochondrial electron transport. Plant J 41:732–743. https://doi.org/10.1111/j.1365-313x.2005.02335.x
Qiu XM, Sun YY, Ye XY, Li ZG (2020) Signaling role of glutamate in plants. Front Plant Sci 10:1743. https://doi.org/10.3389/fpls.2019.01743
R Studio Team (2016). RStudio: Integrated Development for R. RStudio, Inc., Boston, MA. RStudio, Inc., Boston, MA. Retrieved from http://www.rstudio.com/. Accessed 11 Nov 2022
Ramirez JM, Del Compo FF, Peneque A, Lasoda M (1966) Ferredoxin-nitrite reductase from spinach. Biochem Biophys Acta 118:58–74. https://doi.org/10.1016/s0926-6593(66)80144-3
Ren B, Zhang J, Li X, Fan X, Dong S, Liu P, Zhao B (2014) Effects of waterlogging on the yield and growth of summer maize under field conditions. Can J Plant Sci 94:23–31. https://doi.org/10.4141/cjps2013-175
Ren B, Dong S, Zhao B, Liu P, Zhang J (2017a) Responses of nitrogen metabolism, uptake and translocation of maize to waterlogging at different growth stages. Front Plant Sci 8:1216. https://doi.org/10.3389/fpls.2017.01216
Ren B, Zhang J, Dong S, Liu P, Zhao B, Li H (2017b) Nitrapyrin improves grain yield and nitrogen use efficiency of summer maize waterlogged in the field. Agron J 109:185–192. https://doi.org/10.2134/agronj2016.06.0353
Ren B, Hu J, Liu P, Zhao B, Zhang J (2021) Responses of nitrogen efficiency and antioxidant system of summer maize to waterlogging stress under different tillage. PeerJ 9:e11834. https://doi.org/10.7717/peerj.11834
Sadak MSH, Abdelhamid MT, Schmidhalter U (2015) Effect of foliar application of aminoacids on plant yield and some physiological parameters in bean plants irrigated with seawater. Acta Biológica Colombiana 20:141–52. https://doi.org/10.1446/abc.v20n1.42865
Sánchez González JDJ, Ruiz Corral JA, Gracia GM, Ojeda GR, Larious LDIC, Holland JB, Medrano RM, Romero GEG (2018) Ecogeography of teosinte. PLoS ONE 13(2):e0192676. https://doi.org/10.1371/journal.pone.0192676
Sasidharan R, Hartman S, Liu Z, Martopawiro S, Sajeev N, van Veen H, Yeung E, Voesenek LAC (2018) Signal dynamics and interactions during flooding stress. Plant Physiol 176:1106–1117. https://doi.org/10.1104/pp.17.01232
Sauter M (2013) Root responses to flooding. Curr Opin Plant Biol 16:282–286. https://doi.org/10.1016/j.pbi.2013.03.013
Shabala S (2011) Physiological and cellular aspects of phytotoxicity tolerance in plants: the role of membrane transporters and implications for crop breeding for waterlogging tolerance. New Phytol 190:289–298. https://doi.org/10.1111/j.1469-8137.2010.03575.x
Statista (2021) Corn production worldwide 2019/2020, by country. www.statista.com/ Agriculture/Farming. Accessed 20-Jan-2021
Stoychev V, Simova-Stoilova L, Vaseva I, Kostadinova A, Nenkova R, Feller U, Demirevska K (2013) Protein changes and proteolytic degradation in red and white clover plants subjected to waterlogging. Acta Physiol Plant 35:1925–1932
Turner NC, Kramer PJ (1980) Adaptation of plants to water and high temperature stress. Wiley, New York, p 207
Voesenek L, Sasidharan R (2013) Ethylene–and oxygen signalling–drive plant survival during flooding. Plant Biol 15:426–435. https://doi.org/10.1111/plb.12014
Walne CH, Reddy KR (2021) Developing functional relationships between soil water logging and corn shoot and root growth and development. Plants 10:2095–2114. https://doi.org/10.3390/plants10102095
Westra S, Fowler HJ, Evans JP, Alexander LV, Berg P, Johnson F, Kendon EJ, Lenderink G, Roberts NM (2014) Future changes to the intensity and frequency of short-duration extreme rainfall. Rev Geophys 52:522–555. https://doi.org/10.1002/2014RG000464
Wu W, Wang S, Chen H, Song Y, Zhang L, Peng C, Jing L, Li J (2018) Optimal nitrogen regimes compensate for the impacts of seedlings subjected to waterlogging stress in summer maize. PLoS ONE 13(10):e0206210. https://doi.org/10.1371/journal.pone.0206210
Yamauchi T, Shimamura S, Nakazono M, Mochizuki T (2013) Aerenchyma formation in crop species: a review. Field Crops Res 152:8–16. https://doi.org/10.1016/j.fcr.2012.12.008
Yang WC, Lin KH, Wu CW, Chang YJ, Chang YS (2020) Effects of waterlogging with different water resources on plant growth and tolerance capacity of four herbaceous flowers in a bioretention basin. Water 12:1619. https://doi.org/10.3390/w12061619
Yu M, Zhou Z, Deng X, Li J, Mei F, Qi Y (2017) Physiological mechanism of programmed cell death aggravation and acceleration in wheat endosperm cells caused by waterlogging. Acta Physiol Plant 39:23. https://doi.org/10.1007/s11738-016-2324-2
Zhang Y, Liu G, Dong H, Li C (2021) Waterlogging stress in cotton: damage, adaptability, alleviation strategies, and mechanisms. Crop J 9(2):257–270. https://doi.org/10.1016/j.cj.2020.08.005
Zhou B, Guo Z, Xing J, Huang B (2005) Nitric oxide is involved in abscisic acid-induced antioxidant activities in Stylosanthes guianensis. J Exp Bot 56:3223–3228. https://doi.org/10.1093/jxb/eri319
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
KT performed all the experiments discussed in the manuscript. KK conceived the idea, designed the overall project, formulated the objectives, and interpreted the results critically for the manuscript. YS revised and finalized the manuscript. GK provided teosinte accessions and maize genotypes for performing the study.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Serena Varotto.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thukral, K., Kaur, K., Suneja, Y. et al. Distinctive Characteristics of Nitrogen Metabolism in Teosinte Versus Modern Maize for Water-Logging Tolerance. J Plant Growth Regul 42, 4193–4206 (2023). https://doi.org/10.1007/s00344-022-10883-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-022-10883-z