Abstract
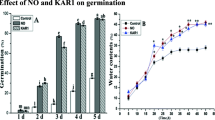
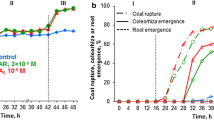
The relationship between ethylene and cyanide (HCN) and karrikin 1 (KAR1) in dormancy release was studied in secondary dormant Brassica oleracea L. (Chinese cabbage) seeds. Freshly harvested seeds of Brassica oleracea usually have poor germination potential. Karrikin1 (KAR1) and cyanide (HCN) are able to stimulate seed germination. However, the stimulatory effects of these two chemicals depend on the activation of the ethylene biosynthesis pathway and on ethylene perception. In this study, KAR1 and HCN application increased the activity of ethylene and of two ethylene biosynthesis enzymes, ACC synthase (ACS) and ACC oxidase (ACO). KAR1 and HCN collectively promoted the accumulation of 1 aminocyclopropane-1-carboxylic acid (ACC). In the presence of NO (nitric oxide) and KAR1, ACS and ACO activities reached their maximum levels after 36 and 42 h, respectively. Ethylene inhibitors suppressed seed germination by approximately 55%, whereas the respiratory inhibitors SHAM and NaN3 inhibited seed germination by 5–10% in the presence of HCN and KAR1. KAR1 and HCN collectively reduced the abscisic acid (ABA) content in seeds, increased the gibberellic acid (GA) content and released seed dormancy. The expression of ethylene biosynthesis genes and ethylene receptor genes (BOACO1, BOACS1, BOACS3, BOACS4, BOACS5, BOACS7, BOACS9, BOACS11, BOETR1 and BOETR2) provided further evidence of the involvement of ethylene in KAR1 and HCN-induced germination. BOACO1, BOACS1, BOACS5, BOACS7, BOACS9, BOACS11, BOETR1 and BOETR2 genes were up regulated in the presence of KAR1 and HCN, while the remaining genes were down regulated. The expression of various ethylene biosynthesis and ethylene receptor genes suggested functional diversification and variations in seed sensitivity in the presence of KAR1 and HCN. Therefore, in the current study, KAR1 and HCN application effectively induced the germination of B. oleracea seeds (approximately 97% germination rate) after 6 days by modifying the ethylene biosynthetic pathway.






Similar content being viewed by others
References
Anzala F, Morere-Le Paven MC, Fournier S, Rondeau D, Limami AM (2006) Physiological and molecular aspects of aspartate-derived amino acid metabolism during germination and post-germination growth in two maize genotypes differing in germination efficiency. J Exp Bot 57(3):645–653
Awan S, Footitt S, Finch-Savage WE (2018) Interaction of maternal environment and allelic differences in seed vigour genes determines seed performance in Brassica oleracea. Plant J 94(6):1098–1108
Babula D, Misztal LH, Jakubowicz M, Kaczmarek M, Nowak W, Sadowski J (2006) Genes involved in biosynthesis and signalisation of ethylene in Brassica oleracea and Arabidopsis thaliana: identification and genome comparative mapping of specific gene homologues. Theoret Appl Genet 112(3):410–420
Bethke PC, Libourel IG, Reinöhl V, Jones RL (2006) Sodium nitroprusside, cyanide, nitrite, and nitrate break Arabidopsis seed dormancy in a nitric oxide-dependent manner. Planta 223(4):805–812
Bewley JD (1997) Seed germination and dormancy. Plant Cell 9(7):1055
Bidonde S, Ferrer MA, Zegzouti H, Ramassamy S, Latché A, Pech JC, Hamilton AJ, Grierson D, Bouzayen M (1998) Expression and characterization of three tomato 1-aminocyclopropane-1-carboxylate oxidase cDNAs in yeast. Eur J Biochem 253(1):20–26
Binnie JE, McManus MT (2009) Characterization of the 1-aminocyclopropane-1-carboxylic acid (ACC) oxidase multigene family of Malus domestica Borkh. Phytochemistry 70(3):348–360
Bogatek R, Lewak S (1988) Effects of cyanide and cold treatment on sugar catabolism in apple seeds during dormancy removal [Malus domestica]. Physiol Plant 73:406
Bogatek R, Lewak S (1991) Cyanide controls enzymes involved in lipid and sugar catabolism in dormant apple embryos during culture. Physiol Plant 83(3):422–426
Bogatek R (1995) The possible role of fructose 2, 6-bisphosphate in the cyanide- mediated removal of embryonic dormancy in apple. Physiol Plant 94(3):460–464
Bogatek R, Côme D, Corbineau F, Picard MA, Żarska-Maciejewska B, Lewak S (1999) Sugar metabolism as related to the cyanide-mediated elimination of dormancy in apple embryos. Plant Physiol Biochem 37(7–8):577–585
Bogatek R, Sykala A, Krysiak C (2004) Cyanide-induced ethylene biosynthesis in dormant apple embryos. Acta Physiol Plant 26(3 suppl.):16–16
Booker MA, DeLong A (2015) Producing the ethylene signal: regulation and diversification of ethylene biosynthetic enzymes. Plant Physiol 169(1):42–50
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254
Cadman CS, Toorop PE, Hilhorst HW, Finch- Savage WE (2006) Gene expression profiles of Arabidopsis Cvi seeds during dormancy cycling indicate a common underlying dormancy control mechanism. Plant J 46(5):805–822
Cembrowska-Lech D, Koprowski M, Kępczyński J (2015) Germination induction of dormant Avena fatua caryopses by KAR1 and GA3 involving the control of reactive oxygen species (H2O2 and O2−) and enzymatic antioxidants (superoxide dismutase and catalase) both in the embryo and the aleurone layers. J Plant Physiol 176:169–179
Chen YF, Etheridge N, Schaller GE (2005) Ethylene signal transduction. Ann Bot 95(6):901–915
Concepcion M, Lizada C, Yang SF (1979) A simple and sensitive assay for 1-aminocyclopropane-1-carboxylic acid. Anal Biochem 100(1):140–145
Corbineau F, Xia Q, Bailly C, El-Maarouf-Bouteau H (2014) Ethylene, a key factor in the regulation of seed dormancy. Front Plant Sci 5:539
Ebbs SD, Kosma DK, Nielson EH, Machingura M, Baker AJ, Woodrow IE (2010) Nitrogen supply and cyanide concentration influence the enrichment of nitrogen from cyanide in wheat (Triticum aestivum L.) and sorghum (Sorghum bicolor L.). Plant Cell Environ 33(7):1152–1160
Esashi Y, Kusuyama K, Tazaki S, Ishihara N (1981) Necessity of a balance between CN-sensitive and CN-resistant respirations for germination of cocklebur seeds. Plant Cell Physiol 22(1):65–71
Finch-Savage WE, Leubner-Metzger G (2006) Seed dormancy and the control of germination. New Phytol 171(3):501–523
Feurtado JA, Kermode AR (2018) A merging of paths: abscisic acid and hormonal cross-talk in the control of seed dormancy maintenance and alleviation. Annu Plant Rev 27:176–223
Flematti GR, Ghisalberti EL, Dixon KW, Trengove RD (2004) A compound from smoke that promotes seed germination. Science 305(5686):977–977
Flematti GR, Ghisalberti EL, Dixon KW, Trengove RD (2009) Identification of alkyl substituted 2 H-furo [2, 3-c] pyran-2-ones as germination stimulants present in smoke. J Agric Food Chem 57(20):9475–9480
Flematti GR, Scaffidi A, Goddard-Borger ED, Heath CH, Nelson DC, Commander LE, Stick RV, Dixon KW, Smith SM, Ghisalberti EL (2010) Structure−activity relationship of karrikin germination stimulants. J Agric Food Chem 58(15):8612–8617
Gómez-Jiménez MDC, García-Olivares E, Matilla AJ (2001) 1-Aminocyclopropane-1 carboxylate oxidase from embryonic axes of germinating chick-pea (Cicer arietinum L.) seeds: cellular immunolocalization and alterations in its expression by indole-3-acetic acid, abscisic acid and spermine. Seed Sci Res 11(3):243–253
Gniazdowska A, Krasuska U, Bogatek R (2010a) Dormancy removal in apple embryos by nitric oxide or cyanide involves modifications in ethylene biosynthetic pathway. Planta 232(6):1397–1407
Gniazdowska A, Krasuska U, Czajkowska K, Bogatek R (2010b) Nitric oxide, hydrogen cyanide and ethylene are required in the control of germination and undisturbed development of young apple seedlings. Plant Growth Regul 61(1):75–84
Gu YQ, Wildermuth MC, Chakravarthy S, Loh YT, Yang C, He X, Han Y, Martin GB (2002) Tomato transcription factors Pti4, Pti5, and Pti6 activate defense responses when expressed in Arabidopsis. Plant Cell 14(4):817–831
Hilhorst HWM, Bradford K, Nonogaki H (2007) Seed development, dormancy and germination. Wiley, New York
Huang S, Gruber S, Stockmann F, Claupein W (2016) Dynamics of dormancy during seed development of oilseed rape (Brassica napus L.). Seed Sci Res 26(3):245–253
International Seed Testing Association (1999) International rules for seed testing. Rules 1999. Seed Science and Technology. CABI, Wallingford
Kendrick MD, Chang C (2008) Ethylene signaling: new levels of complexity and regulation. Curr Opin Plant Biol 11(5):479–485
Kepczynski J, Corbineau F, Côme D (1996) Responsiveness of Amaranthus retroflexus seeds to ethephon, 1-aminocyclopropane 1-carboxylic acid and gibberellic acid in relation to temperature and dormancy. Plant Growth Regul 20(3):259–265
KeÇpczyński J, KeÇpczyńska E (1997) Ethylene in seed dormancy and germination. Physiol Plant 101(4):720–726
Kępczyński J, Van Staden J (2012) Interaction of karrikinolide and ethylene in controlling germination of dormant Avena fatua L. caryopses. Plant Growth Regul 67(2):185–190
Kępczyński J, Cembrowska-Lech D, Van Staden J (2013) Necessity of gibberellin for stimulatory effect of KAR 1 on germination of dormant Avena fatua L. caryopses. Acta Physiol Plant 35(2):379–387
Kucera B, Cohn MA, Leubner-Metzger G (2005) Plant hormone interactions during seed dormancy release and germination. Seed Sci Res 15(4):281–307
Kuyucak N, Akcil A (2013) Cyanide and removal options from effluents in gold mining and metallurgical processes. Miner Eng 50:13–29
Locke JM, Bryce JH, Morris PC (2000) Contrasting effects of ethylene perception and biosynthesis inhibitors on germination and seedling growth of barley (Hordeum vulgare L.). J Exp Bot 51(352):1843–1849
Mathooko FM, Kubo Y, Inaba A, Nakamura R (1993) Partial characterization of 1-aminocyclopropane-1-carboxylate oxidase from excised mesocarp tissue of winter squash [Cucurbita moschata] fruit. Scientific Reports of the Faculty of Agriculture-Okayama University (Japan)
Matilla AJ (2000) Ethylene in seed formation and germination. Seed Sci Res 10(2):111–126
Matilla AJ, Matilla-Vázquez MA (2008) Involvement of ethylene in seed physiology. Plant Sci 175(1–2):87–97
McMahon Smith J, Arteca RN (2000) Molecular control of ethylene production by cyanide in Arabidopsis thaliana. Physiol Plant 109(2):180–187
Mindrebo JT, Nartey CM, Seto Y, Burkart MD, Noel JP (2016) Unveiling the functional diversity of the alpha/beta hydrolase superfamily in the plant kingdom. Curr Opin Struct Biol 41:233–246
Nagashima S (1977) Spectrophotometric determination of cyanide with γ-picoline and barbituric acid. Anal Chim Acta 91(2):303–306
Nelson DC, Riseborough JA, Flematti GR, Stevens J, Ghisalberti EL, Dixon KW, Smith SM (2009) Karrikins discovered in smoke trigger Arabidopsis seed germination by a mechanism requiring gibberellic acid synthesis and light. Plant Physiol 149(2):863–873
Oracz K, Bouteau HEM, Farrant JM, Cooper K, Belghazi M, Job C, Bailly C (2007) ROS production and protein oxidation as a novel mechanism for seed dormancy alleviation. Plant J 50(3):452–465
Oracz K, El-Maarouf-Bouteau H, Bogatek R, Corbineau F, Bailly C (2008) Release of sunflower seed dormancy by cyanide: cross-talk with ethylene signalling pathway. J Exp Bot 59(8):2241–2251
Oracz K, El-Maarouf-Bouteau H, Kranner I, Bogatek R, Corbineau F, Bailly C (2009) The mechanisms involved in seed dormancy alleviation by hydrogen cyanide unravel the role of reactive oxygen species as key factors of cellular signaling during germination. Plant Physiol 150(1):494–505
Peck SC, Kende H (1995) Sequential induction of the ethylene biosynthetic enzymes by indole-3-acetic acid in etiolated peas. Plant Mol Biol 28(2):293–301
Pierik R, Sasidharan R, Voesenek LA (2007) Growth control by ethylene: adjusting phenotypes to the environment. J Plant Growth Regul 26(2):188–200
Pirrello J, Jaimes-Miranda F, Sanchez-Ballesta MT, Tournier B, Khalil-Ahmad Q, Regad F, Latche A, Pech JC, Bouzayen M (2006) Sl-ERF2, a tomato ethylene response factor involved in ethylene response and seed germination. Plant Cell Physiol 47(9):1195–1205
Rahman MM, Banu LA, Rahman MM, Shahjadee UF (2007) Changes of the enzymes activity during germination of different mungbean varieties. Bangl J Sci Ind Res 42(2):213–216
Renata B, Agnieszka G (2006) Nitric oxide and HCN reduce deep dormancy of apple seeds. Acta Physiol Plant 28(3):281–287
Riov J, Yang SF (1982) Effects of exogenous ethylene on ethylene production in citrus leaf tissue. Plant Physiol 70(1):136–141
Ruduś I, Cembrowska-Lech D, Jaworska A, Kępczyński J (2018) Involvement of ethylene biosynthesis and perception during germination of dormant Avena fatua L. caryopses induced by KAR 1 or GA 3. Planta 1:1–20
Sami A, Riaz MW, Zhou X, Zhu Z, Zhou K (2019) Alleviating dormancy in Brassica oleracea seeds using NO and KAR1 with ethylene biosynthetic pathway, ROS and antioxidant enzymes modifications. BMC Plant Biol 19(1):1–15
Sami A, Shah FA, Abdullah M, Yu ZX, Yan Y, He ZZ, Jin ZK (2020) Melatonin mitigates cadmium and aluminum toxicity through modulation of antioxidant potential in Brassica napus L. Plant Biol. https://doi.org/10.1111/plb.13093
Shuai H, Meng Y, Luo X, Chen F, Zhou W, Dai Y, Qi Y, Du J, Yang F, Liu J, Yang W (2017) Exogenous auxin represses soybean seed germination through decreasing the gibberellin/abscisic acid (GA/ABA) ratio. Sci Rep 7(1):1–11
Schierle J, Rohwer F, Bopp M (1989) Distribution of ethylene synthesis along the etiolated pea shoot and its regulation by ethylene. J Plant Physiol 134(3):331–337
Siegień I, Bogatek R (2006) Cyanide action in plants—from toxic to regulatory. Acta Physiol Plant 28(5):483–497
Tonguc M, Elkoyunu R, Erbaş S, Karakurt Y (2012) Changes in seed reserve composition during germination and initial seedling development of safflower (Carthamus tinctorius L.). Turk J Biol 36(1):107–112
Van Staden J, Jager AK, Light ME, Burger BV (2004) Isolation of the major germination cue from plant-derived smoke. S Afr J Bot. https://doi.org/10.1016/S0254-6299(15)30206-4
Waters MT, Scaffidi A, Sun YK, Flematti GR, Smith SM (2014) The karrikin response system of Arabidopsis. Plant J 79(4):623–631
Yang SF, Hoffman NE (1984) Ethylene biosynthesis and its regulation in higher plants. Annu Rev Plant Physiol 35(1):155–189
Yip WK, Yang SF (1988) Cyanide metabolism in relation to ethylene production in plant tissues. Plant Physiol 88(2):473–476
Acknowledgments
The research was financially supported by the 13th Five-Year Plan for Rapeseed-Cotton Industry System of Anhui Province in China (AHCYJSTX-04) and the National Key Research & Development Program (2018YFD0100600).
Author information
Authors and Affiliations
Contributions
AS designed and performed the experiment. SR, AT and ZXY analyzed the data. ZZH and ZKJ provided the guidance during all experiments. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest that might be perceived to influence the results and discussion reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sami, A., Rehman, S., Tanvir, M.A. et al. Assessment of the Germination Potential of Brassica oleracea Seeds Treated with Karrikin 1 and Cyanide, Which Modify the Ethylene Biosynthetic Pathway. J Plant Growth Regul 40, 1257–1269 (2021). https://doi.org/10.1007/s00344-020-10186-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-020-10186-1