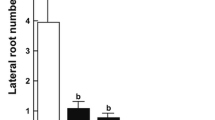
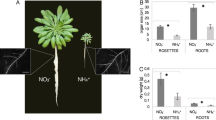
Abstract
Plants adapt to challenging growth conditions, such as the scarcity of nutrients or exposure to toxic metals, via changes in root system architecture. Chromium (Cr) is a non-essential element that when supplied in sublethal concentrations inhibits primary root growth through decreasing meristematic activity and affects photosynthesis. Here, we show that sucrose reverses the inhibitory effects of Cr(VI) on plant growth and development. Sucrose supplementation reactivated primary root growth under repressing Cr(VI) concentrations by restoring cell division and auxin distribution at the root meristem, keeping stem cell niche functioning. Analysis of the growth of Arabidopsis wild-type and mutant seedlings defective in auxin transport or signaling further revealed a critical role of auxin in mediating the effects of sucrose to protect plants from Cr(VI) toxicity. The results suggest that sucrose acts as a regulator in the maintenance of root meristem activity to overcome the stress generated by sublethal Cr(VI) concentrations.





Similar content being viewed by others
References
Aichinger E, Kornet N, Friedrich T, Laux T et al (2012) Plant stem cell niches. Annu Rev Plant Biol 63:615–636
Aida M, Beis D, Heidstra R, Willemsen V, Blilou I, et al (2004) The PLETHORA genes mediate patterning of the Arabidopsis root stem cell niche. Cell 119:109–120
Atkinson NJ, Urwin PE (2012) The interaction of plant biotic and abiotic stresses: from genes to the field. J Exp Bot 63:3523–3543
Benková E, Hejatko J (2009) Hormone interactions at the root apical meristem. Plant Mol Biol 69:383–396
Blilou I, Xu J, Wildwater M, Willemsen V, Paponov I, Friml J, Heidstra R, Aida M, Palme K, Scheres B (2005) The PIN auxin efflux facilitator network controls growth and patterning in Arabidopsis roots. Nature 433:39–44
Cervantes C, Campos-García J, Devars S, Gutiérrez-Corona F, Tavera HL, Torres-Guzmán JC, Moreno-Sánchez R (2001) Interactions of chromium with microorganisms and plants. FEMS Microbiol Rev 25:335–347
Chaudhuri B, Hörmann F, Lalonde S, Brady SM, Orlando DA, Benfey P, Frommer WB (2008) Protonophore-and pH-insensitive glucose and sucrose accumulation detected by FRET nanosensors in Arabidopsis root tips. Plant J 56:948–962
Chen LQ, Qu XQ, Hou BH, Sosso D, Osorio S, Fernie AR, Frommer WB (2012) Sucrose efflux mediated by SWEET proteins as a key step for phloem transport. Science 335:207–211
Colón-Carmona A, Yuo R, Haimovitch-Gal T, Doerner P (1999) Spatiotemporal analysis of mitotic activity with a labile cyclin-GUS fusion protein. Plant J 20:503–508
Davies PJ (2004) Plant hormones: biosynthesis, signal transduction, action! Kluwer Academic Publishers, Dordrecht
Dharmasiri N, Dharmasiri S, Estelle M (2005) The F-box protein TIR1 is an auxin receptor. Nature 435:441–445
Fukaki H, Tameda S, Masuda H, Tasaka M (2002) Lateral root formation is blocked by a gain-of-function mutation in the SOLITARY-ROOT/IAA14 gene of Arabidopsis. Plant J 29:153–168
Gälweiler L, Guan C, Müller A, Wisman E, Mendgen K, Yephremov A, Palme K (1998) Regulation of polar auxin transport by AtPIN1 in Arabidopsis vascular tissue. Science 282:2226–2230
Gottwald JR, Krysan PJ, Young JC, Evert RF, Sussman MR (2000) Genetic evidence for the in planta role of phloem-specific plasma membrane sucrose transporters. Proc Natl Acad Sci USA 7:13979–13984
Imlau A, Truernit E, Sauer N (1999) Cell-to-cell and long-distance trafficking of the green fluorescent protein in the phloem and symplastic unloading of the protein into sink tissues. Plant Cell 11:309–322
Johnson MP (2016) Photosynthesis. Essays Biochem 60:255–273
Kepinski S, Leyser O (2005) The Arabidopsis F-box protein TIR1 is an auxin receptor. Nature 435:446–451
Krecek P, Skupa P, Libus J, Naramoto S, Tejos R, Friml J, Zazímalová E (2009) The PIN-FORMED (PIN) protein family of auxin transporters. Genome Biol 10(249):1–11
Lalonde S, Wipf D, Frommer WB (2004) Transport mechanisms for organic forms of carbon and nitrogen between source and sink. Annu Rev Plant Biol 55:341–372
Li SB, Xie ZZ, Hu CG, Zhang JZ (2016) A review of auxin response factors (ARFs) in plants. Front Plant Sci 7(47):1–6
Lilley JLS, Gee CW, Sairanen I, Ljung K, Nemhauser JL (2012) An endogenous carbon-sensing pathway triggers increased auxin flux and hypocotyl elongation. Plant Physiol 160:2261–2270
Lin XY, Ye YQ, Fan SK, Jin CW, Shao Jian S, Zheng SJS (2015) Increased sucrose accumulation regulates iron-deficiency responses by promoting auxin signalling in Arabidopsis plants. Plant Physiol 170:907–920
Lincoln C, Britton JH, Estelle M (1990) Growth and development of the axr1 mutants of Arabidopsis. Plant Cell 2:1071–1080
Liscum E, Reed JW (2002) Genetics of Aux/IAA and ARF action in plant growth and development. Plant Mol Biol 49:387–400
López-Arredondo DL, Leyva-González MA, González-Morales SI, López-Bucio J, Herrera-Estrella L (2014) Phosphate nutrition: improving low-phosphate tolerance in crops. Annu Rev Plant Biol 65:95–123
López-Bucio J, Hernández-Abreu E, Sánchez-Calderón L, Nieto-Jacobo MF, Simpson J, Herrera-Estrella L (2002) Phosphorus availability alters root architecture and causes changes in hormone sensitivity in the Arabidopsis root system. Plant Physiol 129:244–256
López-Bucio J, Hernández-Madrigal F, Cervantes C, Ortiz-Castro R, Carreón-Abud Y, Martínez-Trujillo M (2014) Phosphate relieves chromium toxicity in Arabidopsis thaliana plants by interfering with chromate uptake. Biometals 27:363–370
López-Bucio J, Ortiz-Castro R, Ruiz-Herrera LF, Vargas-Juárez C, Hernández-Madrigal F, Carreón-Abud Y, Martínez-Trujillo M (2015) Chromate induces adventitious root formation via auxin signalling and SOLITARY ROOT/IAA14 gene function in Arabidopsis thaliana. Biometals 28:353–365
MacGregor DR, Deak KI, Ingram PA, Malamy JE (2008) Root system architecture in Arabidopsis grown in culture is regulated by sucrose uptake in the aerial tissues. The Plant Cell 20:2643–2660
Martínez-Trujillo M, Méndez-Bravo A, Ortiz-Castro R, Hernández-Madrigal F, Ibarra-Laclette E, Ruiz-Herrera LF, Long TA, Cervantes C, Herrera-Estrella L, López-Bucio J (2014) Chromate alters root system architecture and activates expression of genes involved in iron homeostasis and signalling in Arabidopsis thaliana. Plant Mol Biol 86:35–50
Michniewicz M, Brewer PB, Friml J (2007) Polar auxin transport and asymmetric auxin distribution open Access. The Arabidopsis Book 5:e0108. doi:10.1199/tab.0108 2007.
Muday GK, DeLong A (2001) Polar auxin transport: controlling where and how much. Trends Plant Sci 6:535–542
Muday GK, Rahman A, Binder BM (2012) Auxin and ethylene: collaborators or competitors? Trends Plant Sci 17:181–195
Müller A, Guan C, Gälweiler L, Tänzler P, Huijser P, Marchant A, Parry G, Bennett M, Wisman E, Palme K (1998) AtPIN2 defines a locus of Arabidopsis for root gravitropism control. EMBO J 17:6903–6911
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Parry G, Calderon-Villalobos LI, Priggeb M, Peretc B, Dharmasiria S, Itohd H, Lechnera E, Grayd E, Bennett WM, Estelle M M (2009) Complex regulation of the TIR1/AFB family of auxin receptors. Proc Natl Acad Sci USA 106:22540–22545
Péret B, Swarup K, Ferguson A, Seth M, Yang Y, Dhondt S, James N, Casimiro I, Perry P, Syed A, Yang H, Reemmer J, Venison E, Howells C, Perez-Amador MA, Yun J, Alonso J, Beemste TS, Laplaze L, Murphy A, Bennett MJ, Nielsen E, Swarup R (2012) AUX/LAX genes encode a family of auxin influx transporters that perform distinct functions during Arabidopsis development. Plant Cell 24:2874–2885
Pickett FB, Wilson AK, Estelle M (1990) The aux1 mutation of Arabidopsis confers both auxin and ethylene resistance. Plant Physiol 94:1462–1466
Potters G, Pasternak TP, Guisez Y, Palme KJ, Jansen AK (2007) Stress-induced morphogenic responses: growing out of trouble? Trends Plant Sci 12:98–105
Ruan YL (2014) Sucrose metabolism: gateway to diverse carbon use and sugar signalling. Annu Rev Plant Biol 65:33–67
Sabatini S, Beis D, Wolkenfelt H, Murfett J, Guilfoyle T, Malamy J, Benfey P, Leyser O, Bechtold N, Weisbeek P, Scheres B (1999) An auxin-dependent distal organizer of pattern and polarity in the Arabidopsis root. Cell 99:463–472
Sánchez-Calderón L, López-Bucio J, Chacón-López A, Cruz-Ramírez A, Nieto-Jacobo F, Dubrovsky JG, Herrera-Estrella L (2005) Phosphate starvation induces a determinate developmental program in the roots of Arabidopsis thaliana. Plant Cell Physiol 46:174–178
Shanker AK, Cervantes C, Loza-Tavera H, Avudainayagam S (2005) Chromium toxicity in plants. Environ Int 31:739–775
Shupack SI (1991) The chemistry of chromium and some resulting analytical problems. Environ Health Perspect 92:7–11
Stohs SJ, Bagchi D (1995) Mechanisms in the toxicity of metal ions. Free Radical Biol Med 18:321–336
Ulmasov T, Murfett J, Hagen G, Guilfoyle TJ (1997) Aux/IAA proteins repress expression of reporter genes containing natural and highly active synthetic auxin elements. Plant Cell 7:288–299
Xiong Y, Sheen J (2012) Rapamycin and glucose-target of rapamycin (TOR) protein signalling in plants. J Biol Chem 287:2836–2842
Xiong Y, McCormack M, Li L, Hall Q, Xiang C, Sheen Jen J (2013) Glc-TOR signalling leads transcriptome reprogramming and meristem activation. Nature 496:181–186
Yang L, Xu M, Koo Y, He J, Poethig RS (2013) Sugar promotes vegetative phase change in Arabidopsis thaliana by repressing the expression of MIR156A and MIR156C. eLife 2:e00260. doi:10.7554/eLife.00260
Yu S, Cao L, Zhou CM, Zhang TQ, Lian H, Sun Y, Wu J, Huang J, Wang G, Wang JW (2013) Sugar is an endogenous cue for juvenile-to-adult phase transition in plants. eLife 2:e00269. doi:10.7554/eLife.00269
Zakhartsev M, Medvedeva I, Orlov Y, Akberdin I, Krebs O, Schulze WX (2016) Metabolic model of central carbon and energy metabolisms of growing Arabidopsis thaliana in relation to sucrose translocation. BMC Plant Biol 16:262. doi: 10.1186/s12870-016-0868-3
Zimmermann P, Hirsch-Hoffman M, Hennig L, Gruissem W (2004) GENEVESTIGATOR: Arabidopsis microarray database and analysis toolbox. Plant Physiol 136:2621–2632
Acknowledgements
Drs. Mark A. Estelle and Alfredo Cruz-Ramírez are thanked for providing us Arabidopsis mutant and transgenic lines. This work was supported by grants from the Consejo Nacional de Ciencia y Tecnología (CONACYT, México, Grant Nos. 177775, and 169769), and the Consejo de la Investigación Científica (UMSNH, México, Grant No. CIC 2.26).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Hernández-Madrigal, F., Ortiz-Castro, R., Ruiz-Herrera, L.F. et al. Sucrose Protects Arabidopsis Roots from Chromium Toxicity Influencing the Auxin–Plethora Signaling Pathway and Improving Meristematic Cell Activity. J Plant Growth Regul 37, 530–538 (2018). https://doi.org/10.1007/s00344-017-9751-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-017-9751-1