Abstract
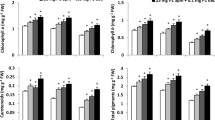
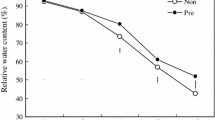
No information is available concerning the influence of dual application of 24-epibrassinolide (EBL) and spermine (Spm) on the nitrogen metabolism in plants subjected to drought conditions. As a first report, this investigation assesses the role of EBL, Spm, and their dual application on polyamine and protein pools in water-stressed plants. It explores the ameliorative effects of these foliar applications under water deficiency. Two maize hybrids (Giza 10 and Giza 129) were treated with or without EBL and/or Spm foliar applications under well-irrigated and drought-stressed conditions (75 and 50 % of field capacity). Dual application (25 mg l−1 Spm + 0.1 mg l−1 EBL) significantly relieved the drought-induced inhibition on the activities of ribulose-1,5-bisphosphate carboxylase and nitrate reductase and the contents of relative water, nitrate, and protein, particularly in hybrid Giza 129. Changes in the content of free polyamines and in the activity of polyamine biosynthetic and catabolic enzymes were detected when water-stressed plants were treated with EBL and/or Spm. Putrescine content and arginine decarboxylase activity were significantly increased in stressed hybrid Giza 10 plants treated by the dual application. However, spermidine and Spm levels as well as ornithine decarboxylase and S-adenosylmethionine decarboxylase activities were significantly increased in stressed hybrid Giza 129 plants treated with the dual application. Diamine oxidase, polyamine oxidase, protease activity, carbonyl content, and ethylene formation were increased in response to water stress and significantly decreased when stressed plants were treated by the dual application. Total free amino acids, phenols, and flavonoids concentration were increased with the increasing water stress level; moreover, they further increased in stressed plants treated with the dual application. Overall, the combined utilization of EBL and Spm serves as complementary tools to confer plant drought tolerance by altering polyamine, ethylene, and protein levels.










Similar content being viewed by others
References
Alcazar R, Altabella T, Marco F, Bortolotti C, Reymond M, Koncz C, Carrasco P, Tiburcio A (2010) Polyamines: molecules with regulatory functions in plant abiotic stress tolerance. Planta 231:1237–1249
Anjum SA, Wang LC, Farooq M, Hussain M, Xue LL, Zou CM (2011) Brassinolide application improves the drought tolerance in maize through modulation of enzymatic antioxidants and leaf gas exchange. J Agron Crop Sci 197:177–185
Arteca RN, Arteca JM (2008) Effects of brassinosteroids, auxin, and cytokinin on ethylene production in Arabidopsis thaliana plants. J Exp Bot 59:3019–3026
Asthir B, Duffus CM, Smith RC, Spoor W (2002) Diamine oxidase is involved in H2O2 production in the chalazal cells during barley grain filling. J Exp Bot 53:677–682
Bajguz A (2000) Effect of brassinosteroids on nucleic acid and protein content in cultured cell of Chlorella vulgaris. Plant Physiol Biochem 38:209–215
Bartoli CG, Gomez F, Martinez DE, Guiamet JJ (2004) Mitochondria are the main target for oxidative damage in leaves of wheat (Triticum aestivum L.). J Exp Bot 55:1663–1669
Behnamnia M, Kalantari KM, Ziaie J (2009) The effects of brassinosteroid on the induction of biochemical changes in Lycopersicon esculentum under drought stress. Turk J Bot 33:417–428
Bouchereau A, Aziz A, Larher F, Martin-Tanguy J (1999) Polyamines and environmental challenges: recent development. Plant Sci 140:103–125
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 7:248–254
Casadebaig P, Debaeke P, Lecoeur J (2008) Thresholds for leaf expansion and transpiration response to soil water deficit in a rang of sunflower genotypes. Eur J Agron 28:646–654
Coombs J, Hall DO, Long SP, Scurlock JMO (1987) Techniques in bioproductivity and photosynthesis. Pergamon, Oxford
Cottenie A, Verloo M, Kiekens L, Velghe G, Camerlynck R (1982) Chemical analysis of plants and soils. Laboratory of analytical and agrochemistry. State University, Ghent, pp 14–24
Drapeau G (1974) Protease from Staphylococcus aureus. In: Lorand L (ed) Method of enzymology, vol 45b. Academic Press, New York
Duan J, Li J, Guo S, Kang Y (2008) Exogenous spermidine affects polyamine metabolism in salinity-stressed Cucumis sativus roots and enhances short-term salinity tolerance. J Plant Physiol 165:1620–1635
Farooq M, Wahid A, Lee DJ (2009) Exogenously applied polyamines increase drought tolerance of rice by improving leaf water status, photosynthesis and membrane properties. Acta Physiol Plant 31:937–945
Fresneau C, Ghashghaie J, Cornic G (2007) Drought effect on nitrate reductase and sucrose-phosphate synthase activities in wheat (Triticum durum L.): role of leaf internal CO2. J Exp Bot 58:2983–2992
Garnica M, Houdusse F, Yvin JC, Garcia-Mina JM (2009) Nitrate supply induced changes in polyamine content and ethylene production in wheat plants grown with ammonium. J Plant Physiol 166:363–374
Groppa MD, Benavides MP (2008) Polyamines and abiotic stress: recent advances. Amino Acids 34:35–45
Gupta K, Dey A, Gupta B (2013) Plant polyamines in abiotic stress responses. Acta Physiol Plant 35:2015–2036
Hameed A, Bibi N, Akhter J, Iqbal N (2011) Differential changes in antioxidants, proteases, and lipid peroxidation in flag leaves of wheat genotypes under different levels of water deficit conditions. Plant Physiol Biochem 49:178–185
Hayat S, Ali B, Hasan SA, Ahmad A (2007) Brassinosteroid enhanced the level of antioxidants under cadmium stress in Brassica juncea. Environ Exp Bot 60:33–41
Huang X, Zhou G, Yang W, Wang A, Hu Z, Lin C, Chen X (2014) Drought-inhibited ribulose-1,5-bisphosphate carboxylase activity is mediated through increased release of ethylene and changes in the ratio of polyamines in pakchoi. J Plant Physiol 171:1392–1400
Jaworski EG (1971) Nitrate reductase assay in intact plant tissues. Biochem Biol Res Commun 43:1274–1279
Kasukabe Y, He L, Nada K, Misawa S, Iharu I, Tachibana S (2004) Overexpression of spermidine synthase enhances tolerance to multiple environmental stress and upregulates the expression of various stress-regulated genes in transgenic Arabidopsis thaliana. Plant Cell Physiol 45:712–722
Kim D, Jeong SW, Lee CY (2003) Antioxidant capacity of phenolic phytochemicals from various cultivars of plums. Food Chem 81:321–326
Krishna P (2003) Brassinosteroid-mediated stress responses. J Plant Growth Regul 22:289–297
Kubis’ J (2008) Exogenous spermidine differently alters activities of some scavenging system enzymes, H2O2 and superoxide radical levels in water-stressed cucumber leaves. J Plant Physiol 165:397–406
Liu JH, Moriguchi T (2007) Changes in free polyamine titers and expression of polyamine biosynthetic genes during growth of peach in vitro callus. Plant Cell Rep 26:125–131
Liu JH, Kitashiba H, Wang J, Ban Y, Moriguchi T (2007) Polyamines and their ability to provide environmental stress tolerance to plants. Plant Biotech 24:117–126
Lowry OH, Rosenbrough NJ, Aarr AL, Randaal RJ (1951) Protein measurement with Folin phenol reagent. J Biol Chem 193:265–275
Mai YY, Lin JM, Zeng XL, Pan RJ (1989) Effect of homobrassinolide on the activity of nitrate reductase in rice seedlings. Plant Physiol Commun 2:50–52
Makino A, Mae T, Ohira K (1988) Differences between wheat and rice in the enzymic properties of ribulose-1,5-bisphosphate carboxylase/oxygenase and the relationship to photosynthetic gas exchange. Planta 174:30–38
Minocha SC, Papa NS, Khan AJ, Samuelsen AI (1991) Polyamines and somatic embryogenesis in carrot. III. Effects of methylglyoxal bis (guanylhydrazone). Plant Cell Physiol 32:302–395
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Mol R, Filek M, Machackova I, Matthys-Rochon E (2004) Ethylene synthesis and auxin augmentation in pistil tissues are important for egg cell differentiation after pollination in maize. Plant Cell Physiol 45:1396–1405
Moore S, Stein WH (1954) A modified ninhydrin reagent for the photometric determination of amino acids and related compounds. J Biol Chem 211:907–913
Radhakrishnan R, Lee I (2013) Spermine promotes acclimation to osmotic stress by modifying antioxidant, abscisic Acid, and jasmonic acid signals in soybean. J Plant Growth Regul 32:22–30
Reznick AZ, Packer L (1994) Oxidative damage to proteins: spectrophotometric method for carbonyl-assay. Methods Enzymol 233:357–363
Ribaut JM, Betran J, Monneveux P, Setter T (2012) Drought tolerance in maize. In: Bennetzen JL, Hake SC (eds) Handbook of maize: its biology. Springer, New York, pp 311–344
Rivero RM, Ruiz JM, García PC, López-Lefebre LR, Sánchez E, Romero L (2001) Resistance to cold and heat stress: accumulation of phenolic compounds in tomato and watermelon plants. Plant Sci 160:315–321
Sawada S, Usuda H, Hasegawa Y, Tsukui T (1990) Regulation of rubisco activity in response to changes of the source/sink balance in single-rooted soybean leaves. Plant Cell Physiol 31:697–704
Singh JP (1988) A rapid method for determination of nitrate in soil and plant extracts. Plant Soil 110:137–139
Snedecor GW, Cochran WG (1980) Statistical Methods, 7th edn. Iowa State University Press, Ames
Szabados L, Savoure A (2010) Proline: a multifunctional amino acid. Trends Plant Sci 15:89–97
Talaat NB, Shawky BT (2012) 24-Epibrassinolide ameliorates the saline stress and improves the productivity of wheat (Triticum aestivum L.). Environ Exp Bot 82:80–88
Talaat NB, Shawky BT (2013a) 24-Epibrassinolide alleviates salt-induced inhibition of productivity by increasing nutrients and compatible solutes accumulation and enhancing antioxidant system in wheat (Triticum aestivum L.). Acta Physiol Plant 35:729–740
Talaat NB, Shawky BT (2013b) Modulation of nutrient acquisition and polyamine pool in salt-stressed wheat (Triticum aestivum L.) plants inoculated with arbuscular mycorrhizal fungi. Acta Physiol Plant 35:2601–2610
Talaat NB, Shawky BT (2014) Protective effects of arbuscular mycorrhizal fungi on wheat (Triticum aestivum L.) plants exposed to salinity. Environ Exp Bot 98:20–31
Talaat NB, Shawky BT, Ibrahim AS (2015) Alleviation of drought-induced oxidative stress in maize (Zea mays L.) plants by dual application of 24-epibrassinolide and spermine. Environ Exp Bot 113:47–58
Xiong Y, Contento AL, Nguyen PQ, Bassham DC (2007) Degradation of oxidized proteins by autophagy during oxidative stress in Arabidopsis. Plant Physiol 143:291–299
Yiu J, Liu C, Fang DY, Lai Y (2009) Waterlogging tolerance of Welsh onion (Allium fistulosum L.) enhanced by exogenous spermidine and spermine. Plant Physiol Biochem 47:710–716
Yuan G, Jia C, Li Z, Sun B, Zhang L, Liu N, Wang Q (2010) Effect of brassinosteroids on drought resistance and abscisic acid concentration in tomato under water stress. Sci Hortic 126:103–108
Zhang C, Huang Z (2013) Effects of endogenous abscisic acid, jasmonic acid, polyamines, and polyamine oxidase activity in tomato seedlings under drought stress. Sci Hortic 159:172–177
Zhang M, Zhai Z, Tian X, Duan L, Li Z (2008) Brassinolide alleviated the adverse effect of water deficits on photosynthesis and the antioxidant of soybean (Glycine max L.). Plant Growth Regul 56:257–264
Zhou Q, Yu B (2010) Changes in content of free, conjugated and bound polyamines and osmotic adjustment in adaptation of vetiver grass to water deficit. Plant Physiol Biochem 48:417–425
Zhu JK (2001) Cell signaling under salt, water and cold stresses. Curr Opin Plant Biol 4:401–406
Acknowledgments
This research was supported by the Academy of Scientific Research and Technology in Egypt and the Bulgaria-Egypt Joint Research Project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Talaat, N.B., Shawky, B.T. Dual Application of 24-Epibrassinolide and Spermine Confers Drought Stress Tolerance in Maize (Zea mays L.) by Modulating Polyamine and Protein Metabolism. J Plant Growth Regul 35, 518–533 (2016). https://doi.org/10.1007/s00344-015-9557-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-015-9557-y