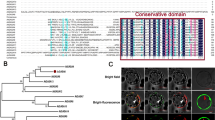
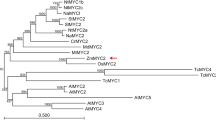
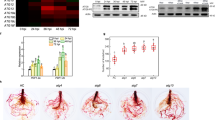
Abstract
Lotus corniculatus is exposed to many forms of biotic stresses, including pathogen and herbivore attack. Jasmonates (JAs) are lipid-based signaling molecules that play important roles in mediating secondary metabolite biosynthesis and plant defense responses. Here, we first characterized LcJAZ1 and LcMYC2, two key components of the JAs signaling pathway. Both LcJAZ1 and LcMYC2 mRNA accumulated quickly in response to JAs. Subcellular localization analysis showed that both LcJAZ1 and LcMYC2 are nuclear proteins. In addition, LcJAZ1 protein was degraded after JAs treatment, but LcMYC2 is not. Yeast two-hybrid, pull-down, and bimolecular fluorescence complementation assays demonstrated that LcJAZ1 interacts with LcMYC2 and AtMYC2. In addition, LcMYC2 interacts with AtJAZ1. The Arabidopsis protoplast transactivation assay revealed that LcJAZ1 acts as repressors of LcMYC2- or AtMYC2-mediated transcription. Our data indicate that the conserved JAZ-MYC complex contributes to the modulation of the JAs signaling pathway.







Similar content being viewed by others
References
Bracha-Drori K, Shichrur K, Katz A, Oliva M, Angelovici R, Yalovsky S, Ohad N (2004) Detection of protein–protein interactions in plants using bimolecular fluorescence complementation. Plant J 40:419–427
Cheng Z, Sun L, Qi T, Zhang B, Peng W, Liu Y, Xie D (2011) The bHLH transcription factor MYC3 interacts with the jasmonate ZIM-domain proteins to mediate jasmonate response in Arabidopsis. Mol Plant 4:279–288
Chini A, Fonseca S, Fernández G, Adie B, Chico JM, Lorenzo O, García-Casado G, López-Vidriero I, Lozano FM, Ponce MR, Micol JL, Solano R (2007) The JAZ family of repressors is the missing link in jasmonate signalling. Nature 448:666–671
Chini A, Fonseca S, Chico JM, Fernández-Calvo P, Solano R (2009) The ZIM domain mediates homo- and heteromeric interactions between Arabidopsis JAZ proteins. Plant J 59:77–87
Chung HS, Howe GA (2009) A critical role for the TIFY motif in repression of jasmonate signaling by a stabilized splice variant of the JASMONATE ZIM-domain protein JAZ10 in Arabidopsis. Plant Cell 21:131–145
Fernández-Calvo P, Chini A, Fernández-Barbero G, Chico JM, Gimenez-Ibanez S, Geerinck J, Eeckhout D, Schweizer F, Godoy M, Franco-Zorrilla JM, Pauwels L, Witters E, Puga MI, Paz-Ares J, Goossens A, Reymond P, De Jaeger G, Solano R (2011) The Arabidopsis bHLH transcription factors MYC3 and MYC4 are targets of JAZ repressors and act additively with MYC2 in the activation of jasmonate responses. Plant Cell 23:701–715
Fonseca S, Chico JM, Solano R (2009) The jasmonate pathway: the ligand, the receptor and the core signalling module. Curr Opin Plant Biol 12:539–554
Gietz D, St Jean A, Woods RA, Schiestl RH (1992) Improved method for high efficiency transformation of intact yeast cells. Nucleic Acids Res 20:1425
Hou X, Lee LYC, Xia K, Yan Y, Yu H (2010) DELLAs modulate jasmonate signaling via competitive binding to JAZs. Dev Cell 19:884–894
James P, Halladay J, Craig EA (1996) Genomic libraries and a host strain designed for highly efficient two-hybrid selection in yeast. Genetics 144:1425–1436
Katsir L, Schilmiller AL, Staswick PE, He SY, Howe GA (2008) COI1 is a critical component of a receptor for jasmonate and the bacterial virulence factor coronatine. Proc Natl Acad Sci USA 105:7100–7105
Melotto M, Mecey C, Niu Y, Chung HS, Katsir L, Yao J, Zeng W, Thines B, Staswick P, Browse J, Howe GA, He SY (2008) A critical role of two positively charged amino acids in the Jas motif of Arabidopsis JAZ proteins in mediating coronatine- and jasmonoyl isoleucine-dependent interactions with the COI1 F-box protein. Plant J 55:979–988
Nakata M, Mitsuda N, Herde M, Koo AJ, Moreno JE, Suzuki K, Howe GA, Ohme-Takagi M (2013) A bHLH-type transcription factor, ABA-INDUCIBLE BHLH-TYPE TRANSCRIPTION FACTOR/JA-ASSOCIATED MYC2-LIKE1, acts as a repressor to negatively regulate jasmonate signaling in Arabidopsis. Plant Cell 25:1641–1656
Niu Y, Figueroa P, Browse J (2011) Characterization of JAZ-interacting bHLH transcription factors that regulate jasmonate responses in Arabidopsis. J Exp Bot 62:2143–2154
Pasquali G, Ouwerkerk PBF, Memelink J (1994) Versatile transformation vectors to assay the promoter activity of DNA elements in plants. Gene 149:373–374
Pauwels L, Goossens A (2011) The JAZ proteins: a crucial interface in the jasmonate signaling cascade. Plant Cell 23:3089–3100
Pauwels L, Barbero GF, Geerinck J, Tilleman S, Grunewald W, Cuéllar Pérez A, Chico JM, Vanden Bossche R, Sewell J, Gil E, García-Casado G, Witters E, Inzé D, Long JA, De Jaeger G, Solano R, Goossens A (2010) NINJAconnects the corepressor TOPLESS to jasmonate signalling. Nature 464:788–791
Pieterse CMJ, Leon-Reyes A, Van der Ent S, Van Wees SCM (2009) Networking by small-molecule hormones in plant immunity. Nat Chem Biol 5:308–316
Qi T, Song S, Ren Q, Wu D, Huang H, Chen Y, Fan M, Peng W, Ren C, Xie D (2011) The jasmonate-ZIM-domain proteins interact with the WD repeat/bHLH/MYB complexes to regulate jasmonate-mediated anthocyanin accumulation and trichome initiation in Arabidopsis thaliana. Plant Cell 23:1795–1814
Sasaki-Sekimoto Y, Jikumaru Y, Obayashi T, Saito H, Masuda S, Kamiya Y, Ohta H, Shirasu K (2013) Basic helix-loop-helix transcription factors JASMONATE-ASSOCIATED MYC2-LIKE1 (JAM1), JAM2, and JAM3 are negative regulators of jasmonate responses in Arabidopsis. Plant Physiol 163:291–304
Sato S, Nakamura Y, Kaneko T, Asamizu E, Kato T, Nakao M, Sasamoto S, Watanabe A, Ono A, Kawashima K, Fujishiro T, Katoh M, Kohara M, Kishida Y, Minami C, Nakayama S, Nakazaki N, Shimizu Y, Shinpo S, Takahashi C, Wada T, Yamada M, Ohmido N, Hayashi M, Fukui K, Baba T, Nakamichi T, Mori H, Tabata S (2008) Genome structure of the legume, Lotus japonicus. DNA Res 15:227–239
Schirawski J, Planchais S, Haenni AL (2000) An improved protocol for the preparation of protoplasts from an established Arabidopsis thaliana cell suspension culture and infection with RNA of turnip yellow mosaic tymovirus: a simple and reliable method. J Virol Methods 86:85–94
Schweizer F, Fernández-Calvo P, Zander M, Diez-Diaz M, Fonseca S, Glauser G, LewseyMG Ecker JR, Solano R, Reymond P (2013) Arabidopsis basic helix-loop-helix transcription factors MYC2, MYC3, and MYC4 regulate glucosinolate biosynthesis, insect performance, and feeding behavior. Plant Cell 25:3117–3132
Sheard LB, Tan X, Mao H, Withers J, Ben-Nissan G, Hinds TR, Kobayashi Y, Hsu FF, Sharon M, Browse J, He SY, Rizo J, Howe GA, Zheng N (2010) Jasmonate perception by inositol-phosphate-potentiated COI1-JAZ co-receptor. Nature 468:400–405
Song S, Qi T, Fan M, Zhang X, Gao H, Huang H, Wu D, Guo H, Xie D (2013) The bHLH subgroup IIId factors negatively regulate jasmonate-mediated plant defense and development. PLoS Genet 9(7):e1003653
Sun ZM, Zhou ML, Xiao XG, Tang YX, Wu YM (2014) Genome-wide analysis of AP2/ERF family genes from Lotus corniculatus shows LcERF054 enhances salt tolerance. Funct Integr Genom 14:453–466
Swain T, Hillis WE (1959) The phenolic constituents of Prunus domestica. I: the quantitative analysis of phenolic constituents. J Agric Food Sci 10:63–68
Thines B, Katsir L, Melotto M, Niu Y, Mandaokar A, Liu G, Nomura K, He SY, Howe GA, Browse J (2007) JAZ repressor proteins are targets of the SCFCOI1 complex during jasmonate signalling. Nature 448:661–665
van der Fits L, Memelink J (1997) Comparison of the activities of CaMV 35S and FMV 34S promoter derivatives in Catharanthus roseus cells transiently and stably transformed by particle bombardment. Plant Mol Biol 33:943–946
Yan Y, Stolz S, Chételat A, Reymond P, Pagni M, Dubugnon L, Farmer EE (2007) A downstream mediator in the growth repression limb of the jasmonate pathway. Plant Cell 19:2470–2483
Yan J, Zhang C, Gu M, Bai Z, Zhang W, Qi T, Cheng Z, Peng W, Luo H, Nan F, Wang Z, Xie D (2009) The Arabidopsis CORONATINE INSENSITIVE1 protein is a jasmonate receptor. Plant Cell 21:2220–2236
Zhou ML, Hou HL, Zhu XM, Shao JR, Wu YM, Tang YX (2011) Soybean transcription factor GmMYBZ2 represses catharanthine biosynthesis in hairy roots of Catharanthus roseus. Appl Microbiol Biotechnol 91:1095–1105
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant No. 31372361) and National Program on Key Basic Research Project (973 Program) (Grant No. 2014CB138701).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Meiliang Zhou and Zhanmin Sun have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
344_2015_9551_MOESM2_ESM.pdf
Supplementary material 2 . Supporting Information Figure S1: AtMYC3 and AtMYC4 interact with LcJAZ1 and AtJAZ1 in yeast. Yeast cells expressing LcJAZ1 and AtJAZ1 proteins fused to the GAL4 BD and AtMYC3 and AtMYC4 fused to the GAL4 AD were spotted on SD/LW (-2) to select for the plasmids and on SD/-LWH with 10 mM 3-AT (-3) to select for transcriptional activation of the His3 gene. Growth was monitored after 7 days. Yeast cells transformed with the empty plasmids pAS2.1 and pACT2, expressing GAL4 BD and AD, respectively, were used as controls. (PDF 41 kb)
344_2015_9551_MOESM3_ESM.pdf
Supplementary material 3. Supporting Information Figure S2: Western blot using anti-HA to identify the positive transgenic plants. W: wild-type plants. (PDF 49 kb)
Rights and permissions
About this article
Cite this article
Zhou, M., Sun, Z., Li, J. et al. Identification of JAZ1-MYC2 Complex in Lotus corniculatus . J Plant Growth Regul 35, 440–448 (2016). https://doi.org/10.1007/s00344-015-9551-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-015-9551-4