Abstract
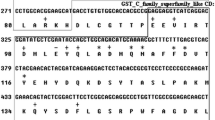
Glutathione S-transferases (GSTs) are a class of enzymes that facilitate the detoxification of xenobiotics, and also play important roles in antioxidant defense. We identified two glutathione S-transferase isoforms (VpGSTS, sigma GST; VpGSTO, omega GST) from Venerupis philippinarum by RACE approaches. The open reading frames of VpGSTS and VpGSTO were of 612 bp and 729 bp, encoding 203 and 242 amino acids with an estimated molecular mass of 22.88 and 27.94 kDa, respectively. The expression profiles of VpGSTS and VpGSTO responded to heavy metals and benzo[a]pyrene (B[a]P) exposure were investigated by quantitative real-time RT-PCR. The expression of VpGSTS and VpGSTO were both rapidly up-regulated, however, they showed differential expression patterns to different toxicants. Cd displayed stronger induction of VpGSTS expression with an approximately 12-fold increase than that of VpGSTO with a maximum 6.4-fold rise. Cu exposure resulted in similar expression patterns for both VpGSTS and VpGSTO. For B[a]P exposure, the maximum induction of VpGSTO was approximately two times higher than that of VpGSTS. Altogether, these findings implied the involvement of VpGSTS and VpGSTO in host antioxidant responses, and highlighted their potential as a biomarker to Cd and B[a]P exposure.
Similar content being viewed by others
References
Banni M, Negri A, Dagnino A, Jebali J, Ameur S, Boussetta H. 2010. Acute effects of benzo[a]pyrene on digestive gland enzymatic biomarkers and DNA damage on mussel Mytilus galloprovincialis. Ecotoxicol. Environ. Saf., 73: 842–848, http://dx.doi.org/10.1016/j.ecoenv.2009.12.032.
Blanchette B, Feng X, Singh B R. 2007. Marine glutathione S-transferases. Mar. Biotechnol., 23: 513–542, http://dx.doi.org/10.1007/s10126-007-9034-0.
Boutet I, Tanguy A, Rousseau S, Auffret M, Moraga D. 2003. Molecular identification and expression of Heat Shock Cognate 70 (hsc70) and Heat Shock Protein 70 (hsp70) genes in the Pacific oyster Crassostrea gigas. Cell Stress Chap., 8: 76–85, http://dx.doi.org/10.1379/1466-1268 (2003).
Boutet I, Tanguy A, Moraga D. 2004. Characterization and expression of four mRNA sequences encoding glutathione S-transferases pi, mu, omega, and sigma classes in the Pacific oyster Crassostrea gigas exposed to hydrocarbons and pesticides. Mar. Biol., 146: 53–64, http://dx.doi.org/10.1007/s00227-004-1423-6.
Bryan G W, Hummerstone L G. 1971. Adaptations of the polychaete Nereis diversicolor to estuarine sediments containing high concentrations of heavy metals. I. General observations and adaptations to copper. J. Mar. Biol. Assoc. U. K. 51: 845–863, http://dx.doi.org/10.1017/S0025315400018014.
Company R, Serafim A, Bebianno M J, Cosson R, Shillito B, Fiala-Médioni A. 2004. Effect of cadmium, copper and mercury on antioxidant enzyme activities and lipid peroxidation on the gills of the hydrothermal vent mussel Bathymodiolus axoricus. Mar. Environ. Res., 58: 377–381, http://dx.doi.org/10.1016/j.marenvres.2004.03.083.
Cunha I, Mangas-Ramirez E, Guilhermino L. 2007. Effects of copper and cadmium on cholinesterase and glutathione S-transferase activities of two marine gastropods ( Monodonta lineata and Nucella lapillus ). Comp. Biochem. Physiol., 145 C: 648–657, http://dx.doi.org//10.1016/j.cbpc.2007.02.014.
Cunha I, García L M, Guilhermino L. 2005. Sea-urchin ( Paracentrotus lividus ) glutathione S-transferases and cholinesterase activities as biomarkers of environmental contamination. J. Environ. Monit., 7: 288–294, http://dx.doi.org/10.1039/B414773A.
Dautrememepuits C, Betoulle S, Vernet G. 2002. Antioxidant response modulated by copper in healthy and parasitized carp ( Cyprinus carpio L.) by Ptychobothrium sp. ( Cestoda ). Biochim. Biophys. Acta, 1 573: 4–8, http://dx.doi.org/10.1016/S0304-4165(02)00328-8.
DeJong J L, Morgenstern R, Jornvall H, DePierre J W, Tu C P. 1988. Gene expression of rat and human microsomal glutathione S-transferases. J. Biol. Chem., 263: 8 430–8 436.
Dondero F, Piacentinia L, Bannia M, Rebeloa M, Burlandoa B, Viarengo A. 2005. Quantitative PCR analysis of two molluscan metallothionein genes unveils differential expression and regulation. Gene, 345: 259–270, http://dx.doi.org/10.1016/j.gene.2004.11.031.
Doyen P, Vasseur P, Rodius F. 2006. Identification, sequencing and expression of selenium-dependent glutathione peroxidase transcript in the freshwater bivalve Unio tumidus exposed to Aroclor 1254. Comp. Biochem. Physiol., 144 C: 122–129, http://dx.doi.org/10.1016/j.cbpc.2006.07.002.
Galloway T S, Sanger R C, Smith K L, Fillmann G, Readman J W, Ford T E, Depledge M H. 2002. Rapid assessment of marine pollution using multiple biomarkers and chemical immunoassays. Environ. Sci. Technol., 36: 2 219–2 226, http://dx.doi.org/10.1021/es010300w.
Gao Q, Song L S, Ni D J, Wu L T, Zhang H, Chang Y Q. 2007. cDNA cloning and mRNA expression of heat shock protein 90 gene in the haemocytes of Zhikong scallop Chlamys farreri. Comp. Biochem. Physiol., 147 B: 704–715, http://dx.doi.org/10.1016/j.cbpb.2007.04.010.
Hayes J D, Flanagan J U, Jowsey I R. 2005. Glutathione transferases. Annu. Rev. Pharmacol. Toxicol., 45: 51–88, http://dx.doi.org/10.1146/annurev.pharmtox.45.120403.095857.
Hoarau P, Damiens G, Roméo M, Gnassia-Barelli M, Bebianno M J. 2006. Cloning and expression of a GST-pi gene in Mytilus galloprovincialis. Attempt to use the GST-pi transcript as a biomarker of pollution. Comp. Biochem. Physiol., 143 C: 196–203, http://dx.doi.org/10.1016/j.cbpc.2006.02.007.
Ji J, Choi H J, Ahn I Y. 2006. Evaluation of Manila clam Ruditapes philippinarum as a sentinel species for metal pollution monitoring in estuarine tidal flats of Korea: effects of size, sex, and spawning on baseline accumulation. Mar. Pollut. Bull. 52: 447–453, http://dx.doi.org/10.1016/j.marpolbul.2005.12.012.
Ketterer B, Coles B, Meyer D J. 1983. The role of glutathione in detoxication. Environ. Health Perspect., 49: 59–69.
Kim J H, Dahms H U, Rhee J S, Lee Y M, Lee J, Han K N, Lee J S. 2010. Expression profiles of seven glutathione S-transferase (GST) genes in cadmium-exposed river pufferfish ( Takifugu obscurus ). Comp. Biochem. Physiol., 151 C: 99–106, http://dx.doi.org/10.1016/j.cbpc.2009.09.001.
Lee K W, Raisuddin S, Rhee J S, Hwang D S, Yu I T, Lee Y M, Park H G, Lee J S. 2008. Expression of glutathione S-transferase (GST) genes in the marine copepod Tigriopus japonicus exposed to trace metals. Aquat. Toxicol., 89: 158–166, http://dx.doi.org/10.1016/j.aquatox.2008.06.011.
Lee Y M, Lee K W, Seo J S, Park H, Park H G, Ahn I Y, Raisuddin S, Lee J S. 2007. Sequence, biochemical characteristics and expression of a novel sigma class of glutathione S-transferase of intertidal copepod, Tigriopus japonicus with a possible role in antioxidant defense. Chemosphere, 69: 893–902, http://dx.doi.org/10.1016/j.chemosphere.2007.05.087.
Liang L N, He B, Jiang G B, Chen D Y, Yao Z W. 2004. Evaluation of mollusks as biomonitors to investigate heavy metal contaminations along the Chinese Bohai Sea. Sci. Total. Environ., 324: 105–113, http://dx.doi.org/10.1016/j.scitotenv.2003.10.021.
Mosleh Y Y, Paris-Palacios S, Biagianti-Risbourg S. 2005. Metallothioneins induction and antioxidative response in aquatic worms Tubifex tubifex (Oligochaeta, tubificiadae) exposed to copper. Chemosphere, 64: 121–128.
Nahrgang J, Camus L, Gonzalez P, Goksøyr A. 2009. PAH biomarker responses in polar cod (Boreogadus saida) exposed to benzo(a)pyrene. Aquat. Toxicol., 94: 309–319, http://dx.doi.org/10.1016/j.aquatox.2009.07.017.
Park H, Ahn I Y, Kim H, Lee J, Shin S C. 2009. Glutathione S-transferase as a biomarker in the Antarctic bivalve Laternula elliptica after exposure to the polychlorinated biphenyl mixture Aroclor 1254. Comp. Biochem. Physio l., 150 C: 528–536, http://dx.doi.org/10.1016/j.cbpc.2009.07.008.
Rainbow P S. 1995. Biomonitoring of heavy metal availability in marine environment. Mar. Pollut. Bull., 31: 183–192, http://dx.doi.org/10.1016/0025-326X(95)00116-5.
Ren H L, Xu D D, Gopalakrishnan S, Qiao K. 2009. Gene cloning of a sigma class glutathione S-transferase from abalone ( Haliotis diversicolor ) and expression analysis upon bacterial challenge. Comp. Biochen. Physiol., 33 C: 980–990, http://dx.doi.org/10.1016/j.dci.2009.04.003.
Sarkar A, Ray D, Shrivastava A N, Sarker S. 2006. Molecular Biomarkers: their significance and application in marine pollution monitoring. Ecotoxicology, 15: 333–340, http://dx.doi.org/10.1007/s10646-006-0069-1.
Wan Q, Whang I, Lee J. 2008a. Molecular cloning and characterization of three sigma glutathione S-transferases from disk abalone ( Haliotis discus discus ). Comp. Biochem. Physiol., 151 B: 257–267, http://dx.doi.org/10.1016/j.cbpb.2008.07.012.
Wan Q, Whang I, Lee J. 2008b. Molecular characterization of mu class glutathione-S-transferase from disk abalone ( Haliotis discus discus ), a potential biomarker of endocrinedisrupting chemicals. Comp. Biochem. Physiol., 150 B: 187–199, http://dx.doi.org/10.1016/j.cbpb.2008.03.002.
Wan Q, Whang I, Lee JS, Lee J. 2009. Novel omega glutathione S-transferases in disk abalone: characterization and protective roles against environmental stress. Comp. Biochem. Physiol., 150C: 558–568, http://dx.doi.org/10.1016/j.cbpc.2009.08.003.
Whang I, Lee J S, Lee J. 2009. Novel omega glutathione S-transferases in disk abalone: Characterization and protective roles against environmental stress. Comp. Biochem. Physiol., 150 C: 558–568, http://dx.doi.org/10.1016/j.cbpc.2009.08.003.
Wang L, Liang X F, Liao WQ, Lei L M, Han B P. 2006. Structural and functional characterization of microcystin detoxification-related liver genes in a phytoplanktivorous fish, Nile tilapia ( Oreochromis niloticu s). Comp. Biochem. Physiol., 144 C: 216–227, http://dx.doi.org/10.1016/j.cbpc.2006.08.009.
Wang L, Song L, Ni D, Zhang H, Liu W. 2009. Alteration of metallothionein mRNA in bay scallop Argopecten irradians under cadmium exposure and bacteria challenge. Comp. Biochem. Physiol., 149 C: 50–57, http://dx.doi.org/10.1016/j.cbpc.2008.07.001.
Xu C, Pan L, Liu N, Wang L, Miao J. 2010. Cloning, characterization and tissue distribution of a pi-class glutathione S-transferase from clam ( Venarupis philippinnarum ): Response to benzo[a]pyrene exposure. Comp. Biochen. Physiol., 152 C: 160–166, http://dx.doi.org/10.1016/j.cbpc.2010.03.011.
Xue W, Warshawsky D. 2006. Metabolic activation of polycyclic and heterocyclic aromatic hydrocarbons and DNA damage: a review. Toxicol. Appl. Pharmacol., 206: 73–93, http://dx.doi.org/10.1016/j.taap.2004.11.006.
Yoshinaga M, Ueki T, Michibata H. 2007. Metal binding ability of glutathione transferases conserved between two animal species, the vanadium-rich ascidian Ascidia sydneiensis samea and the schistosome Schistosoma japonicum. Biochim. Biophys. Acta, 1 770: 1 413–1 418, http://dx.doi.org/10.1016/j.bbagen.2007.05.007.
Zhang L, Liu X, Chen L, You L, Pei D, Cong M, Zhao J, Li C, Liu D, Yu J, Wu H. 2011. Transcriptional regulation of selenium-dependent glutathione peroxidase from Venerupis philippinarum in response to pathogen and contaminants challenge. Fish Shellfish Immunol., 31(6): 831–837, http://dx.doi.org/10.1016/j.fsi.2011.07.026.
Zhang X. 2001. Investigation of pollution of Pb, Cd, Hg, As in sea water and deposit of Bohai Sea area. Heilongjiang Environ J., 25: 87–90. (in Chinese with English abstract)
Zhang Z L, Hong H S, Zhou J L, Yu G. 2004. Phase association of polycyclic aromatic hydrocarbons in the Minjiang River Estuary, China. Sci. Total Environ., 323: 71–86, http://dx.doi.org/10.1016/j.scitotenv.2003.09.026.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supported by the 100 Talents Program of Chinese Academy of Sciences and Key Laboratory of Marine Spill Oil Identification and Damage Assessment Technology (No. 201115), and the State Oceanic Administration of China
Rights and permissions
About this article
Cite this article
Zhang, L., Wu, H., Liu, X. et al. Molecular cloning and differential expression patterns of sigma and omega glutathione S-transferases from Venerupis philippinarum to heavy metals and benzo[a]pyrene exposure. Chin. J. Ocean. Limnol. 30, 413–423 (2012). https://doi.org/10.1007/s00343-012-1173-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00343-012-1173-0