Abstract
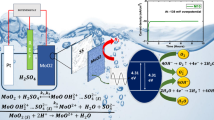
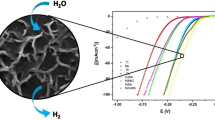
Developing a cost-effective, highly efficient electrocatalyst for the hydrogen evolution reaction (HER) is a major challenge. Herein, we developed metallic molybdenum dioxide (MoO2) films on a stainless steel (SS) substrate by a simple one-step hydrothermal process employing citric acid as a chelating agent. The experiment was carried out with different concentrations of the precursor solution. The porous and compactly arranged interconnected micro-sheets are shown by a MoO2 that enhances the HER in the acidic electrolyte. Its electrochemical study showed excellent water-splitting activity with more than 9 h of stability. The low onset potential of 60 mV and overpotential of 162 mV vs RHE at a current density of 10 mA/cm2 makes this electrode an efficient electro-catalyst for HER. Also, the use of low-cost substrates like SS and normal air annealing, makes the MoO2 sample, cost-effective and better for practical application.












Similar content being viewed by others
Data availability
The raw/processed data required to reproduce these findings cannot be shared at this time as the data also forms part of an ongoing study.
References:
M.A. Khan, H. Zhao, W. Zou, Z. Chen, W. Cao, J. Fang, J. Xu, L. Zhang, J. Zhang, Recent progresses in electrocatalysts for water electrolysis. Electrochemical Energy Reviews 1, 483–530 (2018). https://doi.org/10.1007/s41918-018-0014-z
J.D. Holladay, J. Hu, D.L. King, Y. Wang, An overview of hydrogen production technologies. Catal. Today 139, 244–260 (2009). https://doi.org/10.1016/j.cattod.2008.08.039
B.C.R. Ewan, R.W.K. Allen, A figure of merit assessment of the routes to hydrogen. Int. J. Hydrogen Energy 30–9, 809–819 (2005). https://doi.org/10.1016/j.ijhydene.2005.02.003
Y. Wang, J. Zhang, Structural engineering of transition metal-based nanostructured electrocatalysts for efficient water splitting. Front. Chem. Sci. Eng. 12, 838–854 (2018). https://doi.org/10.1007/s11705-018-1746-3
R. de Levie, Short Communication The electrolysis of water; Journal of Electroanalytical Chemistry 476 (1999) 92–93. www.fbsfvmfr.gb/bjhntf/afbfhefd
H. Cavendish, Experiments on air. Philos. Trans. 74, 119 (1784). https://doi.org/10.1098/rstl.1784.0014
A. Paets Van Troostwijk, J.R. Deiman, electron-proton transfer theory and electrocatalysis. Obs. Phys 35, 369 (1798)
C.G. Morales-Guio, L.A. Stern, X. Hu, Nanostructured hydrotreating catalysts for electrochemical hydrogen evolution. Chem Soc Rev 43, 6555–6569 (2014). https://doi.org/10.1039/C3CS60468C
S. Wang, A. Lu, C.-J. Zhong, Hydrogen production from water electrolysis: role of catalysts. Nano Convergence (2021). https://doi.org/10.1186/s40580-021-00254-x
V.A. Shahraei, Investigation of the water splitting reactions on multi-heteroatom doped cobalt-based carbon catalysts, 2018, chapter 1 (Dissertation)
N.-T. Suen, S.-F. Hung, Q. Quan, N. Zhang, Y.-J. Xu, H. Ming, Electrocatalysis for the oxygen evolution reaction: recent development and future perspectives. Chem. Soc. Rev. 46, 337–365 (2017). https://doi.org/10.1039/c6cs00328a
Z. Lei, J.M. Lee, G. Singh, C.I. Sathish, X. Chu, A.H. Al-Muhtaseb, A. Vinu, J. Yi, Recent advances of layered-transition metal oxides for energy-related applications. Energy Storage Mater. 36, 514–550 (2021). https://doi.org/10.1186/s40580-021-00254-x
Q. Zhang, X. Li, Q. Ma, Q. Zhang, H. Bai, W. Yi, J. Liu, J. Han, G. Xi, A metallic molybdenum dioxide with high stability for surface enhanced Raman spectroscopy. Nat. Commun. 8, 14903 (2017). https://doi.org/10.1038/ncomms14903
V. Ramakrishnan, C. Alex, A.N. Nair, N.S. John, Designing metallic MoO2 nanostructures on rigid substrates for electrochemical water activation. Chem. A Eur. J. 24, 18003–18011 (2018). https://doi.org/10.1002/chem.201803570
Y. Jin, P.K. Shen, Nanoflower-like metallic conductive MoO2 as a high-performance non-precious metal electrocatalyst for the hydrogen evolution reaction. J. Mater. Chem. A 3, 20080–20085 (2015). https://doi.org/10.1039/c5ta06018d
S. Ashraf, C.S. Blackman, G. Hyett, I.P. Parkin, Aerosol assisted chemical vapour deposition of MoO3 and MoO2 thin films on glass from molybdenum polyoxometallate precursors; thermophoresis and gas phase nanoparticle formation. J. Mater. Chem. 16, 3575 (2006). https://doi.org/10.1039/b607335b
Y. Liu, B. Huang, Z. Xie, Hydrothermal synthesis of core-shell MoO2/α-Mo2C heterojunction as high performance electrocatalyst for hydrogen evolution reaction. Appl. Surf. Sci. 427, 693–701 (2018). https://doi.org/10.1016/j.apsusc.2017.08.098
B. Wang, Z. Zhang, S. Zhang, Y. Cao, Y. Sua, S. Liue, W. Tang, J. Yub, Y. Oub, S. Xiea, J. Lig, M. Ma, Surface excited MoO2 to master full water splitting. Electrochim Acta. 359, 136929 (2020). https://doi.org/10.1016/j.electacta.2020.136929
Y. Fan, X. Zhang, Y. Zhang, X. Xie, J. Ding, J. Cai, B. Li, H. Lv, L. Liu, M. Zhu, X. Zheng, Q. Cai, Y. Liu, Lu. Siyu, Decoration of Ru/RuO2 hybrid nanoparticles on MoO2 plane as bifunctional electrocatalyst for overall water splitting. J. Colloid Interface Sci. 604, 508–516 (2021). https://doi.org/10.1016/j.jcis.2021.07.038
L. Yang, Yu. Jiayuan, Z. Wei, G. Li, L. Cao, W. Zhou, S. Chen, Co-N-doped MoO2 nanowires as efficient electrocatalysts for the oxygen reduction reaction and hydrogen evolution reaction. Nano Energy 41, 772–779 (2017). https://doi.org/10.1016/j.nanoen.2017.03.032
S. Deng, X. Liu, T. Huang, T. Zhao, Y. Lu, J. Cheng, T. Shen, J. Liang, D. Wang, MoO2 modulated electrocatalytic properties of Ni: investigate from hydrogen oxidation reaction to hydrogen evolution reaction. Electrochim. Acta 324, 134892 (2019). https://doi.org/10.1016/j.electacta.2019.134892
T. Zhou, Y.Q. Huang, A. Ali, P.K. Shen, Ni-MoO2 nanoparticles heterojunction loaded on stereotaxically-constructed graphene for high-efficiency overall water splitting. J. Electroanalyt. Chem. 897, 115555 (2021). https://doi.org/10.1016/j.jelechem.2021.115555
L. Karuppasamy, L. Gurusamy, S. Anandan, C.H. Liu, J.J. Wu, Defect-enriched heterointerfaces N-MoO2–Mo2C supported Pd nanocomposite as a novel multifunctional electrocatalyst for oxygen, reduction reaction and overall water splitting. Mater. Today Chem. 24, 100799 (2022). https://doi.org/10.1016/j.mtchem.2022.100799
Y.C. Shinta, B. Zaman, S. Sumiyati, Citric Acid and EDTA as chelating agents in phytoremediation of heavy metal in polluted soil: A review. IOP Conf. Ser. Earth Environ. Sci. (2021). https://doi.org/10.1088/1755-1315/896/1/012023
M.A. Pin, Sabir Hussain, Yuan Lin and Zihai Cheng, FeS 2 /carbon hybrids on carbon cloth: a highly efficient and stable counter electrode for dye-sensitized solar cell. Sustain. Energy Fuels 3, 1749–1756 (2019)
Lu. Wang, G.-H. Zhang, K.-C. Chou, Study on oxidation mechanism and kinetics of MoO2 to MoO3 in air atmosphere. Int. J. Refract. Metals Hard Mater. 57, 115–124 (2016). https://doi.org/10.1016/j.ijrmhm.2016.03.001
A. Bhaskar, M. Deepa, M. Deepa, MoO2/multiwalled carbon nanotubes (MWCNT) hybrid for use as a Li-ion battery anode. Appl. Mater. Interfaces 5, 2555–2566 (2013). https://doi.org/10.1021/am3031536
X. Dengab, B. Zhaoabe, Y. Zhongab, Y. Zhuab, Z. Shaoacd, Rational confinement of molybdenum based nanodots in porous carbon for highly reversible lithium storage. J. Mater. Chem. A 4, 10403–10408 (2016)
H.-S. Kim, J.B. Cook, S.H. Tolbert, B. Dunna, The development of pseudocapacitive properties in nanosized-MoO2. J. Electrochem. Soc. 162, A5083–A5090 (2015). https://doi.org/10.1149/2.0141505jes
L. Wang, J. Suna, Molybdenum modified AISI 304 stainless steel bipolar plate for proton exchange membrane fuel cell. J. Renew. Sustain. Energy 5, 021407 (2013). https://doi.org/10.1063/1.4798437
T. Ungar, Microstructural parameters from X-ray diffraction peak broadening. Scripta Mater. 51, 777781 (2004). https://doi.org/10.1016/j.scriptamat.2004.05.007
D. Tahir, E.K. Lee, T.T. Tham, S.K. Oh, H.J. Kang, J. Hua, S. Heo, J.C. Park, J.G. Chung, J.C. Lee, Band alignment of atomic layer deposited (ZrO2)x(SiO2)1–x gate dielectrics on Si (100). Appl. Phys. Lett. 94, 212902 (2009)
A.S. Pratiyush, S. Krishnamoorthy, R. Muralidharan, S. Rajan, D.N. Nath, Advances in Ga2O3 solar-blind UV photodetectors. In: Gallium Oxide Technology, Devices and Applications Metal Oxides, Chapter-16, 369–399, (2019). https://doi.org/10.1016/B978-0-12-814521-0.00016-6
X. Liu, Y. He, S. Wang, Q. Zhang, Preparation of MoO2 sub-micro scale sheets and their optical properties. J. Alloy. Compd. 509, S408–S411 (2011). https://doi.org/10.1016/j.jallcom.2011.01.089
J. Tauc, F. Abeles (eds.), The Optical Properties of Solids (North-Holland, Amsterdam, 1972)
S.H. Mohamed, O. Kappertz, J.M. Ngaruiya, T.P. Leervad Pedersen, R. Drese, M. Wuttig, Correlation between structure, stress and optical properties in direct current sputtered molybdenum oxide films. Thin Solid Films 429, 135–143 (2003). https://doi.org/10.1016/s0040-6090(03)00068-3
S. Ghosh, H.R. Inta, S. Ganguli, G. Tudu, H.V.S.R.M. Koppisetti, V. Mahalingam, MoO2 as a propitious “Pore-Forming Additive” for boosting the water oxidation activity of cobalt oxalate microrods. J. Phys. Chem. 124, 20010–20020 (2020). https://doi.org/10.1021/acs.jpcc
G. Song, J. Shen, F. Jiang, Hu. Ronggui, W. Li, L. An, R. Zou, Z. Chen, Z. Qin, Hu. Junqing, Hydrophilic molybdenum oxide nanomaterials with controlled morphology and strong plasmonic absorption for photo thermal ablation of cancer cells. ACS Appl. Mater. Interfaces 6, 3915–3922 (2014). https://doi.org/10.1021/am4050184
W. Han, P. Yuan, Y. Fan, H. Liu, X. Bao, Synthesis, self-assembly and disassembly of mono-dispersed mo-based inorganic organic hybrid nanocrystals. J. Mater. Chem. 22, 12121–12127 (2012)
L. Fang, Y. Shu, A. Wang, T. Zhang, Green synthesis and characterization of anisotropic uniform single-crystal α-MoO3 nanostructures. J. Phys. Chem. C 111, 2401–2408 (2007). https://doi.org/10.1021/jp065791r
N. Maheswari, G. Muralidharan, Controlled synthesis of nanostructured molybdenum oxide electrodes for high performance supercapacitor devices. Appl. Surf. Sci. 416, 461–469 (2017). https://doi.org/10.1016/j.apsusc.2017.04.094
T. Nagyne-Kovacs, L. Studnicka, I.E. Lukacs, K. Laszlo, P. Pasierb, I.M. Szilogyi, G. Pokol, Hydrothermal synthesis and gas sensing of monoclinic MoO3 nanosheets. Nanomaterials 10, 891 (2020). https://doi.org/10.3390/nano10050891
L.C. Bichara, H.E. Lanús, E.G. Ferrer, M.B. Gramajo, S.A. Brandán, Vibrational study and force field of the citric acid dimer based on the SQM methodology. Adv. Phys. Chem. (2011). https://doi.org/10.1155/2011/347072
Z.C. Xiang, Q. Zhang, Z. Zhang, X. Jin, X.J. Xua, Preparation and photoelectric properties of semiconductor MoO2 micro/nanospheres with wide bandgap. Ceram. Int. 41, 977–981 (2015). https://doi.org/10.1016/j.ceramint.2014.09.017
N. Dukstiene, D. Sinkeviciute, A. Guobiene, Morphological, structural and optical properties of MoO2 films electrodeposited on SnO2∣glass plate. Open Chem. 10, 1106–1118 (2012). https://doi.org/10.2478/s11532-012-0012-7
J. Zhu, S.X. Zhao, X. Wu, Y.F. Wang, L. Yu, C.W. Nan, Wrapping RGO/MoO2/carbon textile as supercapacitor electrode with enhanced flexibility and areal capacitance. Electrochim. Acta 282, 784–791 (2018)
P. Zhang, L. Zou, H. Hu, M. Wang, J. Fang, Y. Lai, J. Li, 3D Hierarchical carbon microflowers decorated with MoO2 nanoparticles for lithium ion batteries. Electrochim. Acta 250, 219–227 (2017)
P.A. Spevack, N. McIntyre, A. Raman, XPS investigation of supported molybdenum oxide thin films. 1. Calcination and reduction studies. J. Phys. Chem. 97, 11020–11030 (1993). https://doi.org/10.1021/j100144a020
M.A. Pin, Sabir Hussain, Yuan Lin and Zihai Cheng, FeS2 /carbon hybrids on carbon cloth: a highly efficient and stable counter electrode for dye-sensitized solar cell, Sustainable. Energy Fuels 3, 1749–1756 (2019). https://doi.org/10.1039/C9SE00240E
D. Momodu, A. Bello, K. Oyedotun, F. Ochai-Ejeh, J. Dangbegnon, M. Madito, N. Manyala, Enhanced electrochemical response of activated carbon nanostructures from tree-bark biomass waste in polymer-gel active electrolytes. RSC Adv. 7, 37286–37295 (2017). https://doi.org/10.1039/c7ra05810a
M. Akia, N. Salinas, S. Luna, E. Medina, A. Valdez, J. Lopez, J. Ayala, M. Alcoutlabi, K. Lozano, In situ synthesis of Fe3O4-reinforced carbon fiber composites as anodes in lithium-ion batteries. J Mater Sci 54, 13479–13490 (2019)
N. Mahmood, Y. Yao, J.-W. Zhang, L. Pan, X. Zhang, J.-J. Zou, Electrocatalysts for hydrogen evolution in alkaline electrolytes: mechanisms. Chall. Prospect. Sol. Adv. Sci. 5–2, 1700464 (2017). https://doi.org/10.1002/advs.201700464
T. Shinagawa, A.T. Garcia-Esparza, K. Takanabe, Insight on Tafel slopes from a microkinetic analysis of aqueous electrocatalysis for energy conversion. Sci. Rep. 5, 13801 (2015)
N.-U.-A. Babar, Y.F. Joya, H. Khalil, F. Hussain, K.S. Joya, Thin-film iron-oxide nanobeads as bifunctional electrocatalyst for high activity overall water splitting. Int. J. Hydrogen Energy 46, 7885–7902 (2021)
S.N. Sapakal, A. Khan, J.M. Khobragade, A.V. Kadam, Synthesis of MoO2 on steel substrate for hydrogen evolution reaction in water splitting: a new approach. J. Mater. Sci. 58, 1403–1414 (2023). https://doi.org/10.1007/s10853-022-08079-7
A.V. Kadam, S.B. Patil, Polyaniline globules as a catalyst for WO3 nanoparticles for supercapacitor application. Mater. Res. Express 5, 085036 (2018). https://doi.org/10.1088/2053-1591/aad406
C. Beasley, M.K. Gnanamani, E. Santillan-Jimenez, M. Martinelli, W.D. Shafer, S.D. Hopps, N. Wanninayake, D.-Y. Kim, Effect of metal work function on hydrogen production from photocatalytic water splitting with MTiO 2 catalysts. Chem. Sel. 5, 1013–1019 (2020). https://doi.org/10.1002/slct.201904151
M.T. Greiner, L. Chai, M.G. Helander, W.-M. Tang, Lu. Zheng-Hong, Metal/metal-oxide interfaces: How metal contacts affect the work function and band structure of MoO3. Adv. Funct. Mater. 23, 215–226 (2013). https://doi.org/10.1002/adfm.201200993
Acknowledgements
This research work is fully funded by DST-SERB, New Delhi under the project [grant number (EEQ/2021/000633)].
Author information
Authors and Affiliations
Contributions
Conceptualization, methodology, investigation, data collection, data curation and writing—original draft preparation was performed by SNS. Editing was handled by Ayesha Khan, Writing—review & editing, supervision was performed by JMK and AVK, all authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no financial conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (MP4 363 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sapakal, S.N., Khan, A., Khobragade, J.M. et al. MoO2-based cost effective catalyst for hydrogen evolution via water splitting. Appl. Phys. A 129, 700 (2023). https://doi.org/10.1007/s00339-023-06987-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-023-06987-2