Abstract
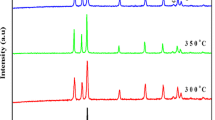
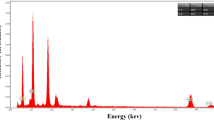
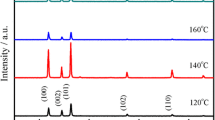
We report the investigation of structure, morphology, photocatalytic degradation of methyl blue using ZnO nanorods (NRs). The ZnO NRs samples were fabricated using electrochemical deposition from aqueous solution of zinc nitrate and hexamethylenetetramine on ITO substrates. The effect of the growth time on the structure, morphology and photocatalytic properties of ZnO NRs was investigated in details.XRD analysis showed that the fabricated NRs have hexagonal wurtzite structure and growth direction along the (002) plane. The morphology of the prepared samples investigated by SEM showed that growth rate of the NRs depends on the time of the growth. The surface area and cross-sectional area of the fabricated ZnO NRs increased with increasing the growth time and led to improvements in the NRs aspect ratios. It was also found that the photocatalytic activity of the grown nanorods enhances by increasing the deposition time. The present results are discussed in terms of the observed structural morphology of the investigated ZnO NRs and correlated with the photocatalytic performance of the grown NRs.







Similar content being viewed by others
References
A. Sedky, S.A. Amin, M. Mohamed, Appl. Phys. A 125, 308 (2019)
A. Sedky, A.M. Ali, M. Mohamed, Opt. Quantum Electron. 52, 42 (2020)
J. Jiang, J. Pi, J. Cai, Bioinorg. Chem. Appl. 2018, 1 (2018).
D. Pourjafari, G. Oska. Nanomater. Sol. Cell Appl. 1 (2019).
J. Tian, G. Weng, Y. Wang, X. Hu, S. Chen, J. Chu, ACS Appl. Nano Mater. 4, 2 (2019).
E. Oh, H.Y. Choi, S.H. Jung, S. Cho, J.C. Kim, K.H. Lee, S.W. Kang, J. Kim, J.Y. Yun, S.H. Jeong, Sens. Actuators B 141, 239 (2009)
M.R. Alenezi, T.H. Alzanki, A.M. Almeshal, A.S. Alshammari, M.J. Beliatis, S.J. Henley, S.R.P. Silva, RSC Adv. 4, 49521 (2014)
F.A. Al-Agel, M.S. Akhtar, H. Alshammari, A. Alshammari, S.A. Khan, Mater. Lett. 147, 119 (2015)
Y. Hames, Z. Alpaslan, A. Kösemen, S.E. San, Y. Yerli, Sol. Energy 84, 426 (2010)
G.C. Yi, C. Wang, W.I. Park, Semicond. Sci. Technol. 0, 22 (2005)
Z. Ye, T. Wang, S. Wu, X. Ji, Q. Zhang, J. Alloys Compd. 690, 189 (2017)
S.K. Mohanta, D.C. Kim, H.K. Cho, S.J. Chua, S. Tripathy, J. Cryst. Growth 310, 3208 (2008)
D.N. Montenegro, V. Hortelano, O. Martínez, M.C. Martínez-Tomas, V. Sallet, V. Muñoz-Sanjosé, J. Jiménez, J. Appl. Phys. 113, 143513 (2013)
S. Sun, S. Jiao, K. Zhang, D. Wang, H. Li, S. Gao, J. Wang, Q. Yu, F. Guo, L. Zhao, S. Su, J. Mater. Sci. Mater. Electron. 24, 85 (2013)
S.A. Taha, A.M. Abd-Elnaiem, M. Mohamed, S. Mostafa, M.S. Mostafa, Desalin. WATER Treat. 100, 160 (2017)
M. Mohamed, S. Moustafa, S.A. Taha, A.M. Abd-Elnaiem, Mater. Res. Express 6, 035026 (2019)
F. Tezcan, A. Mahmood, G. Kardaş, J. Mater. Sci. Mater. Electron. 29, 9547 (2018)
J. Yang, X. Wang, T. Jiang, Y. Li, Q. Ma, J. Han, J. Chen, J. Wang, Y. Wang, Superlatt. Microstruct. 72, 91 (2014)
J. Lv, W. Gong, K. Huang, J. Zhu, F. Meng, X. Song, Z. Sun, Superlatt. Microstruct. 50, 98 (2011)
J. Sakaliuniene, J. Čyviene, B. Abakevičiene, J. Dudonis, Acta Phys. Pol. A 120, 63 (2011)
U. Holzwarth, N. Gibson, Nat. Nanotechnol. 6, 534 (2011)
M. Skompska, K. Zarȩbska, Electrochim. Acta 127, 467 (2014)
S. Baruah, M.A. Mahmood, M.T.Z. Myint, T. Bora, J. Dutta, Beilstein J. Nanotechnol. 1, 14 (2010)
M. Rashad, N.M. Shaalan, M.M. Hafiz, Dig. J. Nanomater. Biostruct. 10, 823 (2015)
H. Ogawa, T. Higuchi, A. Nakamura, S. Tokita, D. Miyazaki, T. Hattori, T. Tsukamoto, J. Alloys Compd. 449, 375 (2008)
A.B.D. Nandiyanto, R. Zaen, R. Oktiani, Arab. J. Chem. 13, 1283 (2020)
K. Tanaka, M.F.V. Capule, T. Hisanaga, Chem. Phys. Lett. 187, 73 (1991)
J. Sharma, M. Vashishtha, D.O. Shah, Glob. J. Sci. Front. Res. B Chem. 14, 19 (2014)
T. Peng, D. Zhao, K. Dai, W. Shi, K. Hirao, J. Phys. Chem. B 109, 4947 (2005)
S. Baruah, M. Jaisai, R. Imani, M.M. Nazhad, J. Dutta, Sci. Technol. Adv. Mater. 11, 055002 (2010)
L.R. Toporovska, A.M. Hryzak, B.I. Turko, V.P. Rudyk, V.S. Tsybulskyi, R.Y. Serkiz, Opt. Quant. Electron. 49, 1 (2017)
Acknowledgements
This research has been funded by Scientific Research Deanship at University of Ha’il, Saudi Arabia, through project number RG-191247. The authors thank the Deanship of Scientific Research at University of Ha’il for funding this research project.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohamed, M., Alshammari, A.S. & Almokhtar, M. Structure, morphology and photocatalytic activity of ZnO nanorods fabricated by electrochemical deposition. Appl. Phys. A 126, 596 (2020). https://doi.org/10.1007/s00339-020-03786-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-020-03786-x