Abstract
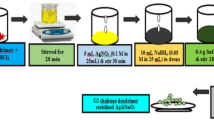
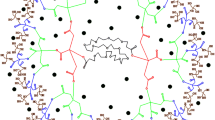
The main focus of this research article is to observe the contributions of chalcone dendrimer in Ag@TiO2 core shell nanoparticles. The samples were synthesized by Redox transmetalation method. The structure of the samples in three molar ratio viz.1:1, 1:2, 1:5 (noted as A,B,C) with Poly Vinyl Pyrrolidone (PVP) as stabilizing agent were compared with the sample Ag@TiO2 of 1:2 molar ratio (noted as D) using zeroth generation triazolyl chalcone dendrimer as stabilizing agent. The prepared samples were structurally characterized by XRD and HRTEM analysis. The XRD and SAED analysis exhibited tetragonal body centered crystal structure with dhkl = 1.6 Å, 2.4 Å and 3.4 Å. The lattice parameters were calculated for all the samples and the variations observed were reported. The grain size and dislocation density of the synthesized nanoparticles were calculated using Debye–Scherrer formula. From Williamson–Hall plot, it was identified that the lattice strain was increased in sample D in which chalcone dendrimer acted as stabilizing agent. The structural investigation showed the impact of stabilizing agent on the surface elements. Similarly, the optical characteristics were discussed based on UV-DRS and Photoluminescence studies. Using Kubelka–Munk function, the band gap energy was calculated and the difference observed in the band gap energy due to the impact of stabilizing agents on metal–metaloxide (Ag@TiO2) combination was analysed. The molar absorptivity (ε) was calculated for all the samples using Beer–Lambert’s law and the difference in absorbance on using different stabilizing agent was discussed.








Similar content being viewed by others
References
T. Pradeep A Textbook of Nanoscience and Nanotechnology (McGraw Hill Edn. (India) Pvt. Ltd., Chennai, 2012) (ISBN 1-25-900732-4)
T. Pradeep, (2007) Nano: The Essentials (McGraw Hill Edn. (India) Pvt. Ltd., Chennai, 2007)
R. Ghosl Chaudhuri, S. Paria, Core/shell nanoparticles: classes, properties, synthesis mechanisms, characterization, and applications. Chem. Rev. 112, 2373–2433 (2012). https://doi.org/10.1021/cr100449n
R. Scott, A. Datye, R. Crooks, Bimetallic palladium—platinum dendrimer encapsulatedcatalysts. J. Am. Chem. Soc. 125, 3708–3709 (2003). https://doi.org/10.1021/ja034176n
G.R. Newkome, C.N. Moorefield, F. Vogtle Dendrons and Dendrimers. Concepts, Synthesis and Applications. (Wiley-VCH, Hoboken, 2001). https://doi.org/10.1002/3527600612
Y. Niu, L. Yeung, R. Crooks, Size-selective hydrogenation of olefins by dendrimer-encapsulated palladium nanoparticles. J. Am. Chem. Soc. 123, 6840–6846 (2001). https://doi.org/10.1021/ja0105257
H.-L. Jiang, Q. Xu, Recent progress in synergistic catalysis over heterometallic nanoparticles. J. Mater. Chem. 21, 13705 (2011). https://doi.org/10.1039/c1jm12020d
X. Chen, S.S. Samuel, Titanium dioxide nanomaterials: synthesis, properties, modifications, and applications. Chem. Rev. 107, 2891–2959 (2007). https://doi.org/10.1021/cr0500535
S. Vaidyaa, A. Patrab, A.K. Gangulia, CdS@TiO2 and ZnS@TiO2 core–shell nanocomposites: synthesis and optical properties. Colloids Surf. A Physicochem. Eng. Aspects. 363, 130–134 (2010). https://doi.org/10.1016/j.colsurfa.2010.04.030
V. Chhabra, V. Pillai, B.K. Mishra, A. Morrone, D.O. Shah (1995) Synthesis, characterization, and properties of microemulsion-mediated nanophase Ti02 particles. Langmuir 11: 3307–3311. https://doi.org/10.1021/la00009a007
D. Mangalaraj, D.N. Devi (2017) Ag/TiO2 (Metal/Metal Oxide) core shell nanoparticles for biological applications. in Springer Proceedings in Physics, 189. https://www.springer.com/in/book/9783319448893. Accessed 5 Apr 2018
D. Zhang, X. Song, R. Zhang, M. Zhang, F. Liu (2005) Preparation and characterization of Ag@TiO2 core-shell nanoparticle water-in-oil emulsions. Eur. J. Inorg. Chem. 1643–1648. https://doi.org/10.1002/ejic.200400811
W. Lee, M. Kim, J. Choi, J. Park, S.J. Ko, S.J. Oh, J. Cheon (2005) Redox—transmetalation process as a generalized synthetic strategy for core–shell magnetic nanoparticles. J. Am. Chem. Soc. 127, 16090–16097. https://doi.org/10.1021/ja053659j
R.V. Vijayalakshmi, A. Kannan, P. Praveen Kumar, K. Ravichandran, P. Rajakumar, Effect of stabilizing agents on the conductivity of Co@TiO2core–shell nanoparticles. Nano-Structures&Nano-Objects 16, 258–265 (2018). https://www.sciencedirect.com/science/article/pii/S2352507X18301215. Accessed 2 Aug 2018
R.V. Vijayalakshmi, A. Kannan, P.Praveen Kumar, K. Ravichandrand, P. Rajakumar, The role of glycodendrimer in the structural and optical studies of Co@AgCl core-shell nanoparticles. Mater. Chem. Phys. 221, 356–360 (2019). https://www.sciencedirect.com/science/article/pii/S0254058418307983?dgcid=rss_sd_all. Accessed 27 Sept 2018
R.V. Vijayalakshmi, P. Praveen Kumar, S. Selvarani, R. Rajakumar, K. Ravichandran, Chalcone dendrimer stabilized core-shell nanoparticles – a comparative study on Co@TiO2, Ag@TiO2 and Co@AgCl nanoparticles for antibacterial and antifungal activity. Mater. Res. Express. 4, 105046 (2017). http://iopscience.iop.org/issue/2053-1591/4/10. Accessed 3 Jan 2018
B.D. Cullity. Elements of X-ray Diffraction. (Addison-Wesley Pub. Comp Inc, California, 1956)
C. Hammond, (2001) The basics of crystallography and diffraction (Oxford University Press, Oxford, 2001)
A.K. Zak, W.H.A. Majid, M.E. Abrishami, R. Yousefi (2011) X-ray analysis of ZnO nanoparticles by Williamson–Hall and size-strain plot methods. Solid State Sci. 13, 251–256. http://repository.um.edu.my/88950/1/solidstatescience2011.pdf. Accessed 12 Dec 2017
G.K. Williamson, W.H. Hall, (1953) X-ray line broadening from filed aluminium and wolfram. Acta Metallurgica. https://www.sciencedirect.com/science/article/pii/0001616053900066. Accessed 12 Dec 2017
B. Manikandan, S.T. Endo, M.K.R. Kaneko, J. Rita, Properties of sol gel synthesized ZnO nanoparticles. J. Mater. Sci. Mater. Electron. (2018). https://doi.org/10.1007/s10854-018-8981-8
P. Bindu, Sabu Thomas, Estimation of lattice strain in ZnO nanoparticles: X-ray peak profile analysis. J Theor Appl Phys 8, 123–134 (2014). https://doi.org/10.1007/s40094-014-0141-9
A.C.J. Wilson, X-ray Optics (UK, London, 1949), https://archive.org/details/X-rayOptics/page/n0
V. Kamlesh, K. Chandekar, K. Mohan, Size-strain analysis and elastic properties of CoFe 2 O 4 nanoplatelets by hydrothermal method. J.Mol.Struc. 1154, 418–427 (2018). https://www.researchgate.net/publication/320061875. Accessed 22 June 2018
K. Ravichandran, D. Nedumaran, Synthesis and characterization of zinc sulphide nanoparticles using inert gas condensation technique. Int J Mech Eng Mater Sci 4(1), 25–31 (2011)
A.E. Morales, E.S. Mora, U. Pal, Use of diffuse reflectance spectroscopy for optical characterization of un-supported nanostructures. Rev. Mexic. De Fisica S53, 18 (2007). https://rmf.smf.mx/pdf/rmf-s/53/5/53_5_18.pdf. Accessed 22 June 2018
T. Silfvast William (2004) Laser fundamentals (Cambridge University Press, New Delhi, 2004) ISBN–0-521-83345-0
B.P. Wang, X. Huang, X. Qin, Y. Zhang, D.J. Wei, W.M. Hwan, Ag@AgCl: a highly efficient and stable photocatalyst active under visible light. Angew. Chem. 47, 7931–7933 (2008). https://doi.org/10.1002/anie.200802483
S. Glaus, G. Calzaferri, The band structures of the silver halides AgF, AgCl, and AgBr: a comparative study. Photochem. Photobiol. Sci. 2, 398–401 (2003). https://doi.org/10.1039/b211678b
F. Moser, R.K. Ahrenkiel, S.L. Lyu (1967) Optical absorption and luminescent emission of the I-center in AgCl. Phys. Rev. 161(3):897–892. https://journals.aps.org/pr/issues/161/3. Accessed 18 Aug 2018
M.I. Tunc, H. Bruns, M. Gliemann, Grunzea, P. Koelsch, Bandgap determination and charge separation in Ag@TiO2 core shell nanoparticle films. Surf. Interface Anal. 42, 835–841 (2010). https://doi.org/10.1002/sia.3558
J.K. Michael Thomas, (1996) Ultraviolet and visible spectroscopy, (John Wiley and sons) ISBN 978-81-265-1723-7
D. Paramelle, A. Sadovoy, S. Gorelik, P. Free, J. Hobleya, D.G. Fernig (2014) A rapid method to estimate the concentration of citrate capped silver nanoparticles from UV-visible light spectra Analyst, 139: 4855–4861. https://doi.org/10.1039/c4an00978a
L.E. Garcia sole, Bass, D. Jaque, (2005) An introduction to the optical spectroscopy solids (John Wiley and sons) ISBN 0-470-868856
Y. Ohko, T. Tatsuma, T. Fujii, K. Naoi, C. Niwa, Y. Kubota, A. Fujishima, Multicolour photochromism of TiO2 films loaded with silver nanoparticles. Nat. Mater. 2, 29 (2003). https://www.nature.com/articles/nmat796
X.Z. Li, F.B. Li (2001) Study of Au/Au3+-TiO2 photocatalysts toward visible photooxidation for water and wastewater treatment, Environ. Sci. Technol. 35, 2381. https://doi.org/10.1021/es001752w
M.I. Litter (1999) Last advances on TiO2—photocatalytic removal of chromium, uranium and arsenic Appl. Catal. B. 2223, 89. https://www.researchgate.net/profile/Marta_Litter/publication/316521226
L.Q. Jing, X.J. Sun, B.F. Xin, W.M. Cai, H.G. Fu, The preparation and characterization of La doped TiO2nanoparticles and their photocatalytic activity. J. Solid State Chem. 177, 3375 (2004). https://www.sciencedirect.com/science/article/pii/S0022459604003068. Accessed 18 Aug 2018
B.F. Xin, L.Q. Jing, Z.Y. Ren, B.Q. Wang, H.G.Fu (2005) Effects of simultaneously doped and deposited Ag on the photocatalytic activity and surface states of TiO2, J.Phys.Chem.B 109 (2005)2805. https://doi.org/10.1021/jp0469618
S. Bagheri, N.M. Julkapli, S. Bee Abd Hamid (2014) Titanium dioxide as a catalyst support in heterogeneous catalysis (Hindawi Publishing Corporation). Sci World J. https://doi.org/10.1155/2014/727496
Acknowledgements
The author thanks Dr.R.Anandhan, Dept. of Organic Chemistry, University of Madras, Chennai for his support to do the entire lab work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Vijayalakshmi, R.V., Selvarani, S., Praveen Kumar, P. et al. Investigations on structural and optical properties of chalcone dendrimer in Ag@TiO2 core–shell nanoparticles. Appl. Phys. A 124, 759 (2018). https://doi.org/10.1007/s00339-018-2177-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-018-2177-1