Abstract
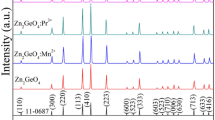
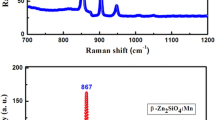
Zn2−2x Mn2x GeO4 (x=0, 0.001, 0.01) phosphors were prepared by conventional solid state reaction technique. X-ray powder diffraction (XRD), field emission scanning electron microscopy (FE-SEM), diffuse reflection spectra, photoluminescence (PL), and cathodoluminescence (CL) spectroscopy were utilized to characterize the synthesized phosphors. The Mn2+-activated Zn2GeO4 phosphors exhibit narrow emission band at 532 nm under the excitation of ultraviolet light, which due to the 4T1(4G)–6A1(6S) transition of Mn2+ ions. Also it is observed that there exists energy transfer between the Zn2GeO4 host lattice and the activator (Mn2+). Under excitation of low-voltage electron beams, Zn2GeO4:Mn2+ shows strong green emission band dominating at 535 nm, corresponding to the 4T1(4G)–6A1(6S) emission of Mn2+ ions. The emission intensity and chromaticity coordinates of Zn2GeO4:Mn2+ as a function of accelerating voltage and the filament current were also investigated.







Similar content being viewed by others
References
S. Itoh, H. Toki, K. Morimoto, T. Kishino, J. Electrochem. Soc. 138, 1509 (1991)
X.M. Liu, J. Lin, J. Mater. Chem. 18, 221 (2008)
J. Silver, R. Withnall, A. Lipman, T.G. Ireland, G.R. Fern, J. Lumin. 122–123, 562 (2007)
S.H. Cho, S.H. Kwon, J.S. Yoo, C.W. Oh, J.D. Lee, K.J. Hong, S.J. Kwon, J. Electrochem. Soc. 147, 3143 (2000)
Y.E. Lee, P.N. David, D.B. John, D.R. Philip, P. Jeff, D.P. Michael, J. Appl. Phys. 91, 2974 (2002)
L. Tian, S.-I. Mho, Solid State Commun. 125, 647 (2003)
Y.C. Kang, H.S. Roh, S.B. Park, Adv. Mater. 12, 451 (2000)
J. Wang, S. Tian, G. Li, F. Liao, X. Jing, J. Electrochem. Soc. 148, H61 (2001)
S.H. Yang, J. Electrochem. Soc. 150, H250 (2003)
S. Yang, C. Stoffers, F. Zhang, S.M. Jacobsen, B.K. Wagner, C.J. Summers, Appl. Phys. Lett. 72, 158 (1998)
F.L. Zhang, S. Yang, C. Stoffers, J. Penczek, P.N. Yocom, D. Zaremba, B.K. Wagner, C.J. Summers, Appl. Phys. Lett. 72, 2226 (1998)
A. Vecht, C. Gibbons, D. Davies, X. Jing, P. Marsh, T. Reland, J. Silver, A. Nowport, D. Barber, J. Vac. Sci. Technol. B 17, 750 (1999)
P. Guo, F. Zhao, G. Li, F. Liao, S. Tian, X. Jing, J. Lumin. 105, 61 (2003)
R. Steven, B.F. Woodfield, J. Boerio-Goates, M.K. Crawford, J. Chem. Thermodyn. 36, 349 (2004)
J. Sato, H. Kobayashi, K. Ikarashi, N. Saito, H. Nishiyama, Y. Inoue, J. Phys. Chem. B 108, 4369 (2004)
J.S. Lewis, P.H. Holloway, J. Electrochem. Soc. 147, 3148 (2000)
J.P. Bender, J.F. Wager, J. Kissick, B.L. Clark, D.A. Keszler, J. Lumin. 99, 311 (2002)
L.C. Williams, D. Norton, J. Budai, P.H. Holloway, J. Electrochem. Soc. 151, H188 (2004)
D.R. Lide, CRC Handbook of Chemistry & Physics, 78th edn. (CRC Press, Boca Raton, 1997)
N. Yamashita, J. Phys. Soc. Jpn. 35, 1089 (1973)
Zh.Sh. Liu, X.P. Jing, L.X. Wang, J. Electrochem. Soc. 154, H500 (2007)
Ch.F. Huang, Ch.F. Lu, T.Y. Tang, J.J. Huang, C.C. Yang, Appl. Phys. Lett. 90, 151122 (2007)
S. Yang, C. Stoffers, F. Zhang, Appl. Phys. Lett. 72, 158 (1998)
Acknowledgements
This work was supported by the National Nature Science Foundation of China (20501023), the Science and Technology Project of Guangzhou (2011J4300012) and the Youth Research Fund Project of Guangzhou Research Institute of Non-ferrous Metals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Q., Wang, J. Synthesis and characterization of Zn2GeO4:Mn2+ phosphor for field emission displays. Appl. Phys. A 108, 943–948 (2012). https://doi.org/10.1007/s00339-012-7003-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00339-012-7003-6