Abstract
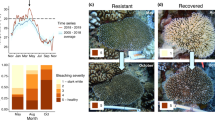
Thermal stress is a major contributor to loss of coral cover, significantly impacting reefs during the third global bleaching event between 2014 and 2017. The long-term persistence of coral reefs depends on acclimatization and adaptation to changing climate, which are influenced greatly by the interactions between bleaching and reproductive success. We observed a genotypically diverse population of Orbicella faveolata before, during, and after consecutive bleaching events in 2014 and 2015 in the Florida Keys. We documented less bleaching during the second event despite 40% more time above local bleaching thresholds and an association between bleaching severity and subsequent spawning. Approximately 75% of colonies experienced the same or less severe bleaching in the second event despite being metabolically compromised, with a substantial minority (~ 35%) faring better in the second event. The second bleaching event also resulted in smaller decreases in chlorophyll content per symbiont cell and symbiont-to-host cell ratio reef-wide, representing less damage to the coral–algal symbiosis. All colonies that recovered quickly (~ 1 month) or did not bleach in 2014 released gametes in 2015, while only 60% of colonies that recovered more slowly did. Bleaching also impacted the amount of gametes released, with more severe bleaching significantly associated with gamete release from < 50% of the colony surface area. Bleaching and spawning outcomes were supported by dynamic physiological changes during bleaching and recovery. Lipid concentration and symbiont-to-host cell ratios collected from the bottom edge of the colony in the middle of the recovery period (February and April) were most important for predicting spawning the following year, highlighting the dynamic interaction between micro-habitats and time in recovery and gametogenesis. This study finds signals of physiological acclimatization in an important reef-building coral and underscores the importance of recovery post-bleaching and reproduction for the persistence of coral reefs.






Similar content being viewed by others
References
Ainsworth TD, Heron SF, Ortiz JC, Mumby PJ, Grech A, Ogawa D, Eakin CM, Leggat W (2016) Climate change disables coral bleaching protection on the Great Barrier Reef. Science 352:338–342
Anthony KRN, Hoogenboom MO, Maynard JA, Grottoli AG, Middlebrook R (2009) Energetics approach to predicting mortality risk from environmental stress: a case study of coral bleaching. Functional Ecology 23:539–550
Ayre DJ, Hughes TP (2000) Genotypic diversity and gene flow in brooding and spawning corals along the Great Barrier Reef, Australia. Evolution 54:1590–1605
Baird A, Marshall P (2002) Mortality, growth and reproduction in scleractinian corals following bleaching on the Great Barrier Reef. Mar Ecol Prog Ser 237:133–141
Baker AC (2004) Symbiont diversity on coral reefs and its relationship to bleaching resistance and resilience. Coral health and disease. Springer, Heidelberg, pp 177–194
Baker AC, Glynn PW, Riegl B (2008) Climate change and coral reef bleaching: an ecological assessment of long-term impacts, recovery trends and future outlook. Estuar Coast Shelf Sci 80:435–471
Baker AC, Rowan R, Knowlton N (1997) Symbiosis ecology of two Caribbean acroporid corals. Proc 8th Int Coral Reef Symposium 2:1295–1300
Bay RA, Palumbi SR (2015) Rapid acclimation ability mediated by transcriptome changes in reef-building corals. Genome Biol Evol 7:1602–1612
Bay RA, Rose N, Barrett R, Bernatchez L, Ghalambor CK, Lasky JR, Brem RB, Palumbi SR, Ralph P (2017) Predicting responses to contemporary environmental change using evolutionary response architectures. The American Naturalist 189:463–473
Bellantuono AJ, Hoegh-Guldberg O, Rodriguez-Lanetty M (2011) Resistance to thermal stress in corals without changes in symbiont composition. Proceedings. Biological Sciences 279:1100–1107
Bellantuono AJ, Granados-Cifuentes C, Miller DJ, Hoegh-Guldberg O, Rodriguez-Lanetty M (2012) Coral thermal tolerance: tuning gene expression to resist thermal stress. PLoS One 7:e50685
Brown BE (1997) Coral bleaching: causes and consequences. Coral Reefs 16:S129–S138
Brown BE, Dunne RP, Ambarsari I, Le Tissier MDA, Satapoomin U (1999) Seasonal fluctuations in environmental factors and variations in symbiotic algae and chlorophyll pigments in four Indo-Pacific coral species. Mar Ecol Prog Ser 191:53–69
Brown BE, Dunne RP, Goodson MS, Douglas AE (2002) Experience shapes the susceptibility of a reef coral to bleaching. Coral Reefs 21:119–126
Costa CF, Sassi R, Amaral FD (2005) Annual cycle of symbiotic dinoflagellates from three species of scleractinian corals from coastal reefs of northeastern Brazil. Coral Reefs 24:191–193
Cowen RK, Sponaugle S (2009) Larval dispersal and marine population connectivity. Ann Rev Mar Sci 1:443–466
Dixon GB, Davies SW, Aglyamova GV, Meyer E, Bay LK, Matz MV (2015) Genomic determinants of coral heat tolerance across latitudes. Science 26:1460–1462
Fagoonee I, Wilson HB, Hassell MP, Turner JF (1999) The dynamics of zooxanthellae populations: a long-term study in the field. Science 283:843–845
Figueiredo J, Baird AH, Cohen MF, Flot JF, Kamiki T, Meziane T, Tsuchiya M, Yamasaki H (2012) Ontogenetic change in the lipid and fatty acid composition of scleractinian coral larvae. Coral Reefs 31:613–619
Fitt WK, McFarland FK, Warner ME, Chilcoat GC (2000) Seasonal patterns of tissue biomass and densities of symbiotic dinoflagellates in reef corals and relation to coral bleaching. Limnol Oceanogr 45:677–685
Fitt WK, Spero HJ, Halas J, White MW, Porter JW (1993) Recovery of the coral Montastrea annularis in the Florida Keys after the 1987 ‘bleaching event’. Coral Reefs 12:57–64
Gilmour JP, Smith LD, Heyward AJ, Baird AH, Pratchett MS (2013) Recovery of an isolated coral reef system following severe disturbance. Science 340:69–71
Gintert BE, Manzello DP, Enochs IC, Kolodziej G, Carlton R, Gleason AC, Gracias N (2018) Marked annual coral bleaching resilience of an inshore patch reef in the Florida Keys: a nugget of hope, aberrance, or last man standing? Coral Reefs 37:533–547
Glynn PW, Maté JL, Baker AC, Calderón MO (2001) Coral bleaching and mortality in Panama and Ecuador during the 1997–1998 El Niño-Southern Oscillation event: spatial/temporal patterns and comparisons with the 1982–1983 event. Bulletin of Marine Science 69:79–109
Grottoli AG, Rodrigues L, Juarez C (2004) Lipids and stable carbon isotopes in two species of Hawaiian corals, Porites compressa and Montipora verrucosa, following a bleaching event. Marine Biology 145:621–631
Grottoli AG, Rodrigues LJ, Palardy JE (2006) Heterotrophic plasticity and resilience in bleached corals. Nature 440:1186–1189
Grottoli AG, Warner ME, Levas SJ, Aschaffenburg M, Schoepf V, McGinley M, Baumann J, Matsui Y (2014) The cumulative impact of annual coral bleaching can turn some coral species winners into losers. Glob Chang Biol 20:3823–3833
Hagedorn M, Carter VL, Lager C, Camperio Ciani JF, Dygert AN, Schleiger RD, Henley EM (2016) Potential bleaching effects on coral reproduction. Reprod Fertil Dev 28:1061–1071
Harii S, Nadaoka K, Yamamoto M, Iwao K (2007) Temporal changes in settlement, lipid content and lipid composition of larvae of the spawning hermatypic coral Acropora tenuis. Mar Ecol Prog Ser 346:89–96
Harland AD, Davies PS, Fixter LM (1992) Lipid content of some Caribbean corals in relation to depth and light. Marine Biology 113:357–361
Harland AD, Navarro JC, Davies PS, Fixter LM (1993) Lipids of some caribbean and red sea corals—total lipid, wax esters, trigylcerides and fatty acids. Marine Biology 117:113–117
Harrison P, Wallace C (1990) Reproduction, dispersal and recruitment of scleractinian corals. Ecosystems of the world 25:133–207
Heron SF, Maynard JA, Van Hooidonk R, Eakin CM (2016) Warming trends and bleaching stress of the world’s coral reefs 1985–2012. Scientific Reports 6:38402
Hoegh-Guldberg O, Smith GJ (1989) The effect of sudden changes in temperature, light and salinity on the population density and export of zooxanthellae from the reef corals Stylophora pistillata Esper and Seriatopora hystrix Dana. J Exp Mar Bio Eco 129:279–303
Holm-Hansen O, Riemann B (1978) Chlorophyll-a Determination: Improvements in Methodology. Oikos 30:438–447
Hughes TP, Tanner JE (2000) Recruitment failure, life histories, and long-term decline of Caribbean corals. Ecology 81:2250–2263
Hughes AR, Inouye BD, Johnson MT, Underwood N, Vellend M (2008) Ecological consequences of genetic diversity. Ecology Letters 11:609–623
Hughes TP, Barnes ML, Bellwood DR, Cinner JE, Cumming GS, Jackson JB, Kleypas J, van de Leemput IA, Lough JM, Morrison TH (2017a) Coral reefs in the Anthropocene. Nature 546:82–90
Hughes TP, Kerry JT, Álvarez-Noriega M, Álvarez-Romero JG, Anderson KD, Baird AH, Babcock RC, Beger M, Bellwood DR, Berkelmans R (2017b) Global warming and recurrent mass bleaching of corals. Nature 543:373
Hughes TP, Anderson KD, Connolly SR, Heron SF, Kerry JT, Lough JM, Baird AH, Baum JK, Berumen ML, Bridge TC, Claar DC (2018) Spatial and temporal patterns of mass bleaching of corals in the Anthropocene. Science 359:80–83
Jones RJ (1997) Changes in zooxanthellar densities and chlorophyll concentrations in corals during and after a bleaching event. Mar Eco Prog Ser 158:51–59
Jones AM, Berkelmans R, van Oppen MJ, Mieog JC, Sinclair W (2008) A community change in the algal endosymbionts of a scleractinian coral following a natural bleaching event: field evidence of acclimatization. Proc Bio Sci 275:1359–1365
Kenkel CD, Matz MV (2017) Gene expression plasticity as a mechanism of coral adaptation to a variable environment. Nat Ecol Evol 1:0014
Kemp DW, Fitt WK, Schmidt GW (2008) A microsampling method for genotyping coral symbionts. Coral Reefs 27:289–293
Levitan DR, Boudreau W, Jara J, Knowlton N (2014) Long-term reduced spawning in Orbicella coral species due to temperature stress. Mar Ecol Prog Ser 515:1–10
Levas S, Schoepf V, Warner ME, Aschaffenburg M, Baumann J, Grottoli AG (2018) Long-term recovery of Caribbean corals from bleaching. J Exp Mar Bio Ecol 506:124–134
Manzello DP, Berkelmans R, Hendee JC (2007) Coral bleaching indices and thresholds for the Florida reef tract, Bahamas, and St. Croix US Virgin Islands. Mar Pollut Bull 54:1923–1931
Matz MV, Treml EA, Aglyamova GV, Bay LK (2018) Potential and limits for rapid genetic adaptation to warming in a Great Barrier Reef coral. PLoS Genetics 14:e1007220
Maynard J, Anthony K, Marshall P, Masiri I (2008) Major bleaching events can lead to increased thermal tolerance in corals. Marine Biology 155:173–182
Mendes J, Woodley J (2002) Effect of the 1995-1996 bleaching event on polyp tissue depth, growth, reproduction and skeletal band formation in Montastraea annularis. Mar Ecol Prog Ser 235:93–102
Mieog JC, Van Oppen MJH, Berklemans R, Stam WT, Olsen JL (2009) Quantification of algal endosymbionts (Symbiodinium) in coral tissue using real-time PCR. Mol Ecol Resour 9:74–82
Middlebrook R, Hoegh-Guldberg O, Leggat W (2008) The effect of thermal history on the susceptibility of reef-building corals to thermal stress. J Exp Biol 211:1050–1056
Miller MW, Piniak GA, Williams DE (2011) Coral mass bleaching and reef temperatures at Navassa Island, 2006. Estuar Coast Shelf Sci 91:42–50
Miller MW, Williams DE, Fisch J (2016) Genet-specific spawning patterns in Acropora palmata. Coral Reefs 35:1393–1398
Miller MW, Baums IB, Pausch RE, Bright AJ, Cameron CM, Williams DE, Moffitt ZJ, Woodley CM (2018) Clonal structure and variable fertilization success in Florida Keys broadcast-spawning corals. Coral Reefs 37:239–249
Munday P, Leis J, Lough J, Paris C, Kingsford M, Berumen M, Lambrechts J (2009) Climate change and coral reef connectivity. Coral Reefs 28:379–395
National Oceanographic and Atmospheric Administration (2014) Endangered and Threatened Wildlife and Plants: Final Listing Determinations on Proposal To List 66 Reef-Building Coral Species and to Reclassify Elkhorn and Staghorn Corals. Federal Register, Document 79 FR 5385:53851–54123
NOAA Coral Reef Watch (2018, updated daily). NOAA Coral Reef Watch Version 3.1 Daily Global 5-km Satellite Coral Bleaching Degree Heating Week Product, Jan. 1, 2014-Dec. 31, 2015. College Park, Maryland, USA: NOAA Coral Reef Watch. Data set accessed 2019-03-25 at https://coralreefwatch.noaa.gov/satellite/hdf/index.php
Oku H, Yamashiro H, Onaga K, Sakai K, Iwasaki H (2003) Seasonal changes in the content and composition of lipids in the coral Goniastrea aspera. Coral Reefs 22:83–85
Ortiz JC, Wolff NH, Anthony KR, Devlin M, Lewis S, Mumby PJ (2018) Impaired recovery of the great barrier reef under cumulative stress. Sci Adv 4:eaar6127
Pausch RE, Williams DE, Miller MW (2018) Impacts of fragment genotype, habitat, and size on outplanted elkhorn coral success under thermal stress. Mar Ecol Prog Ser 592:109–117
Palumbi SR, Barshis DJ, Traylor-Knowles N, Bay RA (2014) Mechanisms of reef coral resistance to future climate change. Science 344:895–898
Pinzón JH, Kamel B, Burge CA, Harvell CD, Medina M, Weil E, Mydlarz LD (2015) Whole transcriptome analysis reveals changes in expression of immune-related genes during and after bleaching in a reef-building coral. R Soc Open Sci 2:140214
Rodrigues LJ, Grottoli AG (2007) Energy reserves and metabolism as indicators of coral recovery from bleaching. Limnol Oceanogr 52:1874–1882
Rowan R, Knowlton N, Baker A, Jara J (1997) Landscape ecology of algal symbionts creates variation in episodes of coral bleaching. Nature 388:265–269
Rowan R, Powers DA (1991) Molecular genetic identification of symbiotic dinoflagellates (zooxanthellae). Mar Ecol Prog Ser 71:65–73
Szmant AM, Gassman NJ (1990) The effects of prolonged bleaching on the tissue biomass and reproduction of the reef coral Montastraea annularis. Coral Reefs 8:217–224
Szmant AM (1991) Sexual reproduction by the Caribbean reef corals Montastrea annularis and M. cavernosa. Mar Ecol Prog Ser 74:13–25
Szmant AM, Weil E, Miller MW, Colon DE (1997) Hybridization within the species complex of the scleractinian coral Montastraea annularis. Marine Biology 129:561–572
Tchernov D, Gorbunov MY, de Vargas C, Yadav SN, Milligan AJ, Häggblom M, Falkowski PG (2004) Membrane lipids of symbiotic algae are diagnostic of sensitivity to thermal bleaching in corals. Proc Natl Acad Sci USA 101:13531–13535
Teece MA, Estes B, Gelsleichter E, Lirman D (2011) Heterotrophic and autotrophic assimilation of fatty acids by two scleractinian corals, Montastraea faveolata and Porites astreoides. Limnol Oceanogr 56:1285–1296
Thomas L, Palumbi SR (2017) The genomics of recovery from coral bleaching. Proc R Soc B 284:20171790
Thornhill DJ, LaJeunesse TC, Kemp DW, Fitt WK, Schmidt GW (2005) Multi-year, seasonal genotypic surveys of coral-algal symbioses reveal prevalent stability or post-bleaching reversion. Marine Biology 148:711–722
Towle EK, Enochs IC, Langdon C (2015a) Threatened Caribbean coral is able to mitigate the adverse effects of ocean acidification on calcification by increasing feeding rate. PLoS One 10:e0123394
Towle EK, Carlton R, Langdon C, Manzello DP (2015b) In-situ measurement of metabolic status in three coral species from the Florida Reef Tract. Reg Stud Mar Sci 2:145–153
Towle EK, Palacio-Castro AM, Baker AC, Langdon C (2017) Source location and food availability determine the growth response of Orbicella faveolata to climate change stressors. Reg Stud Mar Sci 10:107–115
Van Hooidonk R, Maynard J, Planes S (2013) Temporary refugia for coral reefs in a warming world. Nature Climate Change 3:508
Van Oppen MJH, Oliver JK, Putnam HM, Gates RD (2015) Building coral reef resilience through assisted evolution. Proc Natl Acad Sci USA 112:2307–2313
van Veghel MLJ, Bak RPM (1994) Reproductive characteristics of the polymorphic Caribbean coral Montastraea annularis III. Reproduction in damaged and regenerating colonies. Mar Ecol Prog Ser 109:229–233
Wangpraseurt D, Polerecky L, Larkum AW, Ralph PJ, Nielsen DA, Pernice M, Kühl M (2014) The in situ light microenvironment of corals. Limnol Oceanogr 59:917–926
Ward S, Harrison P, Hoegh-Guldberg O (2002) Coral bleaching reduces reproduction of scleractinian corals and increases susceptibility to future stress. Proceedings of the Ninth International Coral Reef Symposium 2:1123–1128
Williams DE, Miller MW, Bright AJ, Pausch RE, Valdivia A (2017) Thermal stress exposure, bleaching response, and mortality in elkhorn coral, Acropora palmata. Mar Pollut Bull 124:189–197
Ziegler M, Seneca FO, Yum LK, Palumbi SR, Voolstra CR (2017) Bacterial community dynamics are linked to patterns of coral heat tolerance. Nat Commun 8:14213
Acknowledgements
We thank Emma Pontes and Camille Akhoudas for assistance with analyzing samples in the laboratory and Phillip Kushlan for assistance in the field with sample collections. We thank Joanne Delaney from the Florida Keys National Marine Sanctuary for all of her help in expediting permits for the research. We especially thank Dana Williams, Allan Bright, and Rachel Pausch for assistance with bleaching and spawning observations in the field supported by the NOAA Coral Reef Conservation Program and NOAA’s Southeast Fisheries Science Center. We thank two anonymous reviewers whose comments improved the quality and clarity of this paper. This work was conducted under permit FKNMS-2014-144. Funding for this project was provided by a competitive grant from the RSMAS Graduate Career Development Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The corresponding author states that there is no conflict of interest.
Additional information
Topic Editor Morgan S. Pratchett
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fisch, J., Drury, C., Towle, E.K. et al. Physiological and reproductive repercussions of consecutive summer bleaching events of the threatened Caribbean coral Orbicella faveolata. Coral Reefs 38, 863–876 (2019). https://doi.org/10.1007/s00338-019-01817-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-019-01817-5