Abstract
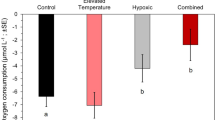
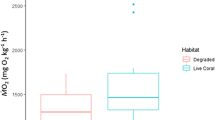
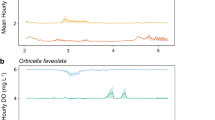
Despite high rates of photosynthetic oxygen production during the day, the warm waters of coral reefs are susceptible to hypoxia at night due to elevated respiration rates at higher temperatures that also reduce the solubility of oxygen. Hypoxia may be a challenge for coral-reef fish that hide in the reef to avoid predators at night. Triggerfishes (Balistidae) are found in a variety of reef habitats, but they also are known to find refuge in reef crevices and holes at night, which may expose them to hypoxic conditions. The critical oxygen tension (P crit) was determined as the point below which oxygen uptake could not be maintained to support standard metabolic rate (SMR) for five species of triggerfish. The triggerfishes exhibited similar levels of hypoxia tolerance as other coral-reef and coastal marine fishes that encounter low oxygen levels in their environment. Two species, Rhinecanthus rectangulus and R. aculeatus, had the lowest P crit (~ 3.0 kPa O2), comparable to the most hypoxia-tolerant obligate coral-dwelling gobies, while Odonus niger and Sufflamen bursa were moderately tolerant to hypoxia (P crit ~ 4.5 kPa), and Xanthichthys auromarginatus was intermediate (P crit ~ 3.7 kPa). These differences in P crit were not due to differences in oxygen demand, as all the species had a similar SMR once mass differences were taken into account. The results suggest that triggerfish species are adapted for different levels of hypoxia exposure during nocturnal sheltering within the reef.





Similar content being viewed by others
References
Bean K, Jones GP, Caley MJ (2002) Relationships among distribution, abundance and microhabitat specialisation in a guild of coral reef triggerfish (family Balistidae). Mar Ecol Prog Ser 233:263–272
Chabot D, Steffensen JF, Farrell AP (2016) The determination of standard metabolic rate in fishes. J Fish Biol 88:81–121
Chapman LJ, Chapman CA, Nordlie FG, Rosenberger AE (2002) Physiological refugia: swamps, hypoxia tolerance and maintenance of fish diversity in the Lake Victoria region. Comp Biochem Physiol A Mol Integ Physiol 133:421–437
Childress JJ, Seibel BA (1998) Life at stable low oxygen levels: Adaptations of animals to oceanic oxygen minimum layers. J Exp Biol 201:1223–1232
Claireaux G, Chabot D (2016) Responses by fishes to environmental hypoxia: integration through Fry’s concept of aerobic metabolic scope. J Fish Biol 88:232–251
Clark TD, Sandblom E, Jutfelt F (2013) Aerobic scope measurements of fishes in an era of climate change: respirometry, relevance and recommendations. J Exp Biol 216:2771–2782
Clarke A, Johnston NM (1999) Scaling of metabolic rate with body mass and temperature in teleost fish. J Anim Ecol 68:893–905
Collins GM, Clark TD, Rummer JL, Carton AG (2013) Hypoxia tolerance is conserved across genetically distinct sub-populations of an iconic, tropical Australian teleost (Lates calcarifer). Conserv Physiol 1:1–9
Diaz RJ, Rosenberg R (2008) Spreading dead zones and consequences for marine ecosystems. Science 321:926–929
Diaz RJ, Breitburg DL (2009) The hypoxic environment. Fish Physiology 27:1–23
Dornburg A, Sidlauskas B, Santini F, Sorenson L, Near TJ, Alfaro ME (2011) The influence of an innovative locomotor strategy on the phenotypic diversification of triggerfish (Family: Balistidae). Evolution 65:1912–1926
Drazen JC, Yeh J, Friedman J, Condon N (2011) Metabolism and enzyme activities of hagfish from shallow and deep water of the Pacific Ocean. Comp Biochem Physiol A Mol Integ Physiol 159:182–187
Farrell AP, Richards JG (2009) Defining hypoxia: An integrative synthesis of the responses of fish to hypoxia. Fish Physiology 27:487–503
Goldshmid R, Holzman R, Weihs D, Genin A (2004) Aeration of corals by sleep-swimming fish. Limnol Oceanogr 49:1832–1839
Graham JB (1990) Ecological, evolutionary, and physical factors influencing aquatic animal respiration. Am Zool 30:137–146
Hobson ES (1972) Activity of Hawaiian reef fishes during the evening and morning transitions between daylight and darkness. Fish Bull 70:715–740
Hobson ES (1974) Feeding relationships of teleostean fishes on coral reefs in Kona, Hawai’i. Fish Bull 72:915–1031
Killen SS, Atkinson D, Glazier DS (2010) The intraspecific scaling of metabolic rate with body mass in fishes depends on lifestyle and temperature. Ecol Lett 13:184–193
Kinsey D, Kinsey E (1967) Diurnal changes in oxygen content of the water over the coral reef platform at Heron I. Mar Freshw Res 18:23–34
Korsmeyer KE, Steffensen JF, Herskin J (2002) Energetics of median and paired fin swimming, body and caudal fin swimming, and gait transition in parrotfish (Scarus schlegeli) and triggerfish (Rhinecanthus aculeatus). J Exp Biol 205:1253–1263
Kühl M, Cohen Y, Dalsgaard T, Jørgensen BB, Revsbech NP (1995) Microenvironment and photosynthesis of zooxanthellae in scleractinian corals studied with microsensors for O2, pH and light. Mar Ecol Prog Ser 117:159–172
Kuwamura T (1991) Habitat segregation, coexistence or interspecific territoriality between two triggerfishes, Rhinecanthus aculeatus and Sufflamen chrysopterus, with notes on distribution of other balistids at Sesoko Island, Okinawa. Galaxea 10:65–78
Levin LA (2003) Oxygen minimum zone benthos: Adaptation and community response to hypoxia. Oceanogr Marine Biol Annu Rev 41:1–45
Mandic M, Todgham AE, Richards JG (2009) Mechanisms and evolution of hypoxia tolerance in fish. Proc R Soc Lond B Biol Sci 276:735–744
Matsuura K (2001) Balistidae. Triggerfishes. In: Carpenter K, Niem V (eds) FAO species identification guide for fishery purposes. The living marine resources of the Western Central Pacific, volume 6: bony fishes part 4 (Labridae to Latimeriidae), estuarine crocodiles, sea turtles, sea snakes and marine mammals. FAO, Rome, pp 3911–3928
McKenzie DJ, Steffensen JF, Korsmeyer K, Whiteley NM, Bronzi P, Taylor EW (2007) Swimming alters responses to hypoxia in the Adriatic sturgeon Acipenser naccarii. J Fish Biol 70:651–658
McRae MG, McRae LB, Fitzsimons JM (2011) Habitats used by juvenile flagtails (Kuhlia spp.; Perciformes: Kuhliidae) on the island of Hawai’i. Pacific Science 65:441–450
Munday PL, McCormick MI, Nilsson GE (2012) Impact of global warming and rising CO2 levels on coral reef fishes: what hope for the future? J Exp Biol 215:3865–3873
Nilsson GE, Östlund-Nilsson S (2004) Hypoxia in paradise: widespread hypoxia tolerance in coral reef fishes. Proc R Soc Lond B Biol Sci 271:S30–S33
Nilsson GE, Östlund-Nilsson S (2008) Does size matter for hypoxia tolerance in fish? Biol Rev Camb Philos Soc 83:173–189
Nilsson GE, Hobbs J-PA, Östlund-Nilsson S (2007a) Tribute to PL Lutz: respiratory ecophysiology of coral-reef teleosts. J Exp Biol 210:1673–1686
Nilsson GE, Hobbs J-P, Munday PL, Östlund-Nilsson S (2004) Coward or braveheart: extreme habitat fidelity through hypoxia tolerance in a coral-dwelling goby. J Exp Biol 207:33–39
Nilsson G, Hobbs JA, Östlund-Nilsson S, Munday P (2007b) Hypoxia tolerance and air-breathing ability correlate with habitat preference in coral-dwelling fishes. Coral Reefs 26:241–248
Portner HO, Farrell AP (2008) Ecology, physiology and climate change. Science 322:690–692
Priede IG (1985) Metabolic scope in fishes. In: Tytler P, Calow P (eds) Fish energetics: new perspectives. Springer, Netherlands, pp 33–64
Randall JE (2005) Reef and shore fishes of the South Pacific: New Caledonia to Tahiti and the Pitcairn Islands. University of Hawai’i Press, Honolulu
Randall JE (2007) Reef and shore fishes of the Hawaiian Islands. University of Hawai’i Sea Grant College Program, University of Hawai’i, Honolulu
Randall JE, Allen GR, Steene RC (1997) Fishes of the Great Barrier Reef and Coral Sea. University of Hawai’i Press, Honolulu
Richards JG (2009) Metabolic and molecular responses of fish to hypoxia. Fish Physiology 27:443–485
Rogers NJ, Urbina MA, Reardon EE, McKenzie DJ, Wilson RW (2016) A new analysis of hypoxia tolerance in fishes using a database of critical oxygen level (P crit). Conserv Physiol 4:cow012
Santini F, Sorenson L, Alfaro ME (2013) A new multi-locus timescale reveals the evolutionary basis of diversity patterns in triggerfishes and filefishes (Balistidae, Monacanthidae; Tetraodontiformes). Mol Phylogenet Evol 69:165–176
Seibel BA (2011) Critical oxygen levels and metabolic suppression in oceanic oxygen minimum zones. J Exp Biol 214:326–336
Seibel BA, Drazen JC (2007) The rate of metabolism in marine animals: environmental constraints, ecological demands and energetic opportunities. Philos Trans R Soc Lond B Biol Sci 362:2061–2078
Shashar N, Cohen Y, Loya Y (1993) Extreme diel fluctuations of oxygen in diffusive boundary layers surrounding stony corals. Biol Bull 185:455–461
Snyder S, Nadler LE, Bayley JS, Svendsen MB, Johansen JL, Domenici P, Steffensen JF (2016) Effect of closed v. intermittent-flow respirometry on hypoxia tolerance in the shiner perch Cymatogaster aggregata. J Fish Biol 88:252–264
Speers-Roesch B, Mandic M, Groom DJ, Richards JG (2013) Critical oxygen tensions as predictors of hypoxia tolerance and tissue metabolic responses during hypoxia exposure in fishes. J Exp Mar Bio Ecol 449:239–249
Steffensen JF (1989) Some errors in respirometry of aquatic breathers: how to avoid and correct for them. Fish Physiol Biochem 6:49–59
Steffensen JF, Bushnell PG, Schurmann H (1994) Oxygen consumption in four species of teleosts from Greenland: no evidence of metabolic cold adaptation. Polar Biol 14:49–54
Svendsen MB, Bushnell PG, Steffensen JF (2016) Design and setup of intermittent-flow respirometry system for aquatic organisms. J Fish Biol 88:26–50
Szczebak JT, Henry RP, Al-Horani FA, Chadwick NE (2013) Anemonefish oxygenate their anemone hosts at night. J Exp Biol 216:970–976
Townhill BL, Pinnegar JK, Righton DA, Metcalfe JD (2017) Fisheries, low oxygen and climate change: how much do we really know? J Fish Biol 90:723–750
White CR, Phillips NF, Seymour RS (2006) The scaling and temperature dependence of vertebrate metabolism. Biol Lett 2:125–127
Yang T, Lai N, Graham J, Somero G (1992) Respiratory, blood, and heart enzymatic adaptations of Sebastolobus alascanus (Scorpaenidae; Teleostei) to the oxygen minimum zone: A comparative study. Biol Bull 183:490–499
Yeager DP, Ultsch GR (1989) Physiological regulation and conformation: a BASIC program for the determination of critical points. Physiol Zool 62:888–907
Zikos A, Seale AP, Lerner DT, Grau EG, Korsmeyer KE (2014) Effects of salinity on metabolic rate and branchial expression of genes involved in ion transport and metabolism in Mozambique tilapia (Oreochromis mossambicus). Comp Biochem Physiol A Mol Integr Physiol 178:121–131
Acknowledgements
The authors thank Captain Brian Quinn for assistance with oxygen data logger deployment and Kat Mowle, Travis Marcoux, and Stacia Goecke for assistance with data collection. We also thank Dr. K. David Hyrenbach for advice on the use of statistics.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Ecology Editor Dr. Alastair Harborne
Rights and permissions
About this article
Cite this article
Wong, C.C., Drazen, J.C., Callan, C.K. et al. Hypoxia tolerance in coral-reef triggerfishes (Balistidae). Coral Reefs 37, 215–225 (2018). https://doi.org/10.1007/s00338-017-1649-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-017-1649-7