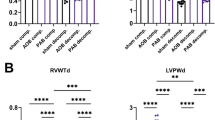
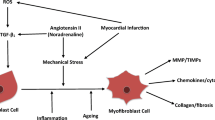
Abstract
Cardiovascular diseases are currently the main cause of death. The study of the pathogenesis and treatment of these diseases is still a major challenge. Traditional 2D cultured cells and animal models have certain limitations. Heart organoids as models can simulate the structure and function of the body, providing a new research strategy. This paper mainly discusses the development of organoids and their application in the study of the cardiac developmental process, drug screening and treatment of genetic and non-genetic diseases, concluding with their strengths and weaknesses.

Similar content being viewed by others
References
Brett Volmert, A.R., Fei Wang, Aniwat Juhong, Artem Kiselev, Aleksandra Kostina, Colin O’Hern, Priyadharshni Muniyandi, Aaron Wasserman, Amanda Huang, Yonatan Lewis-Israeli, ProfileSangbum Park, Zhen Qiu, ProfileChao Zhou, ProfileAitor Aguirre, A patterned human heart tube organoid model generated by pluripotent stem cell self-assembly. bioRxiv, 2022.
Brassard JA, Lutolf MP (2019) Engineering stem cell self-organization to build better organoids. Cell Stem Cell 24(6):860–876
Buono MF et al (2020) Human cardiac organoids for modeling genetic cardiomyopathy. Cells 9(7):1733
Caluori G et al (2019) Non-invasive electromechanical cell-based biosensors for improved investigation of 3D cardiac models. Biosens Bioelectron 124–125:129–135
Corrò C, Novellasdemunt L, Li VSW (2020) A brief history of organoids. Am J Physiol Cell Physiol 319(1):C151–C165
Drakhlis L et al (2021a) Human heart-forming organoids recapitulate early heart and foregut development. Nat Biotechnol 39(6):737–746
Drakhlis L, Devadas SB, Zweigerdt R (2021b) Generation of heart-forming organoids from human pluripotent stem cells. Nat Protoc 16(12):5652–5672
Fatehullah A, Tan SH, Barker N (2016) Organoids as an in vitro model of human development and disease. Nat Cell Biol 18(3):246–254
Firth AL et al (2015) Functional gene correction for cystic fibrosis in lung epithelial cells generated from patient iPSCs. Cell Rep 12(9):1385–1390
Hoang P et al (2018) Generation of spatial-patterned early-developing cardiac organoids using human pluripotent stem cells. Nat Protoc 13(4):723–737
Hoang P et al (2021) Engineering spatial-organized cardiac organoids for developmental toxicity testing. Stem Cell Reports 16(5):1228–1244
Hockemeyer D, Jaenisch R (2016) Induced pluripotent stem cells meet genome editing. Cell Stem Cell 18(5):573–586
Hofbauer P et al (2021) Cardioids reveal self-organizing principles of human cardiogenesis. Cell 184(12):3299-3317.e22
Hughes JP et al (2011) Principles of early drug discovery. Br J Pharmacol 162(6):1239–1249
Kaur S et al (2021) Non-matrigel scaffolds for organoid cultures. Cancer Lett 504:58–66
Kim H et al (2022) Progress in multicellular human cardiac organoids for clinical applications. Cell Stem Cell 29(4):503–514
Kitsuka T et al (2019) 2-Cl-C OXT-A stimulates contraction through the suppression of phosphodiesterase activity in human induced pluripotent stem cell-derived cardiac organoids. PLoS ONE 14(7):e0213114
Lancaster MA et al (2013) Cerebral organoids model human brain development and microcephaly. Nature 501(7467):373–379
Lee J et al (2020) In vitro generation of functional murine heart organoids via FGF4 and extracellular matrix. Nat Commun 11(1):4283
Lewis-Israeli YR et al (2021) Self-assembling human heart organoids for the modeling of cardiac development and congenital heart disease. Nat Commun 12(1):5142
Ma Z et al (2015) Self-organizing human cardiac microchambers mediated by geometric confinement. Nat Commun 6:7413
Mills RJ et al (2017) Functional screening in human cardiac organoids reveals a metabolic mechanism for cardiomyocyte cell cycle arrest. Proc Natl Acad Sci U S A 114(40):E8372-e8381
Mills RJ et al (2019) Drug screening in human PSC-cardiac organoids identifies pro-proliferative compounds acting via the mevalonate pathway. Cell Stem Cell 24(6):895-907.e6
Ming Y et al (2022) Longitudinal morphological and functional characterization of human heart organoids using optical coherence tomography. Biosens Bioelectron 207:114136
Nakano T et al (2012) Self-formation of optic cups and storable stratified neural retina from human ESCs. Cell Stem Cell 10(6):771–785
Noor N et al (2019) 3D Printing of personalized thick and perfusable cardiac patches and hearts. Adv Sci (weinh) 6(11):1900344
Nugraha B et al (2019) Human cardiac organoids for disease modeling. Clin Pharmacol Ther 105(1):79–85
Park SE, Georgescu A (2019) Organoids-on-a-chip. Science 364(6444):960–965
Pauli C et al (2017) Personalized in vitro and in vivo cancer models to guide precision medicine. Cancer Discov 7(5):462–477
Prior N, Inacio P, Huch M (2019) Liver organoids: from basic research to therapeutic applications. Gut 68(12):2228–2237
Richards DJ et al (2020) Human cardiac organoids for the modelling of myocardial infarction and drug cardiotoxicity. Nat Biomed Eng 4(4):446–462
Rossi G et al (2021) Capturing Cardiogenesis in Gastruloids. Cell Stem Cell 28(2):230-240.e6
Roth TL, Marson A (2021) Genetic Disease and Therapy. Annu Rev Pathol 16:145–166
Sato T et al (2009) Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459(7244):262–265
Seguret M et al (2021) Cardiac organoids to model and heal heart failure and cardiomyopathies. Biomedicines 9(5):563
Seidlitz T, Koo BK (2021) Gastric organoids-an in vitro model system for the study of gastric development and road to personalized medicine. Cell Death Differ 28(1):68–83
Shinnawi R et al (2019) Modeling reentry in the short QT syndrome with human-induced pluripotent stem cell-derived cardiac cell sheets. J Am Coll Cardiol 73(18):2310–2324
Skardal A et al (2020) Drug compound screening in single and integrated multi-organoid body-on-a-chip systems. Biofabrication 12(2):025017
Voges HK et al (2017) Development of a human cardiac organoid injury model reveals innate regenerative potential. Development 144(6):1118–1127
Wimmer RA et al (2019) Generation of blood vessel organoids from human pluripotent stem cells. Nat Protoc 14(11):3082–3100
Xiang Y et al (2017) Fusion of regionally specifieD hPSC-derived organoids models human brain development and interneuron migration. Cell Stem Cell 21(3):383-398.e7
Yonatan Israeli, M.G., Kristen Ball, Aaron Wasserman, Jinyun Zou, Guangming Ni, View ORCID ProfileChao Zhou, View ORCID ProfileAitor Aguirre, Generation of Heart Organoids Modeling Early Human Cardiac Development Under Defined Conditions. bioRxiv, 2020.
Funding
This study was supported by the National Key R&D Program of China (2022YFA1104300, 2021YFA1101902), the National Natural Science Foundation of China (82241202, 82170364, 81970223, 82000263), the Natural Science Foundation of Jiangsu Province (BK20201409), Jiangsu Cardiovascular Medicine Innovation Center (CXZX202210).
Author information
Authors and Affiliations
Contributions
JL, JY, DZ, WL and SH: wrote and revised the manuscript. All authors read, edited and approved the manuscript.
Corresponding authors
Ethics declarations
Conflicts of interest
All the authors have no conflicts of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, J., Yang, J., Zhao, D. et al. Promises and challenges of cardiac organoids. Mamm Genome 34, 351–356 (2023). https://doi.org/10.1007/s00335-023-09987-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00335-023-09987-y