Abstract
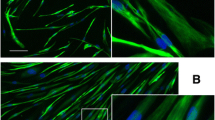
The availability of an in vitro canine cell line would reduce the need for dogs for primary in vitro cell culture and reduce overall cost in pre-clinical studies. An immortalized canine muscle cell line, named Myok9, from primary myoblasts of a normal dog has been developed by the authors. Immortalization was performed by SV40 viral transfection of the large T antigen into the primary muscle cells. Proliferation assays, growth curves, quantitative PCR, western blotting, mass spectrometry, and light microscopy were performed to characterize the MyoK9 cell line at different stages of growth and differentiation. The expression of muscle-related genes was determined to assess myogenic origin. Myok9 cells expressed dystrophin and other muscle-specific proteins during differentiation, as detected with mass spectrometry and western blotting. Using the Myok9 cell line, new therapies before moving to pre-clinical studies to enhance the number and speed of analyses and reduce the cost of early experimentation can be tested now. This cell line will be made available to the research community to further evaluate potential therapeutics.







Similar content being viewed by others
Data availability
All data generated for the LC–MS/MS can be available upon request.
References
Ahuja D, Saenz-Robles MT, Pipas JM (2005) SV40 large T antigen targets multiple cellular pathways to elicit cellular transformation. Oncogene 24:7729–7745. https://doi.org/10.1038/sj.onc.1209046
Bailey GP, Marien D (2011) The value of juvenile animal studies "What have we learned from preclinical juvenile toxicity studies? II". Birth Defects Res B 92:273–291. https://doi.org/10.1002/bdrb.20328
Berg Z, Beffa LR, Cook DP, Cornelison DD (2011) Muscle satellite cells from GRMD dystrophic dogs are not phenotypically distinguishable from wild type satellite cells in ex vivo culture. Neuromuscul Disord 21:282–290. https://doi.org/10.1016/j.nmd.2010.12.009
Buckingham M, Rigby PW (2014) Gene regulatory networks and transcriptional mechanisms that control myogenesis. Dev Cell 28:225–238. https://doi.org/10.1016/j.devcel.2013.12.020
Cannell IG, Kong YW, Bushell M (2008) How do microRNAs regulate gene expression? Biochem Soc Trans 36:1224–1231. https://doi.org/10.1042/BST0361224
Douillard-Guilloux G, Mouly V, Caillaud C, Richard E (2009) Immortalization of murine muscle cells from lysosomal alpha-glucosidase deficient mice: a new tool to study pathophysiology and assess therapeutic strategies for Pompe disease. Biochem Biophys Res Commun 388:333–338. https://doi.org/10.1016/j.bbrc.2009.08.006
Eun K et al (2019) Establishment of TP53-knockout canine cells using optimized CRIPSR/Cas9 vector system for canine cancer research. BMC Biotechnol 19:1. https://doi.org/10.1186/s12896-018-0491-5
Fang J, Wei Y, Teng X, Zhao S, Hua J (2018) Immortalization of canine adipose-derived mesenchymal stem cells and their seminiferous tubule transplantation. J Cell Biochem 119:3663–3670. https://doi.org/10.1002/jcb.26574
Gois Beghini D, Iwao Horita S, Monteiro Fonseca da Cardoso L, Anastacio Alves L, Nagaraju K, Henriques-Pons A (2019) A promising future for stem-cell-based therapies in muscular dystrophies-in vitro and in vivo treatments to boost cellular engraftment. Int J Mol Sci. https://doi.org/10.3390/ijms20215433
Gurda BL, Bradbury AM, Vite CH (2017) Canine and feline models of human genetic diseases and their contributions to advancing clinical therapies. Yale J Biol Med 90:417–431
Haider SR, Wang W, Kaufman SJ (1994) SV40 T antigen inhibits expression of MyoD and myogenin, up-regulates Myf-5, but does not affect early expression of desmin or alpha 7 integrin during muscle development. Exp Cell Res 210:278–286. https://doi.org/10.1006/excr.1994.1040
Health NIo. https://osp.od.nih.gov/scientific-sharing/hela-cells-landing/.
Hong SG et al (2009) Generation of red fluorescent protein transgenic dogs. Genesis 47:314–322. https://doi.org/10.1002/dvg.20504
Hopf-Jensen S, Marques J, Lehrke S, Michael P, Muller-Hulsbeck S (2019) Influence of vessel morphology and anatomical variation on technical and clinical success in thrombectomy: in vitro and in vivo analyses. J Neuroradiol. https://doi.org/10.1016/j.neurad.2019.05.007
Jensen ON, Wilm M, Shevchenko A, Mann M (1999) Sample preparation methods for mass spectrometric peptide mapping directly from 2-DE gels. Methods Mol Biol 112:513–530
Jha KK, Banga S, Palejwala V, Ozer HL (1998) SV40-Mediated immortalization. Exp Cell Res 245:1–7. https://doi.org/10.1006/excr.1998.4272
Kang HY, Choi YK, Jeong YI, Choi KC, Hyun SH, Hwang WS, Jeung EB (2017) Immortalization of porcine 11beta-hydroxysteroid dehydrogenase type 1-transgenic liver cells using SV40 large T antigen. Int J Mol Sci. https://doi.org/10.3390/ijms18122625
Karlgren M et al (2017) A CRISPR-Cas9 generated MDCK cell line expressing human MDR1 without endogenous canine MDR1 (cABCB1): an improved tool for drug efflux studies. J Pharm Sci 106:2909–2913. https://doi.org/10.1016/j.xphs.2017.04.018
Kirchhoff C et al (2004) Immortalization by large T-antigen of the adult epididymal duct epithelium. Mol Cell Endocrinol 216:83–94. https://doi.org/10.1016/j.mce.2003.10.073
Kornegay JN (2017) The golden retriever model of Duchenne muscular dystrophy. Skelet Muscle 7:9. https://doi.org/10.1186/s13395-017-0124-z
Kornegay JN, Childers KM (2016) Canine-inherited dystrophinopathies and centronuclear myopathies. Stem cell biology and regenerative medicine. Humana Press, New York. https://doi.org/10.1007/978-1-4939-3228-3_12
La Rovere RM et al (2014) Myogenic potential of canine craniofacial satellite cells. Front Aging Neurosci 6:90. https://doi.org/10.3389/fnagi.2014.00090
Land H, Parada LF, Weinberg RA (1983) Tumorigenic conversion of primary embryo fibroblasts requires at least two cooperating oncogenes. Nature 304:596–602. https://doi.org/10.1038/304596a0
Li Y, Pan H, Huard J (2010) Isolating stem cells from soft musculoskeletal tissues. J Vis Exp. https://doi.org/10.3791/2011
Lucey BP, Nelson-Rees WA, Hutchins GM (2009) Henrietta Lacks, HeLa cells, and cell culture contamination. Arch Pathol Lab Med 133:1463–1467. https://doi.org/10.1043/1543-2165-133.9.1463
Many GM et al (2016) OPN-a induces muscle inflammation by increasing recruitment and activation of pro-inflammatory macrophages. Exp Physiol 101:1285–1300. https://doi.org/10.1113/EP085768
Mata Lopez S, Balog-Alvarez C, Vitha S, Bettis AK, Canessa EH, Kornegay JN, Nghiem PP (2020) Challenges associated with homologous directed repair using CRISPR-Cas9 and TALEN to edit the DMD genetic mutation in canine Duchenne muscular dystrophy. PLoS ONE 15:e0228072. https://doi.org/10.1371/journal.pone.0228072
Moran JL, Li Y, Hill AA, Mounts WM, Miller CP (2002) Gene expression changes during mouse skeletal myoblast differentiation revealed by transcriptional profiling. Physiol Genomics 10:103–111. https://doi.org/10.1152/physiolgenomics.00011.2002
Motaghed M, Al-Hassan FM, Hamid SS (2013) Cellular responses with thymoquinone treatment in human breast cancer cell line MCF-7. Pharmacognosy Res 5:200–206. https://doi.org/10.4103/0974-8490.112428
Nagata Y, Kiyono T, Okamura K, Goto YI, Matsuo M, Ikemoto-Uezumi M, Hashimoto N (2017) Interleukin-1beta (IL-1beta)-induced Notch ligand Jagged1 suppresses mitogenic action of IL-1beta on human dystrophic myogenic cells. PLoS ONE 12:e0188821. https://doi.org/10.1371/journal.pone.0188821
Nghiem PP, Kornegay JN (2019) Gene therapies in canine models for Duchenne muscular dystrophy. Hum Genet 138:483–489. https://doi.org/10.1007/s00439-019-01976-z
Nghiem PP et al (2017) Osteopontin is linked with AKT, FoxO1, and myostatin in skeletal muscle cells. Muscle Nerve 56:1119–1127. https://doi.org/10.1002/mus.25752
Nichols TC, Hough C, Agerso H, Ezban M, Lillicrap D (2016) Canine models of inherited bleeding disorders in the development of coagulation assays, novel protein replacement and gene therapies. J Thromb Haemost 14:894–905. https://doi.org/10.1111/jth.13301
Park JS et al (2016) Canine cancer immunotherapy studies: linking mouse and human. J Immunother Cancer 4:97. https://doi.org/10.1186/s40425-016-0200-7
Ramboer E, De Craene B, De Kock J, Vanhaecke T, Berx G, Rogiers V, Vinken M (2014) Strategies for immortalization of primary hepatocytes. J Hepatol 61:925–943. https://doi.org/10.1016/j.jhep.2014.05.046
Rotondo JC, Mazzoni E, Bononi I, Tognon M, Martini F (2019) Association between simian virus 40 and human tumors. Front Oncol 9:670. https://doi.org/10.3389/fonc.2019.00670
Sampaolesi M et al (2006) Mesoangioblast stem cells ameliorate muscle function in dystrophic dogs. Nature 444:574–579. https://doi.org/10.1038/nature05282
Sanarica F et al (2019) Proof-of-concept validation of the mechanism of action of Src tyrosine kinase inhibitors in dystrophic mdx mouse muscle: in vivo and in vitro studies. Pharmacol Res 145:104260. https://doi.org/10.1016/j.phrs.2019.104260
Schachter J, Moncada JV (1989) vitro activity of ofloxacin against Chlamydia trachomatis. Am J Med 87:14S–16S
Schneider SM et al (2018) Glucose metabolism as a pre-clinical biomarker for the golden retriever model of Duchenne muscular dystrophy. Mol Imaging Biol. https://doi.org/10.1007/s11307-018-1174-2
Sneddon LU, Halsey LG, Bury NR (2017) Considering aspects of the 3Rs principles within experimental animal biology. J Exp Biol 220:3007–3016. https://doi.org/10.1242/jeb.147058
Song Y et al (2019) Non-immunogenic utrophin gene therapy for the treatment of muscular dystrophy animal models. Nat Med. https://doi.org/10.1038/s41591-019-0594-0
Turner T (2012) Development of the polio vaccine: a historical perspective of Tuskegee University's role in mass production and distribution of HeLa cells. J Health Care Poor Underserved 23:5–10. https://doi.org/10.1353/hpu.2012.0151
Uaesoontrachoon K, Wasgewatte Wijesinghe DK, Mackie EJ, Pagel CN (2013) Osteopontin deficiency delays inflammatory infiltration and the onset of muscle regeneration in a mouse model of muscle injury. Dis Model Mech 6:197–205. https://doi.org/10.1242/dmm.009993
Villarnovo D, McCleary-Wheeler AL, Richards KL (2017) Barking up the right tree: advancing our understanding and treatment of lymphoma with a spontaneous canine model. Curr Opin Hematol 24:359–366. https://doi.org/10.1097/MOH.0000000000000357
Wang M, Wu B, Shah SN, Lu P, Lu Q (2018) Saponins as natural adjuvant for antisense morpholino oligonucleotides delivery in vitro and in mdx mice. Mol Ther Nucleic Acids 11:192–202. https://doi.org/10.1016/j.omtn.2018.02.004
Yagisawa T, Chandhoke PS, Fan J, Lucia S (1998) Renal osteopontin expression in experimental urolithiasis. J Endourol 12:171–176. https://doi.org/10.1089/end.1998.12.171
Yajing S et al (2018) Establishment and characterization of pygmy killer whale (Feresa attenuata) dermal fibroblast cell line. PLoS ONE 13:e0195128. https://doi.org/10.1371/journal.pone.0195128
Yang B, Qiu Y, Zhou N, Ouyang H, Ding J, Cheng B, Sun J (2017) Application of stem cells in oral disease therapy: progresses and perspectives. Front Physiol 8:197. https://doi.org/10.3389/fphys.2017.00197
Zou Q et al (2015) Generation of gene-target dogs using CRISPR/Cas9 system. J Mol Cell Biol 7:580–583. https://doi.org/10.1093/jmcb/mjv061
Acknowledgements
The authors acknowledge Solid Biosciences for providing the funding for this project and for SML’s salary. The Olympus FV1000 confocal microscope acquisition was supported by the Office of the Vice President of Research at Texas A&M University. Finally, they acknowledge Creative Bioarray for immortalizing the cell line.
Funding
This project was funded by Solid Biosciences Inc.
Author information
Authors and Affiliations
Contributions
SML developed the experimental design, data collection, analysis, and manuscript writing. CBA was involved in data analysis, supervision, and experimental design. EC was in charge of mass spectrometry data analysis and acquisition. YH was involved in mass spectrometry data analysis and supervision. KJB was involved in supervision and experimental design. SV was in charge of imaging acquisition. AB was in charge of dog care and colony supervision. JNK was involved in supervision and data interpretation. PPN was involved in experimental design, manuscript writing, data analysis, and supervision. All the authors were involved in manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
SML’s salary was funded by Solid Biosciences. KJB and JB is an employee of Solid Biosciences. JNK is a paid consultant for Solid Biosciences. PPN is a paid consultant for AGADA Biosciences. The rest of the authors declare no competing interests.
Ethical approval
The dogs were used and cared for according to principles outlined in the National Research Council’s Guide for the Care and Use of Laboratory Animals. Procedures were approved by the Texas A&M IACUC through protocols 2018–0182 (Standard Operating Procedures-Canine X-Linked Muscular Dystrophy) and 2017–0147 (Studies of Murine Models for Human Disease and Treatment).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary file5
(DOCX 41 kb)
Rights and permissions
About this article
Cite this article
López, S.M., Balog-Alvarez, C., Canessa, E.H. et al. Creation and characterization of an immortalized canine myoblast cell line: Myok9. Mamm Genome 31, 95–109 (2020). https://doi.org/10.1007/s00335-020-09833-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00335-020-09833-5