Abstract
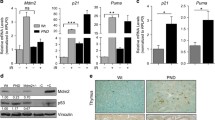
Supv3L1 is a conserved and ubiquitously expressed helicase found in numerous tissues and cell types of many species. In human cells, SUPV3L1 was shown to suppress apoptotic death and sister chromatid exchange, and impair mitochondrial RNA metabolism and protein synthesis. In vitro experiments revealed binding of SUPV3L1 to BLM and WRN proteins, suggesting a role in genome maintenance processes. Disruption of the Supv3L1 gene in the mouse has been reported to be embryonic lethal at early developmental stages. We generated a conditional mouse in which the phenotypes associated with the removal of exon 14 can be tested in a variety of tissues. Disruption mediated by a Mx1 promoter-driven Cre displayed a postnatal growth delay, reduced lifespan, loss of adipose tissue and muscle mass, and severe skin abnormalities manifesting as ichthyosis, thickening of the epidermis, and atrophy of the dermis and subcutaneous tissue. Using a tamoxifen-activatable Esr1/Cre driver, Supv3L1 disruption resulted in growth retardation and aging phenotypes, including loss of adipose tissue and muscle mass, kyphosis, cachexia, and premature death. Many of the abnormalities seen in the Mx1-Cre mice, such as hyperkeratosis characterized by profound scaling of feet and tail, could also be detected in tamoxifen-inducible Cre mice. Conditional ablation of Supv3L1 in keratinocytes confirmed atrophic changes in the skin and ichthyosis-like changes. Together, these data indicate that Supv3L1 is important for the maintenance of the skin barrier. In addition, loss of Supv3L1 function leads to accelerated aging-like phenotypes.








Similar content being viewed by others
References
Adams DJ, Biggs PJ, Cox T, Davies R, van der Weyden L et al (2004) Mutagenic insertion and chromosome engineering resource (MICER). Nat Genet 36:867–871
Arnheiter H, Skuntz S, Noteborn M, Chang S, Meier E (1990) Transgenic mice with intracellular immunity to influenza virus. Cell 62:51–61
Babbe H, Chester N, Leder P, Reizis B (2007) The Bloom’s syndrome helicase is critical for development and function of the alphabeta T-cell lineage. Mol Cell Biol 27:1947–1959
Boehncke WH, Schon MP (2007) Animal models of psoriasis. Clin Dermatol 25:596–605
Butow RA, Zhu H, Perlman P, Conrad-Webb H (1989) The role of a conserved dodecamer sequence in yeast mitochondrial gene expression. Genome 31:757–760
Campisi J, d’Adda di Fagagna F (2007) Cellular senescence: when bad things happen to good cells. Nat Rev Mol Cell Biol 8:729–740
Collado M, Blasco MA, Serrano M (2007) Cellular senescence in cancer and aging. Cell 130:223–233
Conrad-Webb H, Perlman PS, Zhu H, Butow RA (1990) The nuclear SUV3-1 mutation affects a variety of post-transcriptional processes in yeast mitochondria. Nucleic Acids Res 18:1369–1376
Danielian PS, Muccino D, Rowitch DH, Michael SK, McMahon AP (1998) Modification of gene activity in mouse embryos in utero by a tamoxifen-inducible form of Cre recombinase. Curr Biol 8:1323–1326
de Boer J, de Wit J, van Steeg H, Berg RJ, Morreau H et al (1998) A mouse model for the basal transcription/DNA repair syndrome trichothiodystrophy. Mol Cell 1:981–990
Dmochowska A, Kalita K, Krawczyk M, Golik P, Mroczek K et al (1999) A human putative Suv3-like RNA helicase is conserved between Rhodobacter and all eukaryotes. Acta Biochim Pol 46:155–162
Dziembowski A, Piwowarski J, Hoser R, Minczuk M, Dmochowska A et al (2003) The yeast mitochondrial degradosome. Its composition, interplay between RNA helicase and RNase activities and the role in mitochondrial RNA metabolism. J Biol Chem 278:1603–1611
Fawell SE, Lees JA, White R, Parker MG (1990) Characterization and colocalization of steroid binding and dimerization activities in the mouse estrogen receptor. Cell 60:953–962
Fehling HJ, Krotkova A, Saint-Ruf C, von Boehmer H (1995) Crucial role of the pre-T-cell receptor alpha gene in development of alpha beta but not gamma delta T cells. Nature 375:795–798
German J (1995) Bloom’s syndrome. Dermatol Clin 13:7–18
Giorgio M, Trinei M, Migliaccio E, Pelicci PG (2007) Hydrogen peroxide: a metabolic by-product or a common mediator of ageing signals? Nat Rev Mol Cell Biol 8:722–728
Gudjonsson JE, Johnston A, Dyson M, Valdimarsson H, Elder JT (2007) Mouse models of psoriasis. J Invest Dermatol 127:1292–1308
Hantke J, Rogers T, French L, Tournev I, Guergueltcheva V et al (2003) Refined mapping of the HMSNR critical gene region—construction of a high-density integrated genetic and physical map. Neuromuscul Disord 13:729–736
Hayashi S, McMahon AP (2002) Efficient recombination in diverse tissues by a tamoxifen-inducible form of Cre: a tool for temporally regulated gene activation/inactivation in the mouse. Dev Biol 244:305–318
Hickson ID (2003) RecQ helicases: caretakers of the genome. Nat Rev Cancer 3:169–178
Hoffman ES, Passoni L, Crompton T, Leu TM, Schatz DG et al (1996) Productive T-cell receptor beta-chain gene rearrangement: coincident regulation of cell cycle and clonality during development in vivo. Genes Dev 10:948–962
Hoki Y, Araki R, Fujimori A, Ohhata T, Koseki H et al (2003) Growth retardation and skin abnormalities of the Recql4-deficient mouse. Hum Mol Genet 12:2293–2299
Horisberger MA, De Staritzky K (1989) Expression and stability of the Mx protein in different tissues of mice, in response to interferon inducers or to influenza virus infection. J Interferon Res 9:583–590
Khidr L, Wu G, Davila A, Procaccio V, Wallace D et al (2008) Role of SUV3 helicase in maintaining mitochondrial homeostasis in human cells. J Biol Chem 283:27064–27073
Klysik J, Singer JD (2005) Mice with the enhanced green fluorescent protein gene knocked in to chromosome 11 exhibit normal transmission ratios. Biochem Genet 43:321–333
Kuhn R, Schwenk F, Aguet M, Rajewsky K (1995) Inducible gene targeting in mice. Science 269:1427–1429
Littlewood TD, Hancock DC, Danielian PS, Parker MG, Evan GI (1995) A modified oestrogen receptor ligand-binding domain as an improved switch for the regulation of heterologous proteins. Nucleic Acids Res 23:1686–1690
Liu P, Jenkins NA, Copeland NG (2003) A highly efficient recombineering-based method for generating conditional knockout mutations. Genome Res 13:476484
Lloyd C, Yu QC, Cheng J, Turksen K, Degenstein L et al (1995) The basal keratin network of stratified squamous epithelia: defining K15 function in the absence of K14. J Cell Biol 129:1329–1344
Maeda I, Kohara Y, Yamamoto M, Sugimoto A (2001) Large-scale analysis of gene function in Caenorhabditis elegans by high-throughput RNAi. Curr Biol 11:171–176
Magin TM (1998) Lessons from keratin transgenic and knockout mice. Subcell Biochem 31:141–172
Mann MB, Hodges CA, Barnes E, Vogel H, Hassold TJ et al (2005) Defective sister-chromatid cohesion, aneuploidy and cancer predisposition in a mouse model of type II Rothmund-Thomson syndrome. Hum Mol Genet 14:813–825
Margossian SP, Li H, Zassenhaus HP, Butow RA (1996) The DExH box protein Suv3p is a component of a yeast mitochondrial 3′-to-5′ exoribonuclease that suppresses group I intron toxicity. Cell 84:199–209
Mattioni T, Louvion JF, Picard D (1994) Regulation of protein activities by fusion to steroid binding domains. Methods Cell Biol 43 Pt A:335–352
Minczuk M, Piwowarski J, Papworth MA, Awiszus K, Schalinski S et al (2002) Localisation of the human hSuv3p helicase in the mitochondrial matrix and its preferential unwinding of dsDNA. Nucleic Acids Res 30:5074–5086
Minczuk M, Mroczek S, Pawlak SD, Stepien PP (2005) Human ATP-dependent RNA/DNA helicase hSuv3p interacts with the cofactor of survivin HBXIP. FEBS J 272:5008–5019
Moll R, Franke WW, Schiller DL, Geiger B, Krepler R (1982) The catalog of human cytokeratins: patterns of expression in normal epithelia, tumors and cultured cells. Cell 31:11–24
Nehlin JO, Skovgaard GL, Bohr VA (2000) The Werner syndrome. A model for the study of human aging. Ann N Y Acad Sci 908:167–179
Oberdoerffer P, Sinclair DA (2007) The role of nuclear architecture in genomic instability and ageing. Nat Rev Mol Cell Biol 8:692–702
Pereira M, Mason P, Szczesny RJ, Maddukuri L, Dziwura S et al (2007) Interaction of human SUV3 RNA/DNA helicase with BLM helicase: loss of the SUV3 gene results in mouse embryonic lethality. Mech Aging Dev 128:609–617
Picard D (1994) Regulation of protein function through expression of chimaeric proteins. Curr Opin Biotechnol 5:511–515
Promislow DE (1994) DNA repair and the evolution of longevity: a critical analysis. J Theor Biol 170:291–300
Ramirez-Solis R, Liu P, Bradley A (1995) Chromosome engineering in mice. Nature 378:720–724
Rogers T, Chandler D, Angelicheva D, Thomas PK, Youl B et al (2000) A novel locus for autosomal recessive peripheral neuropathy in the EGR2 region on 10q23. Am J Hum Genet 67:664–671
Schneider A, Zhang Y, Guan Y, Davis LS, Breyer MD (2003) Differential, inducible gene targeting in renal epithelia, vascular endothelium, and viscera of Mx1Cre mice. Am J Physiol Renal Physiol 284:F411–F417
Schon MP (1999) Animal models of psoriasis—what can we learn from them? J Invest Dermatol 112:405–410
Sharpless NE, DePinho RA (2007) How stem cells age and why this makes us grow old. Nat Rev Mol Cell Biol 8:703–713
Shu Z, Vijayakumar S, Chen CF, Chen PL, Lee WH (2004) Purified human SUV3p exhibits multiple-substrate unwinding activity upon conformational change. Biochemistry 43:4781–4790
Slijepcevic P (2007) DNA damage response, telomere maintenance and ageing in light of the integrative model. Mech Ageing Dev 129:11–16
Stepien PP, Margossian SP, Landsman D, Butow RA (1992) The yeast nuclear gene suv3 affecting mitochondrial post-transcriptional processes encodes a putative ATP-dependent RNA helicase. Proc Natl Acad Sci USA 89:6813–6817
Sun LQ, Lee DW, Zhang Q, Xiao W, Raabe EH et al (2004) Growth retardation and premature aging phenotypes in mice with disruption of the SNF2-like gene, PASG. Genes Dev 18:1035–1046
Szczesny RJ, Obriot H, Paczkowska A, Jedrzejczak R, Dmochowska A et al (2007) Down-regulation of human RNA/DNA helicase SUV3 induces apoptosis by a caspase- and AIF-dependent pathway. Biol Cell 99:323–332
Taylor RW, Turnbull DM (2005) Mitochondrial DNA mutations in human disease. Nat Rev Genet 6:389–402
Terman A, Brunk UT (2004) Aging as a catabolic malfunction. Int J Biochem Cell Biol 36:2365–2375
Thomas PK, Kalaydjieva L, Youl B, Rogers T, Angelicheva D et al (2001) Hereditary motor and sensory neuropathy-russe: new autosomal recessive neuropathy in Balkan Gypsies. Ann Neurol 50:452–457
Tyynismaa H, Mjosund KP, Wanrooij S, Lappalainen I, Ylikallio E et al (2005) Mutant mitochondrial helicase Twinkle causes multiple mtDNA deletions and a late-onset mitochondrial disease in mice. Proc Natl Acad Sci USA 102:17687–17692
Vasioukhin V, Degenstein L, Wise B, Fuchs E (1999) The magical touch: genome targeting in epidermal stem cells induced by tamoxifen application to mouse skin. Proc Natl Acad Sci USA 96:8551–8556
Vennos EM, James WD (1995) Rothmund-Thomson syndrome. Dermatol Clin 13:143–150
Vennos EM, Collins M, James WD (1992) Rothmund-Thomson syndrome: review of the world literature. J Am Acad Dermatol 27:750–762
Wang X, Zinkel S, Polonsky K, Fuchs E (1997) Transgenic studies with a keratin promoter-driven growth hormone transgene: prospects for gene therapy. Proc Natl Acad Sci USA 94:219–226
Wanrooij S, Luoma P, van Goethem G, van Broeckhoven C, Suomalainen A et al (2004) Twinkle and POLG defects enhance age-dependent accumulation of mutations in the control region of mtDNA. Nucleic Acids Res 32:3053–3064
Warming S, Costantino N, Court DL, Jenkins NA, Copeland NG (2005) Simple and highly efficient BAC recombineering using galK selection. Nucleic Acids Res 33:e36
Xia YP, Zhao Y, Marcus J, Jimenez PA, Ruben SM et al (1999) Effects of keratinocyte growth factor-2 (KGF-2) on wound healing in an ischaemia-impaired rabbit ear model and on scar formation. J Pathol 188:431–438
Zhu H, Conrad-Webb H, Liao XS, Perlman PS, Butow RA (1989) Functional expression of a yeast mitochondrial intron-encoded protein requires RNA processing at a conserved dodecamer sequence at the 3’ end of the gene. Mol Cell Biol 9:1507–1512
Acknowledgments
The authors thank Paul Monfills and Mandy Pereira for their assistance with preparation of tissue sections and collecting the images. This work was supported by grants 5P20RR015578-07 and P42ES013660 from the National Institute of Environmental Health Sciences. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Environmental Health Sciences or the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Paul, E., Cronan, R., Weston, P.J. et al. Disruption of Supv3L1 damages the skin and causes sarcopenia, loss of fat, and death. Mamm Genome 20, 92–108 (2009). https://doi.org/10.1007/s00335-008-9168-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00335-008-9168-z