Abstract
Objectives
To evaluate the diagnostic performance of ultrasound guided attenuation parameter (UGAP) for evaluating liver fat content with different probe forces and body positions, in relation to sex, and compared with proton density fat fraction (PDFF).
Methods
We prospectively enrolled a metabolic dysfunction-associated steatotic liver disease (MASLD) cohort that underwent UGAP and PDFF in the autumn of 2022. Mean UGAP values were obtained in supine and 30° left decubitus body position with normal 4 N and increased 30 N probe force. The diagnostic performance was evaluated by the area under the receiver operating characteristic curve (AUC).
Results
Among 60 individuals (mean age 52.9 years, SD 12.9; 30 men), we found the best diagnostic performance with increased probe force in 30° left decubitus position (AUC 0.90; 95% CI 0.82–0.98) with a cut-off of 0.58 dB/cm/MHz. For men, the best performance was in supine (AUC 0.91; 95% CI 0.81–1.00) with a cut-off of 0.60 dB/cm/MHz, and for women, 30° left decubitus position (AUC 0.93; 95% CI 0.83–1.00), with a cut-off 0.56 dB/cm/MHz, and increased 30 N probe force for both genders. No difference was in the mean UGAP value when altering body position. UGAP showed good to excellent intra-reproducibility (Intra-class correlation 0.872; 95% CI 0.794–0.921).
Conclusion
UGAP provides excellent diagnostic performance to detect liver fat content in metabolic dysfunction-associated steatotic liver diseases, with good to excellent intra-reproducibility. Regardless of sex, the highest diagnostic accuracy is achieved with increased probe force with men in supine and women in 30° left decubitus position, yielding different cut-offs.
Clinical relevance statement
The ultrasound method ultrasound-guided attenuation parameter shows excellent diagnostic accuracy and performs with good to excellent reproducibility. There is a possibility to alter body position and increase probe pressure, and different performances for men and women should be considered for the highest accuracy.
Key Points
• There is a possibility to alter body position when performing the ultrasound method ultrasound-guided attenuation parameter.
• Increase probe pressure for the highest accuracy.
• Different performances for men and women should be considered.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Quantitative ultrasound (QUS) attenuation imaging of liver fat content can be difficult to perform depending on how the patient participates and their body constitution. How probe pressure and body position affect the diagnostic accuracy of QUS, especially in relation to sex, is currently unknown.
The estimated global prevalence of metabolic dysfunction-associated steatotic liver diseases (MASLD) is as high as 38% in some regions [1]. A recent nationwide adult cohort study in Sweden found a significantly higher hazard ratio for overall mortality in all individuals with MASLD, including simple steatosis, compared to controls [2].
The presence of hepatic steatosis is a prerequisite for diagnosing MASLD [3, 4]. Liver biopsy (LB) used to be the gold standard for determining liver fat content but has a risk of adverse events [5,6,7,8]. However, the proton density fat fraction (PDFF) determined by magnetic resonance imaging (MRI) is an excellent non-invasive method, surpassing and replacing LB as the reference method for hepatic fat quantification [9,10,11,12]. MRI has some limitations of accessibility and contraindications; therefore, conventional ultrasound (US) is recommended for diagnosing MASLD [13]. US uses a qualitative grading scale [14] with 85% sensitivity in grading S2–S3 [14, 15], but only 12% sensitivity in grading S0–S1 [16], as underlying liver diseases and low reproducibility reduce accuracy [17,18,19].
The US wave loses energy (i.e., attenuates) as it propagates in the tissue, and the attenuation can be quantitatively determined [20, 21]. All imaging modes in US start from radiofrequency (RF) data processing, and QUS for liver fat content is mostly based on RF data for calculation of the speed of sound, attenuation, and backscatter. Several studies have been performed for QUS determination of liver fat content with different technologies and US systems [22,23,24,25,26,27,28,29,30,31,32]. However, fat distribution differs between men and women [33], and reliability is important regardless of sex. To date, no standardized method for evaluating liver fat content by QUS has been published, and how increased probe pressure and body position affect diagnostic performance with the new technology US-guided attenuation parameter (UGAP) is not known [34].
The aims of the present study were to investigate the influence of probe pressure and body position on the diagnostic accuracy of UGAP in individuals with and without MASLD and to examine whether different thresholds are warranted for men and women for identifying the presence of hepatic steatosis (i.e., steatosis more than 5% according to MRI).
Materials and methods
This prospective single-center study was approved by the Swedish Ethical Review Authority (No. 2022–01610-01). The research was performed in accordance with The World Medical Association Declaration of Helsinki, 2013. All study participants provided informed written consent. Data collection was performed between 24 September and 10 December 2022.
Study population
Patients with a clinically verified diagnosis of MASLD were asked to participate in this study. Inclusion was based on the availability of a magnetic resonance examination including an MRI-PDFF > 10% performed within the last 12 months. A control population of healthy individuals with low risk of MASLD (i.e., BMI < 25 kg/m2 and absence of components of metabolic syndrome) were also asked to participate in this study. The study population quota was deemed met when 40 individuals with ≥ 5% liver fat content and 20 individuals with < 5% liver fat content according to PDFF had been included and equal distribution of sex was fulfilled in the whole study group. All patients were recruited at the Department of Gastroenterology and Hepatology, Linköping University Hospital. Exclusion criteria were declining to participate, age < 18 years, pregnancy, or contraindications to MRI (i.e., claustrophobia or presence of medical implants or implants with ferromagnetic properties). Thus, a total of 60 individuals were included after giving written informed consent (Fig. 1).
Ultrasound measurements and performance
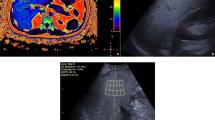
The US-guided attenuation parameter (UGAP) was measured using a LOGIQ E10 rev 3 US system (General Electric Healthcare) with a convex probe (C1-6). In the UGAP program, the US display was divided into two, with a quality map to select the best image for the attenuation coefficient measure and an attenuation map where attenuation is quantified and shown in a large area using a real-time color-coded map. With the UGAP technique, an entire measurement loop is automatically recorded from five measurements, skipping 10 frames between each measurement. Measurements were performed with a rectangular-shaped region of interest (ROI), the position of which could be altered only in the lateral direction in the liver parenchyma. Reliable UGAP measurements were defined as the median values of five measurements with a ratio of the interquartile range of the attenuation value to the median being < 30% and an approved quality map. Failure was defined as a red-marked ROI in the display attenuation map. UGAP values were expressed in dB/cm/MHz and obtained from a homogeneous area of liver parenchyma free of big vessels and rib shadow with the patient’s right arm elevated above their head.
Five UGAP measurements were performed with normal 4 Newton (N) or increased 30 N probe force and with the body in supine or 30° left decubitus position using two identical pillows to ensure an identical 30° position. For a description of applied probe force measurements, see Supplementary Material 1 (Method description of applied probe force measurements).
To evaluate the reliability of UGAP, sixty measurements were performed in the supine position with normal 4 N probe force, and sampling was repeated approximately 1–3 months later with the same operator, blinded to the earlier results, from recorded volumes.
All UGAP measurements were obtained on the same day as PDFF and magnetic resonance elastography (MRE), and all operators were blinded to each method´s results. Prior to the examination, all participants were fasting for at least 5 h.
As steatosis in early stages commonly has heterogeneous distribution in the liver, there may be a need for increased exactness in the evaluation of the diagnostic performance of UGAP and the influence of probe force, which is provided in Supplementary Material 2 (Evaluation of UGAP diagnostic performance with single measurement in same spot of liver parenchyma and Supplementary Material 3, Evaluation of probe force with single measurement in same spot of liver parenchyma).
Magnetic resonance measurements and performance
PDFF and MRE were performed using an Ingenia 3 T (Philips Healthcare, Best). The PDFF [35] covered the entire liver, and the MRE acquisitions were planned in four slices according to a standardized protocol [36]. Complete information is provided in Supplementary Material 4 (Method description of magnetic resonance measurements).
Statistical analysis
The normality of the continuous data for UGAP and MRI-PDFF was tested by the one-sample Kolmogorov–Smirnov test.
Intra-class correlations (ICCs) were estimated for UGAP, and their 95% confidence intervals (CIs) were based on a mean-rating (k = 2), absolute-agreement, and 2-way mixed-effects model. ICC values < 0.50 indicate poor reliability, values between 0.50 and 0.75 indicate moderate reliability, values between 0.75 and 0.90 indicate good reliability, and values > 0.90 indicate excellent reliability [37].
The paired t-test was used to evaluate differences between mean UGAP values in different body positions and different probe forces.
To evaluate the diagnostic accuracy, ROC analysis was performed. From the area under the receiver operating characteristic curve (AUC), a cut-off was determined for the mean UGAP value at sensitivity > 90% and 95% and specificity > 90% and 95%. We used an MRI-PDFF cut-off of 5% to define UGAP diagnostic performance for distinguishing steatosis grade S0 vs. ≥ S1. The evaluation was made in different body positions and probe forces and in subgroups of men and women.
The corresponding levels of MRI-PDFF for the different grades of steatosis were set in accordance with Imajo et al [30]: steatosis grade 1 (S1) = 5.0–11.2%, S2 = 11.3–17.0%, and S3 ≥ 17.1%.
The corresponding levels of MRE for the different grades of fibrosis were set in accordance with Guglielmo et al [38]: normal = < 2.9 kPa, fibrosis stage 1 = 2.9–3.5 kPa, fibrosis stage 2 = 3.5–4.0, fibrosis stage 3 = 4.0–5.0 kPa, and fibrosis stage 4 or cirrhosis = > 5.0 kPa.
Continuous variables are expressed as the mean ± standard deviation (min–max). Categorical variables are expressed as frequency and percentage.
For all statistical tests, 95% CIs were calculated. A p value < 0.05 was considered significant, and all statistical tests were two-sided.
Statistical analyses were performed in the software Statistical Package for Social Sciences (SPSS) version 29.
Results
No technical failure occurred in the performance of technical measures of UGAP. A total of 60 individuals (30 men, mean age 52.9 years ± 12.9 and 30 women, 53.9 years ± 11.6) were included in the analysis, including 40 individuals with MASLD and 20 control individuals without MASLD. The characteristics of all participants in the study, stratified by sex, are presented in Table 1.
Reliability of UGAP
Sixty measurements performed with the body in the supine position with normal 4 N probe force yielded a mean UGAP value of 0.65 ± 0.14 dB/cm/MHz. Approximately 1–3 months later, the same operator, blinded to the earlier results, obtained 60 measurements from recorded volumes, yielding a mean UGAP value of 0.66 ± 0.12 dB/cm/MHz. There was no significant difference in UGAP values between the two time points (p = 0.38, N = 60), and the estimated ICC was 0.872 (95% CI 0.794–0.921). Based on the ICC, the intra-rater and test–retest reliability of this new method shows good to excellent reliability.
UGAP performance for staging hepatic steatosis in MASLD
There was a significant difference between hepatic steatosis stages S0–S3 in the supine position with normal 4 N probe force (Fig. 2). Differences in the mean UGAP values were S0 0.53 vs. S1 0.61 dB/cm/MHz (p = 0.027, n = 20; n = 18), S1 0.61 vs. S2 0.71 dB/cm/MHz (p = 0.025, n = 18; n = 8), S2 0.71 vs. S3 0.84 dB/cm/MHz (p = 0.003, n = 8; n = 14).
UGAP diagnostic performance for staging hepatic steatosis in MASLD. There was a significant difference between hepatic steatosis stages S0-S3 in the supine position with normal 4 N probe force. S0, n = 20; S1, n = 18; S2, n = 8; S3, n = 14. Differences in the mean UGAP values: S0 0.53 vs. S1 0.61 dB/cm/MHz (p = 0.027), S1 0.61 vs. S2 0.71 dB/cm/MHz (p = 0.025), S2 0.71 vs. S3 0.84 dB/cm/MHz (p = 0.003). MASLD= metabolic dysfunction-associated steatotic liver disease (defined as ≥ 5% MRI-PDFF), UGAP = ultrasound-guided attenuation parameter, MRI = magnetic resonance imaging, PDFF = proton density fat fraction. PDFF defined cut-offs for steatosis grade 1 (S1) = 5.0–11.2%, S2 = 11.3–17.0%, and S3 ≥ 17.1%
Influence of probe force and body position in UGAP
For the whole group in a supine position, there was a significant difference between the mean UGAP values obtained with normal 4 N and increased 30 N probe force (p < 0.001, N = 60). However, there was no significant difference with the body in the 30° left decubitus position (p = 0.195, n = 59). For men, there was no difference in the supine (p = 0.093, n = 30) or 30° left decubitus position (p = 0.104, n = 29). For women, there was a difference in the supine position (p = 0.004, n = 30), but not in the 30° left decubitus position (p = 0.104, n = 30; Table 2).
No significant differences were found for the mean UGAP values in different body positions (Table 3).
Increased probe force showed a significant impact on skin-to-liver capsule distance; complete results are provided in Supplementary Material 5 (Results for probe force influence on skin-to-liver capsule distance).
Validity of UGAP in MASLD
For the whole group, the best diagnostic performance of UGAP was found with the body in the 30° left decubitus position with increased 30 N probe force (AUC 0.90, 95% CI 0.82–0.98) with a cut-off of 0.58 dB/cm/MHz, 90% sensitivity, and 70% specificity (Fig. 3 and Table 4).
AUC plots illustrating the diagnostic performance of the ultrasound-guided attenuation parameter (UGAP) for the body in a supine position with normal 4 N probe force, yielding an AUC of 0.87 (95% CI 0.77–0.97), and with increased 30 N probe force (AUC 0.90, 95% CI 0.82–0.98). Body in 30° left decubitus body position with normal 4 N probe force yielded an AUC of 0.87 (95% CI 0.76–0.98), and with increased 30 N probe force an AUC of 0.90 (95% CI 0.82–0.98). MRI-PDFF ≥ 5% was defined as the cut-off for steatosis grade S1. CI = confidence interval, AUC = area under the curve, MASLD= metabolic dysfunction-associated steatotic liver disease (defined as ≥ 5% MRI-PDFF), MRI = magnetic resonance imaging, PDFF = proton density fat fraction
In the group of men, the best performance was in the supine position with increased 30 N probe force (AUC 0.91, 95% CI 0.80–1.00), with a cut-off of 0.60 dB/cm/MHz, sensitivity of 90%, and specificity of 82%. For women, the best performance was in the 30° left decubitus position with increased 30 N probe force (AUC 0.93, (95% CI 0.82–1.00), with a cut-off of 0.56 dB/cm/MHz, sensitivity of 95%, and specificity of 78% (Figs. 4 and 5 and Table 4).
AUC plots illustrating the diagnostic performance of ultrasound-guided attenuation parameter (UGAP) for men (n = 30). UGAP was performed with normal 4 N force, increased 30 N probe force, and with the body in the supine or 30° left decubitus position. MRI-PDFF < 5% was the cut-off for steatosis grade S0 (n = 11) and ≥ S1 (n = 19). With the body in the supine position and with normal 4 N probe force had an AUC of 0.83 (95% CI 0.67–0.99), and with increased 30 N probe force an AUC of 0.91 (95% CI 0.81–1.00). With the body in 30° left decubitus position and with normal 4 N probe force, the AUC was 0.85 (95% CI 0.68–1.00), and with increased 30 N probe force 0.89 (95% CI 0.77–1.00). AUC = area under the curve, CI = confidence interval, MASLD= metabolic dysfunction-associated steatotic liver disease (defined as ≥ 5% MRI-PDFF), MRI = magnetic resonance imaging, PDFF = proton density fat fraction
AUC plots illustrating the diagnostic performance of ultrasound-guided attenuation parameter (UGAP) for women (n = 30). UGAP was performed with normal 4 N probe force, increased 30 N probe force, and with the body in the supine or 30° left decubitus position. MRI-PDFF < 5% was the cut-off for steatosis grade S0 (n = 11) and ≥ S1 (n = 19). The supine position with normal 4 N probe force had an AUC of 0.93 (95% CI 0.82–1.00) and with increased 30 N probe force 0.90 (95% CI 0.79–1.00). With the body in 30° left decubitus position with normal 4 N probe force, the AUC was 0.88 (95% CI 0.76–1.00), and with increased 30 N probe force 0.93 (95% CI 0.83–1.00). AUC = area under the curve, CI = confidence interval, MASLD= metabolic dysfunction-associated steatotic liver disease (defined as ≥ 5% MRI-PDFF), MRI = magnetic resonance imaging, PDFF = proton density fat fraction
With normal 4 N probe force, the best diagnostic accuracy for men was with the body in the 30° left decubitus position (AUC 0.85, 95% CI 0.68–1.00), with a cut-off of 0.58 dB/cm/MHz, 90% sensitivity, and 82% specificity. For women, the best diagnostic accuracy was with the body in the supine position (AUC 0.93, 95% CI 0.82–1.00), with a cut-off of 0.52 dB/cm/MHz, 95% sensitivity, and 78% specificity (Figs. 4 and 5 and Table 4).
MRI PDFF and MRE measurement results
Using Imajio et al PDFF defined cut-offs for liver steatosis grade S0–S3 yielded in 20 participants with S0, 18 participants with S1, 8 participants with S2, and 14 participants with S3.
Using Guglielmo et al defined cut-offs for liver fibrosis grades 0–4 yielded in 58 participants with no liver fibrosis (< 2.9 kPa), 1 participant with liver fibrosis stages 1–2 (2.9–3.5 kPa) and steatosis grade S3, and 1 participant with liver fibrosis stages 2–3 (3.5–4.0 kPa) and steatosis grade S2.
Discussion
We assessed 60 individuals using the new QUS method UGAP to stage liver fat content in different body positions and with different probe forces and assessed men and women separately to detect potential sex differences. The results showed that an increased 30 N probe force yielded increased mean UGAP values and higher diagnostic accuracy compared to normal 4 N probe force, body position did not influence the mean UGAP values, and differences between sexes indicated different examination techniques for men and women.
To the best of our knowledge, no study has been conducted with increased probe force or to evaluate the differences between men and women examined by QUS for liver fat content. Consistent with our results, earlier studies of UGAP have shown good inter-reproducibility [39] and no correlation with liver fibrosis [40, 41].
An intercostal approach in the supine position with normal probe force is recommended for the estimation of liver fat content by US attenuation [42]. The distance to the liver increases in obesity and overweight, and the B-mode image of US is known to be affected during these conditions. Therefore, altering body position and increasing probe pressure are common techniques to obtain acceptable B-mode images in abdominal US examinations. However, the possibility of using these examination techniques for UGAP has not yet been evaluated. In the present study, we showed that the body positions from supine to 30° left decubitus are interchangeable, and higher mean UGAP values were obtained with increased 30 N probe force, though the best accuracy was obtained with higher probe force.
There are well-known differences between the sexes in how body fat is distributed; women have a greater ability to store fat in subcutaneous sections, whereas men have more visceral fat than women [43, 44], which may indicate a need for different examination techniques for men and women to achieve higher diagnostic accuracy. In the present study, the best diagnostic accuracy for women was obtained with the body in the 30° left decubitus position with increased 30 N probe force and for men with the body in the supine position with increased 30 N probe force. Accordingly, our results suggest that the operator should consider the patient’s sex when performing UGAP measurements and consider the use of different cut-offs for women and men. Interpreting our results in clinical settings may result in reclassification of the diagnosis.
The impact on the reliability of QUS due to increased distance to the liver has been confirmed previously [45]. Increased probe pressure induces tissue deformation in the subcutaneous layers between the probe and costal rib, and the increased 30 N probe force in our study decreased the skin-to-liver capsule distance. In comparison, in QUS shear wave elastography for liver fibrosis, the use of increased probe pressure yielded improved technical success with more feasible investigations [46]. The positive effect of increased probe force can be explained by a reduced impact of scattering, refraction, absorption, and reflection in the boundary region, which enables the US waves to better traverse in compressed subcutaneous fatty layers than non-compressed layers between the skin and costal rib, with less energy loss. This hypothesis is consistent with earlier findings implying that the QUS shear wave method is affected by tissue attenuation [47]. Also, increased skin-to-liver capsule distance has higher amounts of aberrations in the RF signal which might have a negative impact on QUS attenuation estimation, and has been earlier discussed as a possible confounding factor [31].
In our study, we determined the attenuation coefficient in an MASLD cohort with equal numbers of men and women, which is a strength. Liver fat content was determined with PDFF as the reference method for all participants, which also is a strength, and UGAP and MRI measurements were performed on the same day for each participant. Moreover, this study was performed with UGAP rev 3 in US device GE E10 LOGIQ and defined PDFF steatosis stages were similar to previously published data, which ensures the possibility of reliable comparisons [30]. In our study, 58 participants did not have fibrosis according to our MRE measurements. Only 1 participant with liver fibrosis stages 1–2 (2.9–3.5 kPa) and steatosis grade S3, and 1 participant with liver fibrosis stages 2–3 (3.5–4.0 kPa) and steatosis grade S2. It is known that with increasing levels of fibrosis, the grade of steatosis declines over time, and can therefore be seen as a confounding factor in determining the presence of steatosis. Therefore, another strength in our study is the evaluation of fibrosis performed with MRE which is valuable for comparison with other similar research. It is also valuable since we do not know the full impact of liver fibrosis on hepatic steatosis measurements with UGAP.
This work has some limitations. The clinical usefulness of our results may be limited due to a lack of available commercial probe pressure devices, and a technical solution for recording the probe pressure was previously requested for QUS [48]. However, we showed that as much as 30 N of probe force does not compromise the UGAP attenuation coefficient results, but indicated that increased probe force is beneficial for increased diagnostic accuracy. Moreover, only one operator performed UGAP. Error sources, such as backscattering variation, speed of sound variation, focus location, imaging artifacts, image resolution, and signal-to-noise ratio [42], can increase intra-inter operator variability, which may impact the benefits of increased probe force for diagnostic accuracy. The differences found in our study between men and women should also be evaluated in a larger cohort to confirm the need for different performance and cut-offs for men and women for the highest diagnostic accuracy. Another limitation is the lack of liver biopsy used as reference for hepatic steatosis stages S2 and S3 in this study; however, the cut-off to detect hepatic steatosis ≥ S1 used in this study was 5%.
There are several commercial QUS methods available for measuring liver fat content, though not all possible confounders have been evaluated [31, 32, 49]. Moreover, in medical imaging, different patterns of hepatic steatosis have been described (diffuse, geographic, focal, subcapsular, multifocal, perivascular) [50, 51]. In cases of geographic pattern, with UGAP, there is the possibility to alter the ROI in lateral directions to select parts in the liver parenchyma with increased fat content, but how to perform measurements with QUS in these cases has not yet been discussed. In UGAP the ROI size and position in depth are fixed, due to the integrated reference phantom, which seems to be beneficial compared to other technologies in which high variability is seen for attenuation estimates depending on ROI depth [26]. In lower steatosis stages, the distribution of steatosis is known to be heterogenous, which has an impact on diagnostic accuracy. However, with single measurements and small PDFF ROIs, we showed excellent diagnostic performance for UGAP, Supplementary Material 2, (Evaluation of UGAP diagnostic performance with single measurement in same spot of liver parenchyma). A standardized method is needed for the estimation of liver fat content by QUS attenuation.
Conclusion
The UGAP provides excellent diagnostic performance to detect ≥ S1 in MASLD (defined as MRI-PDFF 5%) and has good to excellent reproducibility. Altering body position to 30° left decubitus is possible when performing UGAP, and increased 30 N probe force results in increased mean UGAP values. Regardless of sex, the highest diagnostic accuracy is obtained with increased probe force, with men in the supine and women in the 30° left decubitus position, rendering different cut-offs, which should be considered to avoid missing individuals with increased hepatic steatosis. We therefore conclude that both probe force and body position influence the accuracy of UGAP in individuals with MASLD and that different thresholds should be used for men and women.
Abbreviations
- AUC:
-
Area under the receiver operating characteristic curve
- MASLD:
-
Metabolic dysfunction-associated steatotic liver disease
- MRE:
-
Magnetic resonance elastography
- MRI:
-
Magnetic resonance imaging
- N:
-
Newton
- PDFF:
-
Proton density fat fraction
- QUS:
-
Quantitative ultrasound
- RF:
-
Radiofrequency
- ROI:
-
Region of interest
- SCD:
-
Distance between liver capsule and liver surface
- UGAP:
-
Ultrasound-guided attenuation parameter
- US:
-
Ultrasound
References
Younossi ZM, Wong G, Anstee QM, Henry L (2023) The Global Burden of Liver Disease. Clin Gastroenterol Hepatol. https://doi.org/10.1016/j.cgh.2023.04.015
Simon TG, Roelstraete B, Khalili H, Hagström H, Ludvigsson JF (2021) Mortality in biopsy-confirmed nonalcoholic fatty liver disease: results from a nationwide cohort. Gut 70:1375–1382
Angulo P (2002) Nonalcoholic fatty liver disease. N Engl J Med 346:1221–1231
Adams LA, Lindor KD (2007) Nonalcoholic fatty liver disease. Ann Epidemiol 17:863–869
Akay S, Karasu Z, Noyan A et al (2007) Liver biopsy: is the pain for real or is it only the fear of it? Dig Dis Sci 52:579–581
Ratziu V, Charlotte F, Heurtier A et al (2005) Sampling variability of liver biopsy in nonalcoholic fatty liver disease. Gastroenterology 128:1898–1906
Abdi W, Millan JC, Mezey E (1979) Sampling variability on percutaneous liver biopsy. Arch Intern Med 139:667–669
Vuppalanchi R, Unalp A, Van Natta ML et al (2009) Effects of liver biopsy sample length and number of readings on sampling variability in nonalcoholic Fatty liver disease. Clin Gastroenterol Hepatol 7:481–486
Weingärtner S, Desmond KL, Obuchowski NA et al (2022) Development, validation, qualification, and dissemination of quantitative MR methods: overview and recommendations by the ISMRM quantitative MR study group. Magn Reson Med 87:1184–1206
Dulai PS, Sirlin CB, Loomba R (2016) MRI and MRE for non-invasive quantitative assessment of hepatic steatosis and fibrosis in NAFLD and NASH: clinical trials to clinical practice. J Hepatol 65:1006–1016
Qu Y, Li M, Hamilton G, Zhang YN, Song B (2019) Diagnostic accuracy of hepatic proton density fat fraction measured by magnetic resonance imaging for the evaluation of liver steatosis with histology as reference standard: a meta-analysis. Eur Radiol 29:5180–5189
Nasr P, Forsgren MF, Ignatova S et al (2017) Using a 3% proton density fat fraction as a cut-off value increases sensitivity of detection of hepatic steatosis, based on results from histopathology analysis. Gastroenterology 153:53-55.e57
EASL-EASD-EASO, (2016) EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J Hepatol 64:1388–1402
Hamaguchi M, Kojima T, Itoh Y et al (2007) The severity of ultrasonographic findings in nonalcoholic fatty liver disease reflects the metabolic syndrome and visceral fat accumulation. Am J Gastroenterol 102:2708–2715
Hernaez R, Lazo M, Bonekamp S et al (2011) Diagnostic accuracy and reliability of ultrasonography for the detection of fatty liver: a meta-analysis. Hepatology 54:1082–1090
Ryan CK, Johnson LA, Germin BI, Marcos A (2002) One hundred consecutive hepatic biopsies in the workup of living donors for right lobe liver transplantation. Liver Transpl 8:1114–1122
Perez NE, Siddiqui FA, Mutchnick MG et al (2007) Ultrasound diagnosis of fatty liver in patients with chronic liver disease: a retrospective observational study. J Clin Gastroenterol 41:624–629
Macaluso FS, Maida M, Cammà C et al (2014) Body mass index and liver stiffness affect accuracy of ultrasonography in detecting steatosis in patients with chronic hepatitis C virus genotype 1 infection. Clin Gastroenterol Hepatol 12:878-884.e871
Strauss S, Gavish E, Gottlieb P, Katsnelson L (2007) Interobserver and intraobserver variability in the sonographic assessment of fatty liver. AJR Am J Roentgenol 189:w320-323
Bigelow T (2010) Estimating the total ultrasound attenuation along the propagation path by applying multiple filters to backscattered echoes from a single spherically focused source. IEEE Trans Ultrason Ferroelectr Freq Control 57:900–907
Parker KJ, Waag RC (1983) Measurement of ultrasonic attenuation within regions selected from B-scan images. IEEE Trans Biomed Eng 30:431–437
Bae JS, Lee DH, Lee JY et al (2019) Assessment of hepatic steatosis by using attenuation imaging: a quantitative, easy-to-perform ultrasound technique. Eur Radiol. https://doi.org/10.1007/s00330-019-06272-y
Popa A, Șirli R, Popescu A et al (2021) Ultrasound-based quantification of fibrosis and steatosis with a new software considering transient elastography as reference in patients with chronic liver diseases. Ultrasound Med Biol 47:1692–1703
Cerit M, Şendur HN, Cindil E et al (2020) Quantification of liver fat content with ultrasonographic attenuation measurement function: correlation with unenhanced multidimensional computerized tomography. Clin Imaging 65:85–93
Kaliaev A, Chavez W, Soto J et al (2022) Quantitative ultrasound assessment of hepatic steatosis. J Clin Exp Hepatol 12:1091–1101
Ferraioli G, Raimondi A, Maiocchi L et al (2023) Liver fat quantification with ultrasound. J Ultrasound Med 42:2247–2255
Han A, Zhang YN, Boehringer AS et al (2019) Inter-platform reproducibility of ultrasonic attenuation and backscatter coefficients in assessing NAFLD. Eur Radiol 29:4699–4708
Nam K, Torkzaban M, Halegoua-DeMarzio D, Wessner CE, Lyshchik A (2023) Improving diagnostic accuracy of ultrasound texture features in detecting and quantifying hepatic steatosis using various beamforming sound speeds. Phys Med Biol 68. https://doi.org/10.1088/1361-6560/acb635
Rónaszéki AD, Budai BK, Csongrády B et al (2022) Tissue attenuation imaging and tissue scatter imaging for quantitative ultrasound evaluation of hepatic steatosis. Medicine (Baltimore) 101:e29708
Imajo K, Toyoda H, Yasuda S et al (2022) Utility of ultrasound-guided attenuation parameter for grading steatosis with reference to MRI-PDFF in a large cohort. Clin Gastroenterol Hepatol 20:2533-2541.e2537
Ferraioli G, Kumar V, Ozturk A, Nam K, Korte CLd, Barr RG (2022) US attenuation for liver fat quantification: an AIUM-RSNA QIBA pulse-echo quantitative ultrasound initiative. Radiology 302:495–506
Ferraioli G, Berzigotti A, Barr RG et al (2021) Quantification of liver fat content with ultrasound: a WFUMB position paper. Ultrasound Med Biol 47:2803–2820
Wirth A, Steinmetz B (1998) Gender differences in changes in subcutaneous and intra-abdominal fat during weight reduction: an ultrasound study. Obes Res 6:393–399
Fujiwara Y, Kuroda H, Abe T et al (2018) The B-mode image-guided ultrasound attenuation parameter accurately detects hepatic steatosis in chronic liver disease. Ultrasound Med Biol 44:2223–2232
Kukuk GM, Hittatiya K, Sprinkart AM et al (2015) Comparison between modified Dixon MRI techniques, MR spectroscopic relaxometry, and different histologic quantification methods in the assessment of hepatic steatosis. Eur Radiol 25:2869–2879
Alliance QIB (2022) MR Elastography of the Liver, quantitative imaging biomarkers alliance. profile stage: technically confirmed RSNA. Committee, Q. M. B, Chicago
Koo TK, Li MY (2016) A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med 15:155–163
Guglielmo FF, Venkatesh SK, Mitchell DG (2019) Liver MR elastography technique and image interpretation: pearls and pitfalls. Radiographics 39:1983–2002
Zhao Y, Jia M, Zhang C et al (2022) Reproducibility of ultrasound-guided attenuation parameter (UGAP) to the noninvasive evaluation of hepatic steatosis. Sci Rep 12:2876
Cassinotto C, Jacq T, Anselme S et al (2022) Diagnostic Performance of Attenuation to Stage Liver Steatosis with MRI proton density fat fraction as reference: a prospective comparison of three US machines. Radiology 305:353–361
Tada T, Kumada T, Toyoda H et al (2020) Liver stiffness does not affect ultrasound-guided attenuation coefficient measurement in the evaluation of hepatic steatosis. Hepatol Res 50:190–198
Guglielmo FF, Barr RG, Yokoo T et al (2023) Liver Fibrosis, fat, and iron evaluation with mri and fibrosis and fat evaluation with US: a practical guide for radiologists. Radiographics 43:e220181
Lemieux S, Prud’homme D, Bouchard C, Tremblay A, Després JP (1993) Sex differences in the relation of visceral adipose tissue accumulation to total body fatness. Am J Clin Nutr 58:463–467
Tchernof A, Bélanger C, Morisset AS et al (2006) Regional differences in adipose tissue metabolism in women: minor effect of obesity and body fat distribution. Diabetes 55:1353–1360
Byenfeldt M, Elvin A, Fransson P (2018) On patient related factors and their impact on ultrasound-based shear wave elastography of the liver. Ultrasound Med Biol 44:1606–1615
Byenfeldt M, Elvin A, Fransson P (2019) Influence of probe pressure on ultrasound-based shear wave elastography of the liver using Comb-Push 2-D technology. Ultrasound Med Biol 45:411–428
Carrascal CA, Aristizabal S, Greenleaf JF, Urban MW (2016) Phase aberration and attenuation effects on acoustic radiation force-based shear wave generation. IEEE Trans Ultrason Ferroelectr Freq Control 63:222–232
Groth M, Fischer L, Herden U et al (2023) Impact of probe-induced abdominal compression on two-dimensional shear wave elastography measurement of split liver transplants in children. RoFo: Fortschritte auf dem Gebiete der Röntgenstrahlen und der Nuklearmedizin. Rofo 195:905–912. https://doi.org/10.1055/a-2049-9369
Ferraioli G, Soares Monteiro LB (2019) Ultrasound-based techniques for the diagnosis of liver steatosis. World J Gastroenterol 25:6053–6062
Décarie PO, Lepanto L, Billiard JS et al (2011) Fatty liver deposition and sparing: a pictorial review. Insights Imaging 2:533–538
Dioguardi Burgio M, Bruno O, Agnello F et al (2016) The cheating liver: imaging of focal steatosis and fatty sparing. Expert Rev Gastroenterol Hepatol 10:671–678
Acknowledgements
We express our great gratitude to all participants in the study.
We also express our great gratitude to nurse Carola Fagerström, Linköping University Hospital in Sweden, for her professional contribution to this study.
Funding
Open access funding provided by Linköping University. This study has received funding by grants from the following:
- County of Jämtland Cancer and Nursing Foundation Sweden (grant no. 930), Lion's Cancer Research Foundation Umeå University Sweden (LP 20–2221) and Unit of Research, Education and Development Region Jämtland Härjedalen Sweden (to MB).
- Swedish Research Council and Region Östergötland Sweden (to PL).
- ALF Grants, Region Östergötland, Medical Research Council of Southeast Sweden (grant no. 752871) (to ME).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Marie Byenfeldt.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was obtained from all participants in this study.
Ethical approval
Approval from the Swedish Ethical Review Authority was obtained.
Study subjects or cohorts overlap
No study subjects or cohorts have been previously reported.
Methodology
• prospective
• diagnostic and experimental study
• performed at one institution
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Byenfeldt, M., Kihlberg, J., Nasr, P. et al. Altered probe pressure and body position increase diagnostic accuracy for men and women in detecting hepatic steatosis using quantitative ultrasound. Eur Radiol 34, 5989–5999 (2024). https://doi.org/10.1007/s00330-024-10655-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-024-10655-1