Abstract
Objectives
A watch and wait strategy with the goal of organ preservation is an emerging treatment paradigm for rectal cancer following neoadjuvant treatment. However, the selection of appropriate patients remains a challenge. Most previous efforts to measure the accuracy of MRI in assessing rectal cancer response used a small number of radiologists and did not report variability among them.
Methods
Twelve radiologists from 8 institutions assessed baseline and restaging MRI scans of 39 patients. The participating radiologists were asked to assess MRI features and to categorize the overall response as complete or incomplete. The reference standard was pathological complete response or a sustained clinical response for > 2 years.
Results
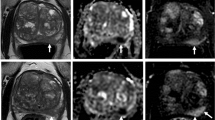
We measured the accuracy and described the interobserver variability of interpretation of rectal cancer response between radiologists at different medical centers. Overall accuracy was 64%, with a sensitivity of 65% for detecting complete response and specificity of 63% for detecting residual tumor. Interpretation of the overall response was more accurate than the interpretation of any individual feature. Variability of interpretation was dependent on the patient and imaging feature investigated. In general, variability and accuracy were inversely correlated.
Conclusions
MRI-based evaluation of response at restaging is insufficiently accurate and has substantial variability of interpretation. Although some patients’ response to neoadjuvant treatment on MRI may be easily recognizable, as seen by high accuracy and low variability, that is not the case for most patients.
Key Points
• The overall accuracy of MRI-based response assessment is low and radiologists differed in their interpretation of key imaging features.
• Some patients’ scans were interpreted with high accuracy and low variability, suggesting that these patients’ pattern of response is easier to interpret.
• The most accurate assessments were those of the overall response, which took into consideration both T2W and DWI sequences and the assessment of both the primary tumor and the lymph nodes.



Similar content being viewed by others
Abbreviations
- Cap-OX:
-
Capecitabine-oxaliplatin
- cCR:
-
Clinical complete response
- CRT:
-
Chemoradiotherapy
- FOLFOX:
-
Leucovorin-fluorouracil-oxaliplatin
- NAT:
-
Neoadjuvant treatment
- T2W:
-
T2 weighted
- TME:
-
Total mesorectal excision
- TNT:
-
Total neoadjuvant therapy
- WW:
-
Watch and wait
References
Maas M, Nelemans PJ, Valentini V et al (2010) Long-term outcome in patients with a pathological complete response after chemoradiation for rectal cancer: a pooled analysis of individual patient data. Lancet Oncol 11:835–844
Garcia-Aguilar J, Chow OS, Smith DD et al (2015) Effect of adding mFOLFOX6 after neoadjuvant chemoradiation in locally advanced rectal cancer: a multicentre, phase 2 trial. Lancet Oncol 16:957–966
Cercek A, Roxburgh CSD, Strombom P et al (2018) Adoption of total neoadjuvant therapy for locally advanced rectal cancer. JAMA Oncol 4:e180071
Chen TY, Wiltink LM, Nout RA et al (2015) Bowel function 14 years after preoperative short-course radiotherapy and total mesorectal excision for rectal cancer: report of a multicenter randomized trial. Clin Colorectal Cancer 14:106–114
Maas M, Beets-Tan RG, Lambregts DM et al (2011) Wait-and-see policy for clinical complete responders after chemoradiation for rectal cancer. J Clin Oncol 29:4633–4640
Araujo RO, Valadao M, Borges D et al (2015) Nonoperative management of rectal cancer after chemoradiation opposed to resection after complete clinical response. A comparative study. Eur J Surg Oncol 41:1456–1463
Martens MH, Maas M, Heijnen LA et al (2016) Long-term outcome of an organ preservation program after neoadjuvant treatment for rectal cancer. J Natl Cancer Inst 108
Appelt AL, Ploen J, Harling H et al (2015) High-dose chemoradiotherapy and watchful waiting for distal rectal cancer: a prospective observational study. Lancet Oncol 16:919–927
Smith JD, Ruby JA, Goodman KA et al (2012) Nonoperative management of rectal cancer with complete clinical response after neoadjuvant therapy. Ann Surg 256:965–972
Renehan AG, Malcomson L, Emsley R et al (2016) Watch-and-wait approach versus surgical resection after chemoradiotherapy for patients with rectal cancer (the OnCoRe project): a propensity-score matched cohort analysis. Lancet Oncol 17:174–183
Smith JJ, Strombom P, Chow OS et al (2019) Assessment of a watch-and-wait strategy for rectal cancer in patients with a complete response after neoadjuvant therapy. JAMA Oncol 5:e185896
Maas M, Lambregts DM, Nelemans PJ et al (2015) Assessment of clinical complete response after chemoradiation for rectal cancer with digital rectal examination, endoscopy, and MRI: selection for organ-saving treatment. Ann Surg Oncol 22:3873–3880
Lambregts DMJ, Delli Pizzi A, Lahaye MJ et al (2018) A pattern-based approach combining tumor morphology on MRI with distinct signal patterns on diffusion-weighted imaging to assess response of rectal tumors after chemoradiotherapy. Dis Colon Rectum 61:328–337
Sclafani F, Brown G, Cunningham D et al (2017) Comparison between MRI and pathology in the assessment of tumour regression grade in rectal cancer. Br J Cancer 117:1478–1485
Lambregts DM, Vandecaveye V, Barbaro B et al (2011) Diffusion-weighted MRI for selection of complete responders after chemoradiation for locally advanced rectal cancer: a multicenter study. Ann Surg Oncol 18:2224–2231
Aker M, Boone D, Chandramohan A, Sizer B, Motson R, Arulampalam T (2018) Diagnostic accuracy of MRI in assessing tumor regression and identifying complete response in patients with locally advanced rectal cancer after neoadjuvant treatment. Abdom Radiol (NY) 43:3213–3219
Smith JJ, Chow OS, Gollub MJ et al (2015) Organ Preservation in Rectal Adenocarcinoma: a phase II randomized controlled trial evaluating 3-year disease-free survival in patients with locally advanced rectal cancer treated with chemoradiation plus induction or consolidation chemotherapy, and total mesorectal excision or nonoperative management. BMC Cancer 15:767
Garcia-Aguilar J, Patil S, Gollub MJ et al (2022) Organ preservation in patients with rectal adenocarcinoma treated with total neoadjuvant therapy. J Clin Oncol. https://doi.org/10.1200/JCO.22.00032:JCO2200032
Beets-Tan RGH, Lambregts DMJ, Maas M et al (2018) Magnetic resonance imaging for clinical management of rectal cancer: updated recommendations from the 2016 European Society of Gastrointestinal and Abdominal Radiology (ESGAR) consensus meeting. Eur Radiol 28:1465–1475
Santiago I, Barata M, Figueiredo N et al (2020) The split scar sign as an indicator of sustained complete response after neoadjuvant therapy in rectal cancer. Eur Radiol 30:224–238
de Jong EA, ten Berge JC, Dwarkasing RS, Rijkers AP, van Eijck CH (2016) The accuracy of MRI, endorectal ultrasonography, and computed tomography in predicting the response of locally advanced rectal cancer after preoperative therapy: a metaanalysis. Surgery 159:688–699
van den Broek JJ, van der Wolf FS, Lahaye MJ et al (2017) Accuracy of MRI in restaging locally advanced rectal cancer after preoperative chemoradiation. Dis Colon Rectum 60:274–283
Haak HE, Maas M, Lahaye MJ et al (2020) Selection of patients for organ preservation after chemoradiotherapy: MRI identifies poor responders who can go straight to surgery. Ann Surg Oncol 27:2732–2739
Siddiqui MR, Gormly KL, Bhoday J et al (2016) Interobserver agreement of radiologists assessing the response of rectal cancers to preoperative chemoradiation using the MRI tumour regression grading (mrTRG). Clin Radiol 71:854–862
Yuval JB, Thompson HM, Garcia-Aguilar J (2020) Organ preservation in rectal cancer. J Gastrointest Surg 24:1880–1888
Gollub MJ, Das JP, Bates DDB et al (2021) Rectal cancer with complete endoscopic response after neoadjuvant therapy: what is the meaning of a positive MRI? Eur Radiol. https://doi.org/10.1007/s00330-020-07657-0
Garcia-Aguilar J, Patil S, Kim JK et al (2020) Preliminary results of the organ preservation of rectal adenocarcinoma (OPRA) trial. J Clin Oncol 38:4008–4008
Thompson H, Kim JK, Yuval JB et al (2021) Survival and organ preservation according to clinical response after total neoadjuvant therapy in locally advanced rectal cancer patients: a secondary analysis from the Organ Preservation in Rectal Adenocarcinoma (OPRA) trial. J Clin Oncol 39:3509–3509
Kahneman D, Rosenfield A, Gandhi L, Blaser T (2016) Noise. Harvard Bus Rev:38–46
Acknowledgements
The authors thank Arthur Gelmis, BS, for editing the manuscript and Floris S. Verheij for help with production of the manuscript. JBY acknowledges Yael Renert-Yuval for insightful conversations.
Funding
Research at Memorial Sloan Kettering is funded in part by grant P30 CA008748 from the National Cancer Institute. JBY’s research fellowship at Memorial Sloan Kettering was funded in part by grant T32 CA009501 from the National Cancer Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Marc J. Gollub.
Conflict of interest
Patil is a consultant for ByHeart. Julio Garcia-Aguilar has received honoraria from Medtronic, Intuitive Surgical, and Johnson & Johnson and owns stock in Intuitive Surgical. Marc J. Gollub holds stocks in Pfizer. All other authors have no disclosures to declare. Other authors have no conflicts of interest to disclose.
Statistics and biometry
SP is an experienced biostatistician and is one of the authors.
Informed consent
Written informed consent was waived by the institutional review board.
Ethical approval
Institutional review board approval was obtained.
Methodology
• retrospective
• observational
• multicenter study
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yuval, J.B., Patil, S., Gangai, N. et al. MRI assessment of rectal cancer response to neoadjuvant therapy: a multireader study. Eur Radiol 33, 5761–5768 (2023). https://doi.org/10.1007/s00330-023-09480-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-023-09480-9