Abstract
Objective
To determine if artificial intelligence–based modification of the Thyroid Imaging Reporting Data System (TI-RADS) would be better than the current American College of Radiology (ACR) TI-RADS for risk stratification of thyroid nodules.
Methods
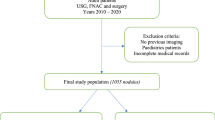
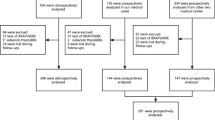
A total of 2061 thyroid nodules (in 1859 patients) sampled with fine-needle aspiration or operation were retrospectively analyzed between January 2017 and July 2020. Two radiologists blinded to the pathologic diagnosis evaluated nodule features in five ultrasound categories and assigned TI-RADS scores by both ACR TI-RADS and AI TI-RADS. Inter-rater agreement was assessed by asking another two radiologists to score a set of 100 nodules independently. The reference standard was postoperative pathological or cytopathological diagnosis according to the Bethesda system. Inter-rater agreement was determined using intraclass correlation coefficient (ICC).
Results
AI TI-RADS assigned lower TI-RADS risk levels than ACR TI-RADS (p < 0.001) and had larger area under receiver operating characteristic curve (0.762 vs. 0.679, p < 0.001). The sensitivities of ACR TI-RADS and AI TI-RADS were similar (86.7% vs. 82.2%, p = 0.052), but specificity was higher with AI TI-RADS (70.2% vs. 49.2%, p < 0.001). AI TI-RADS downgraded 743 (48.63%) benign nodules, indicating that 328 (42.3% of 776 biopsied nodules) unnecessary fine-needle aspirations (FNA) could have been avoided. Inter-rater agreement was better with AI TI-RADS than with ACR TI-RADS (ICC, 0.808 vs. 0.861, p < 0.001).
Conclusion
AI TI-RADS can achieve meaningful reduction in the number of benign thyroid nodules recommended for biopsy and significantly improve specificity despite a slight decrease in sensitivity.
Key Points
• AI TI-RADS assigned lower TI-RADS risk levels than ACR TI-RADS, showing similar sensitivity but higher specificity.
• Half of the benign nodules can be downgraded of which 42.3% of biopsy nodules avoided unnecessary fine-needle aspiration (FNA).
• AI TI-RADS had a better overall inter-rater agreement.




Similar content being viewed by others
Abbreviations
- ACR:
-
American College of Radiology
- AI:
-
Artificial intelligence
- AUC:
-
Area under receiver operating characteristic curve
- CI:
-
Confidence interval
- FNA:
-
Fine-needle aspiration
- ICC:
-
Interclass correlation coefficient
- ROC:
-
Receiver operating characteristic curve
- SD:
-
Standard deviation
- TI-RADS:
-
Thyroid Imaging Reporting and Data System
- US:
-
Ultrasound
References
Wiltshire J, Drake T, Uttley L, Balasubramanian S (2016) Systematic review of trends in the incidence rates of thyroid cancer. Thyroid 26:1541–1552
Vaccarella S, Franceschi S, Bray F, Wild C, Plummer M, Dal Maso L (2016) Worldwide thyroid-cancer epidemic? The increasing impact of overdiagnosis. N Engl J Med 375:614–617
Li Y, Teng D, Ba J et al (2020) Efficacy and safety of long-term universal salt iodization on thyroid disorders: epidemiological evidence from 31 provinces of mainland China. Thyroid 30:568–579
Singh Ospina N, Brito J, Maraka S et al (2016) Diagnostic accuracy of ultrasound-guided fine needle aspiration biopsy for thyroid malignancy: systematic review and meta-analysis. Endocrine 53:651–661
Hoang JK, Middleton WD, Farjat AE et al (2018) Interobserver variability of sonographic features used in the American College of Radiology Thyroid Imaging Reporting and Data System. AJR Am J Roentgenol 211:162–167
Smith-Bindman R, Lebda P, Feldstein V et al (2013) Risk of thyroid cancer based on thyroid ultrasound imaging characteristics: results of a population-based study. JAMA Intern Med 173:1788–1796
Ahn H, Kim H, Welch H (2014) Korea’s thyroid-cancer “epidemic”--screening and overdiagnosis. N Engl J Med 371:1765–1767
Srinivas M, Amogh V, Gautam M et al (2016) A prospective study to evaluate the reliability of Thyroid Imaging Reporting and Data System in differentiation between benign and malignant thyroid lesions. J Clin Imaging Sci 6:5
Horvath E, Majlis S, Rossi R et al (2009) An ultrasonogram reporting system for thyroid nodules stratifying cancer risk for clinical management. J Clin Endocrinol Metab 94:1748–1751
Tessler F, Middleton W, Grant E et al (2017) ACR Thyroid Imaging, Reporting and Data System (TI-RADS): white paper of the ACR TI-RADS Committee. J Am Coll Radiol 14:587–595
Wildman-Tobriner B, Buda M, Hoang J et al (2019) Using artificial intelligence to revise ACR TI-RADS risk stratification of thyroid nodules: diagnostic accuracy and utility. Radiology 292:112–119
Hoang J, Middleton W, Farjat A et al (2018) Reduction in thyroid nodule biopsies and improved accuracy with American College of Radiology Thyroid Imaging Reporting and Data System. Radiology 287:185–193
Pandya A, Caoili E, Jawad-Makki F et al (2020) Retrospective cohort study of 1947 thyroid nodules: a comparison of the 2017 American College of Radiology TI-RADS and the 2015 American Thyroid Association Classifications. AJR Am J Roentgenol 214:900–906
Tan L, Tan Y, Tan S (2020) Diagnostic accuracy and ability to reduce unnecessary FNAC: a comparison between four Thyroid Imaging Reporting Data System (TI-RADS) versions. Clin Imaging 65:133–137
Cibas E, Ali S (2017) The 2017 Bethesda system for reporting thyroid cytopathology. Thyroid 27:1341–1346
Middleton W, Teefey S, Reading C et al (2018) Comparison of performance characteristics of American College of Radiology TI-RADS, Korean Society of Thyroid Radiology TIRADS, and American Thyroid Association Guidelines. AJR Am J Roentgenol 210:1148–1154
Lauria Pantano A, Maddaloni E, Briganti S et al (2018) Differences between ATA, AACE/ACE/AME and ACR TI-RADS ultrasound classifications performance in identifying cytological high-risk thyroid nodules. Eur J Endocrinol 178:595–603
Yoon J, Lee H, Kim E, Moon H, Kwak J (2016) Malignancy risk stratification of thyroid nodules: comparison between the Thyroid Imaging Reporting and Data System and the 2014 American Thyroid Association Management Guidelines. Radiology 278:917–924
Kim D, Chung S, Choi S, Kim K (2020) Accuracy of thyroid imaging reporting and data system category 4 or 5 for diagnosing malignancy: a systematic review and meta-analysis. Eur Radiol 30:5611–5624
Ha SM, Baek JH, Na DG et al (2019) Diagnostic performance of practice guidelines for thyroid nodules: thyroid nodule size versus biopsy rates. Radiology 291:92–99
Grani G, Lamartina L, Ascoli V et al (2019) Reducing the number of unnecessary thyroid biopsies while improving diagnostic accuracy: toward the “right” TIRADS. J Clin Endocrinol Metab 104:95–102
La Vecchia C, Malvezzi M, Bosetti C et al (2015) Thyroid cancer mortality and incidence: a global overview. Int J Cancer 136:2187–2195
Lee M, Kim E, Kwak J, Kim M (2009) Partially cystic thyroid nodules on ultrasound: probability of malignancy and sonographic differentiation. Thyroid 19:341–346
Frates M, Benson C, Doubilet P et al (2006) Prevalence and distribution of carcinoma in patients with solitary and multiple thyroid nodules on sonography. J Clin Endocrinol Metab 91:3411–3417
Bellantone R, Lombardi C, Raffaelli M et al (2004) Management of cystic or predominantly cystic thyroid nodules: the role of ultrasound-guided fine-needle aspiration biopsy. Thyroid 14:43–47
Shi Y, Jin Y, Zheng L (2020) Partially cystic thyroid nodules on ultrasound: the associated factors for malignancy. Clin Hemorheol Microcirc 74:373–381
Kim D, Park J, In H, Choo H, Ryu J, Jung S (2012) Ultrasound-based diagnostic classification for solid and partially cystic thyroid nodules. AJNR Am J Neuroradiol 33:1144–1149
Kim D, Lee E, In H, Kim S (2010) Sonographic differentiation of partially cystic thyroid nodules: a prospective study. AJNR Am J Neuroradiol 31:1961–1966
Middleton W, Teefey S, Reading C et al (2017) Multiinstitutional analysis of thyroid nodule risk stratification using the American College of Radiology Thyroid Imaging Reporting and Data System. AJR Am J Roentgenol 208:1331–1341
Lee Y, Kim D, In H et al (2011) Differentiation between benign and malignant solid thyroid nodules using an US classification system. Korean J Radiology 12:559–567
Choi Y, Kim S, Choi S (2008) Diagnostic accuracy of ultrasound features in thyroid microcarcinomas. Endocr J 55:931–938
Kim E, Park C, Chung W et al (2002) New sonographic criteria for recommending fine-needle aspiration biopsy of nonpalpable solid nodules of the thyroid. AJR Am J Roentgenol 178:687–691
Moon W, Jung S, Lee J et al (2008) Benign and malignant thyroid nodules: US differentiation--multicenter retrospective study. Radiology 247:762–770
Moon W, Baek J, Jung S et al (2011) Ultrasonography and the ultrasound-based management of thyroid nodules: consensus statement and recommendations. Korean J Radiology 12:1–14
Lee J, Lee S, Cha S, Cho B, Kang M, Lee O (2013) Fine-needle aspiration of thyroid nodules with macrocalcification. Thyroid 23:1106–1112
Xu T, Gu J, Ye X et al (2017) Thyroid nodule sizes influence the diagnostic performance of TIRADS and ultrasound patterns of 2015 ATA guidelines: a multicenter retrospective study. Sci Rep 7:43183
Trimboli P, Treglia G, Guidobaldi L et al (2014) Clinical characteristics as predictors of malignancy in patients with indeterminate thyroid cytology: a meta-analysis. Endocrine 46:52–59
Ahn H, Na D, Baek J, Sung J, Kim J (2019) False negative rate of fine-needle aspiration in thyroid nodules: impact of nodule size and ultrasound pattern. Head Neck 41:967–973
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Jianhua Zhou.
Conflict of Interest
The authors of this manuscript declare no relationships with any companies, whose productsor services may be related to the subject matter of the article.
Statistics and Biometry
No complex statistical methods were necessary for this paper.
Informed Consent
Written informed consent was waived by the Institutional Review Board.
Ethical Approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• diagnostic study
• multicenter study
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 34 kb)
Rights and permissions
About this article
Cite this article
Liu, Y., Li, X., Yan, C. et al. Comparison of diagnostic accuracy and utility of artificial intelligence–optimized ACR TI-RADS and original ACR TI-RADS: a multi-center validation study based on 2061 thyroid nodules. Eur Radiol 32, 7733–7742 (2022). https://doi.org/10.1007/s00330-022-08827-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-022-08827-y