Abstract
Objective
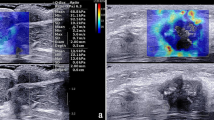
To determine the ability of conventional ultrasound (US) combined with shear wave elastography (SWE) to reveal axillary status after neoadjuvant chemotherapy (NAC) in breast cancer patients.
Methods
From September 2016 to December 2021, 201 patients with node-positive breast cancer who underwent NAC were enrolled in this prospective study. Conventional US features of axillary lymph nodes and SWE characteristics of breast lesions after NAC were analyzed. The diagnostic performances of US, SWE, and their combination were assessed using multivariate logistic regression and receiver operator characteristic curve (ROC) analyses.
Results
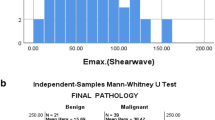
The area under the ROC curve (AUC) for the ability of conventional US features to determine axillary status after NAC was 0.82, with a sensitivity of 85.23%, a specificity of 67.39%, and an accuracy of 76.11%. Shear wave velocity (SWV) displayed moderate performance for predicting axilla status after NAC with SWVmean demonstrating an AUC of 0.85. Cortical thickness and shape of axillary nodes and SWVmean of breast tumors were independently associated with axillary nodal metastasis after NAC. Compared to conventional US, the combination of conventional US of axillary lymph nodes with SWE of breast lesions achieved a significantly higher AUC (0.90 vs 0.82, p < 0.01, Delong’s test) with a sensitivity of 87.50%, improved specificity of 82.61% and accuracy of 85.00%.
Conclusions
Breast SWE was independently associated with residual metastasis of axillary node after NAC in patients with initially diagnosed positive axilla. Combining SWE with conventional US showed good diagnostic performance for axillary node disease after NAC.
Key Points
• Breast SWE can serve as a supplement to axilla US for the evaluation of the axilla after NAC.
• The combination of axilla US with breast SWE may be a promising method to facilitate less-invasive treatment in patients receiving NAC.






Similar content being viewed by others
Abbreviations
- ALND:
-
Axillary lymph node dissection
- AUC:
-
Area under the ROC
- CI:
-
Confidence interval
- CT:
-
Computerized tomography
- ER:
-
Estrogen receptor
- FNR:
-
False-negative rates
- HE:
-
Hematoxylin and eosin
- HER2:
-
Human epidermal growth factor receptor 2
- IHC:
-
Immunohistochemistry
- LN:
-
Lymph node
- NAC:
-
Neoadjuvant chemotherapy
- MRI:
-
Magnetic resonance imaging
- OR:
-
Odds ratio
- pCR:
-
Pathological complete response
- PET:
-
Positron emission tomography
- PR:
-
Progesterone receptor
- ROC:
-
Receiver operating characteristic
- SE:
-
Strain elastography
- SLNB:
-
Sentinel lymph node biopsy
- SWE:
-
Shear wave elastography
- SWV:
-
Shear wave velocity
- UE:
-
Ultrasound elastography
- US:
-
Ultrasound
References
Teshome M, Hunt KK (2014) Neoadjuvant therapy in the treatment of breast cancer. Surg Oncol Clin 23:505–523. https://doi.org/10.1016/j.soc.2014.03.006
Gradishar WJ, Anderson BO, Abraham J et al (2020) Breast Cancer, Version 3.2020, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 18:452–478. https://doi.org/10.6004/jnccn.2020.0016
Galimberti V, Cole BF, Viale G et al (2018) Axillary dissection versus no axillary dissection in patients with breast cancer and sentinel-node micrometastases (IBCSG 23-01): 10-year follow-up of a randomised, controlled phase 3 trial. Lancet Oncol 19:1385–1393. https://doi.org/10.1016/S1470-2045(18)30380-2
Boughey JC, Suman VJ, Mittendorf EA et al (2013) Sentinel lymph node surgery after neoadjuvant chemotherapy in patients with node-positive breast cancer: the ACOSOG Z1071 (Alliance) clinical trial. JAMA 310:1455–1461. https://doi.org/10.1001/jama.2013.278932
Kuehn T, Bauerfeind I, Fehm T et al (2013) Sentinel-lymph-node biopsy in patients with breast cancer before and after neoadjuvant chemotherapy (SENTINA): a prospective, multicentre cohort study. Lancet Oncol 14:609–618. https://doi.org/10.1016/S1470-2045(13)70166-9
Magnoni F, Galimberti V, Corso G et al (2020) Axillary surgery in breast cancer: an updated historical perspective. Semin Oncol 47:341–352. https://doi.org/10.1053/j.seminoncol.2020.09.001
Boughey JC, Ballman KV, Hunt KK et al (2015) Axillary ultrasound after neoadjuvant chemotherapy and its impact on sentinel lymph node surgery: results from the American College of Surgeons Oncology Group Z1071 Trial (Alliance). J Clin Oncol 33:3386–3393. https://doi.org/10.1200/JCO.2014.57.8401
Peppe A, Wilson R, Pope R et al (2017) The use of ultrasound in the clinical re-staging of the axilla after neoadjuvant chemotherapy (NACT). Breast 35:104–108. https://doi.org/10.1016/j.breast.2017.05.015
Banys-Paluchowski M, Gruber IV, Hartkopf A et al (2020) Axillary ultrasound for prediction of response to neoadjuvant therapy in the context of surgical strategies to axillary dissection in primary breast cancer: a systematic review of the current literature. Arch Gynecol Obstet 301:341–353. https://doi.org/10.1007/s00404-019-05428-x
Expert Panel on Breast Imaging, Slanetz PJ, Moy L et al (2017) ACR Appropriateness Criteria® monitoring response to neoadjuvant systemic therapy for breast cancer. J Am Coll Radiol 14:462–475. https://doi.org/10.1016/j.jacr.2017.08.037
Chang JM, Leung JWT, Moy L, Ha SM, Moon WK (2020) Axillary nodal evaluation in breast cancer: state of the art. Radiology 295:500–515. https://doi.org/10.1148/radiol.2020192534
Almerey T, Villacreses D, Li Z et al (2019) Value of axillary ultrasound after negative axillary MRI for evaluating nodal status in high-risk breast Cancer. J Am Coll Surg 228:792–797. https://doi.org/10.1016/j.jamcollsurg.2019.01.022
Barr RG, Nakashima K, Amy D et al (2015) WFUMB guidelines and recommendations for clinical use of ultrasound elastography: Part 2: breast. Ultrasound Med Biol 41:1148–1160. https://doi.org/10.1016/j.ultrasmedbio.2015.03.008
Cosgrove DO, Berg WA, Doré CJ et al (2012) Shear wave elastography for breast masses is highly reproducible. Eur Radiol 22:1023–1032. https://doi.org/10.1007/s00330-011-2340-y
Ma Y, Zhang S, Li J et al (2017) Comparison of strain and shear-wave ultrasounic elastography in predicting the pathological response to neoadjuvant chemotherapy in breast cancers. Eur Radiol 27:2282–2291. https://doi.org/10.1007/s00330-016-4619-5
Evans A, Whelehan P, Thompson A et al (2018) Identification of pathological complete response after neoadjuvant chemotherapy for breast cancer: comparison of greyscale ultrasound, shear wave elastography, and MRI. Clin Radiol 73:910.e1–910.e6. https://doi.org/10.1016/j.crad.2018.05.030
Jing H, Cheng W, Li ZY et al (2016) Early evaluation of relative changes in tumor stiffness by shear wave elastography predicts the response to neoadjuvant chemotherapy in patients with breast cancer. J Ultrasound Med 35:1619–1627. https://doi.org/10.7863/ultra.15.08052
Fernandes J, Sannachi L, Tran WT et al (2019) Monitoring breast cancer response to neoadjuvant chemotherapy using ultrasound strain elastography. Transl Oncol 12:1177–1184. https://doi.org/10.1016/j.tranon.2019.05.004
Ma Y, Zhang S, Zang L et al (2016) Combination of shear wave elastography and Ki-67 index as a novel predictive modality for the pathological response to neoadjuvant chemotherapy in patients with invasive breast cancer. Eur J Cancer 69:86–101. https://doi.org/10.1016/j.ejca.2016.09.031
Evans A, Whelehan P, Thompson A et al (2018) Prediction of pathological complete response to neoadjuvant chemotherapy for primary breast cancer comparing interim ultrasound, shear wave elastography and MRI. Ultraschall Med 39:422–431. https://doi.org/10.1055/s-0043-111589
Lee SH, Chang JM, Han W et al (2015) Shear-wave elastography for the detection of residual breast cancer after neoadjuvant chemotherapy. Ann Surg Oncol 22:376–384. https://doi.org/10.1245/s10434-015-4828-1
Evans A, Armstrong S, Whelehan P et al (2013) Can shear-wave elastography predict response to neoadjuvant chemotherapy in women with invasive breast cancer? Br J Cancer 109:2798–2802. https://doi.org/10.1038/bjc.2013.660
Reinert T, de Souza ABA, Sartori GP, Obst FM, Barrios CH (2021) Highlights of the 17th St Gallen International Breast Cancer Conference 2021: customising local and systemic therapies. Ecancermedicalscience 15:1236. https://doi.org/10.3332/ecancer.2021.1236
Ai X, Liao X, Wang M et al (2020) Prognostic value of lymph node ratio in breast cancer patients with adequate pathologic evidence after neoadjuvant chemotherapy. Med Sci Monit 26:e922420. https://doi.org/10.12659/MSM.922420
American Institute of Ultrasound in Medicine, American Society of Breast Surgeons (2009) AIUM practice guideline for the performance of a breast ultrasound examination. J Ultrasound Med 28:105–109. https://doi.org/10.7863/jum.2009.28.1.105
Eun NL, Son EJ, Gweon HM, Kim JA, Youk JH (2020) Prediction of axillary response by monitoring with ultrasound and MRI during and after neoadjuvant chemotherapy in breast cancer patients. Eur Radiol 30:1460–1469. https://doi.org/10.1007/s00330-019-06539-4
Hammond ME, Hayes DF, Dowsett M et al (2010) American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. Arch Pathol Lab Med 134:907–922. https://doi.org/10.1200/JCO.2009.25.6529
Li DD, Xu HX, Guo LH et al (2016) Combination of two-dimensional shear wave elastography with ultrasound breast imaging reporting and data system in the diagnosis of breast lesions: a new method to increase the diagnostic performance. Eur Radiol 26:3290–3300. https://doi.org/10.1007/s00330-015-4163-8
Goldhirsch A, Winer EP, Coates AS et al (2013) Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol 24:2206–2223. https://doi.org/10.1093/annonc/mdt303
Waks AG, Winer EP et al (2019) Breast cancer treatment: a review. JAMA 321:288–300. https://doi.org/10.1001/jama.2018.19323
Le-Petross HT, McCall LM, Hunt KK et al (2018) Axillary ultrasound identifies residual nodal disease after chemotherapy: results from the American College of Surgeons Oncology Group Z1071 Trial (Alliance). AJR Am J Roentgenol 210:669–676. https://doi.org/10.2214/AJR.17.18295
Shin K, Weaver O, Wei W et al (2020) Sonographic features of benign and malignant axillary nodes post-neoadjuvant chemotherapy. Breast J 26:182–187. https://doi.org/10.1111/tbj.13488
Morency D, Dumitra S, Parvez E et al (2019) Axillary lymph node ultrasound following neoadjuvant chemotherapy in biopsy-proven node-positive breast cancer: results from the SN FNAC Study. Ann Surg Oncol 26:4337–4345. https://doi.org/10.1245/s10434-019-07809-7
Skarping I, Förnvik D, Zackrisson S, Borgquist S, Rydén L (2021) Predicting pathological axillary lymph node status with ultrasound following neoadjuvant therapy for breast cancer. Breast Cancer Res Treat 189:131–144. https://doi.org/10.1007/s10549-021-06283-8
Sun CY, Lei KR, Liu BJ et al (2017) Virtual touch tissue imaging and quantification (VTIQ) in the evaluation of thyroid nodules: the associated factors leading to misdiagnosis. Sci Rep 7:41958. https://doi.org/10.1038/srep41958
Hajian-Tilaki K (2013) Receiver Operating Characteristic (ROC) Curve analysis for medical diagnostic test evaluation. Caspian J Intern Med 4:627–635
de Meric de Bellefon M, Lemanski C, Ducteil A et al (2018) Management of the axilla in the era of breast cancer heterogeneity. Front Oncol 8:84. https://doi.org/10.3389/fonc.2018.00084
Boileau JF, Poirier B, Basik M et al (2015) Sentinel node biopsy after neoadjuvant chemotherapy in biopsy-proven node-positive breast cancer: the SN FNAC study. J Clin Oncol 33:258–264. https://doi.org/10.1200/JCO.2014.55.7827
Mamtani A, Barrio AV, King TA et al (2016) How often does neoadjuvant chemotherapy avoid axillary dissection in patients with histologically confirmed nodal metastases? Results of a prospective study. Ann Surg Oncol 23:3467–3474. https://doi.org/10.1245/s10434-016-5246-8
Croshaw R, Shapiro-Wright H, Svensson E et al (2011) Accuracy of clinical examination, digital mammogram, ultrasound, and MRI in determining postneoadjuvant pathologic tumor response in operable breast cancer patients. Ann Surg Oncol 18:3160–3163. https://doi.org/10.1245/s10434-011-1919-5
Vijayaraghavan GR, Vedantham S, Kataoka M et al (2017) The relevance of ultrasound imaging of suspicious axillary lymph nodes and fine-needle aspiration biopsy in the post-ACOSOG Z11 era in early breast cancer. Acad Radiol 24:308–315. https://doi.org/10.1016/j.acra.2016.10.005
Kim R, Chang JM, Lee HB et al (2019) Predicting axillary response to neoadjuvant chemotherapy: breast MRI and US in patients with node-positive breast cancer. Radiology 293:49–57. https://doi.org/10.1148/radiol.2019190014
Zhao Q, Sun JW, Zhou H et al (2018) Pre-operative conventional ultrasound and sonoelastography evaluation for predicting axillary lymph node metastasis in patients with malignant breast lesions. Ultrasound Med Biol 44:2587–2595. https://doi.org/10.1016/j.ultrasmedbio.2018.07.017
Zhao QL, Xia XN, Zhang Y et al (2018) Elastosonography and two-dimensional ultrasonography in diagnosis of axillary lymph node metastasis in breast cancer. Clin Radiol 73:312–318. https://doi.org/10.1016/j.crad.2017.09.013
Xiao-Long L, Hui-Xiong X, Dan-Dan L et al (2017) A risk model based on ultrasound, ultrasound elastography, and histologic parameters for predicting axillary lymph node metastasis in breast invasive ductal carcinoma. Sci Rep 7:3029. https://doi.org/10.1038/s41598-017-03582-3
Wang RY, Zhang YW, Gao ZM, Wang XM (2020) Role of sonoelastography in assessment of axillary lymph nodes in breast cancer: a systematic review and meta-analysis. Clin Radiol 75:320.e1–320.e7. https://doi.org/10.1016/j.crad.2019.11.016
Maeshima Y, Sakai T, Ogiya A et al (2021) Assessment of axillary node status by ultrasound after neoadjuvant chemotherapy in patients with clinically node-positive breast cancer according to breast cancer subtype. Sci Rep 11:10858. https://doi.org/10.1038/s41598-021-89738-8
Laws A, Dillon K, Kelly BN et al (2020) Node-positive patients treated with neoadjuvant chemotherapy can be spared axillary lymph node dissection with wireless non-radioactive localizers. Ann Surg Oncol 27:4819–4827. https://doi.org/10.1245/s10434-020-08902-y
Acknowledgements
The authors would like to thank all the involved study investigators for dedicating their time and skills to the completion of this study. The authors would also like to thank Medjaden Bioscience Limited for their assistance in the preparation of the manuscript.
Funding
This study has received funding from the Science and Technology Project Funds of Guangzhou (No. 1563000308).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Xiao-Qing Pei.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• prospective
• diagnostic or prognostic study
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Huang, JX., Lin, SY., Ou, Y. et al. Combining conventional ultrasound and sonoelastography to predict axillary status after neoadjuvant chemotherapy for breast cancer. Eur Radiol 32, 5986–5996 (2022). https://doi.org/10.1007/s00330-022-08751-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-022-08751-1