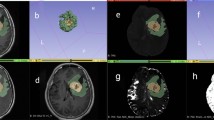
Abstract
Objectives
To construct a radiomics nomogram based on multiparametric MRI data for predicting isocitrate dehydrogenase 1 mutation (IDH +) and loss of nuclear alpha thalassemia/mental retardation syndrome X‐linked expression (ATRX −) in patients with lower‐grade gliomas (LrGG; World Health Organization [WHO] 2016 grades II and III).
Methods
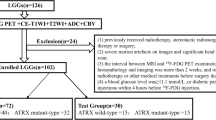
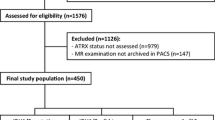
A total of 111 LrGG patients (76 mutated IDH and 35 wild-type IDH) were enrolled, divided into a training set (n = 78) and a validation set (n = 33) for predicting IDH mutation. IDH + LrGG patients were further stratified into the ATRX − (n = 38) and ATRX + (n = 38) subtypes. A total of 250 radiomics features were extracted from the region of interest of each tumor, including that from T2 fluid-attenuated inversion recovery (T2 FLAIR), contrast-enhanced T1 WI, ASL-derived cerebral blood flow (CBF), DWI-derived ADC, and exponential ADC (eADC). A radiomics signature was selected using the Elastic Net regression model, and a radiomics nomogram was finally constructed using the age, gender information, and above features.
Results
The radiomics nomogram identified LrGG patients for IDH mutation (C-index: training sets = 0.881, validation sets = 0.900) and ATRX loss (C-index: training sets = 0.863, validation sets = 0.840) with good calibration. Decision curve analysis further confirmed the clinical usefulness of the two nomograms for predicting IDH and ATRX status.
Conclusions
The nomogram incorporating age, gender, and the radiomics signature provided a clinically useful approach in noninvasively predicting IDH and ATRX mutation status for LrGG patients. The proposed method could facilitate MRI-based clinical decision-making for the LrGG patients.
Key Points
• Non-invasive determination of IDH and ATRX gene status of LrGG patients can be obtained with a radiomics nomogram.
• The proposed nomogram is constructed by radiomics signature selected from 250 radiomics features, combined with age and gender.
• The proposed radiomics nomogram exhibited good calibration and discrimination for IDH and ATRX gene mutation stratification of LrGG patients in both training and validation sets.






Similar content being viewed by others
Abbreviations
- ATRX:
-
Alpha thalassemia/mental retardation syndrome X-linked
- CBF:
-
Cerebral blood flow
- DCA:
-
Decision curve analysis
- FLAIR:
-
Fluid-attenuated inversion recovery
- GLCM:
-
Gray-level co-occurrence matrix
- GLRLM:
-
Gray-level run-length matrix
- GLSZM:
-
Gray-level size zone matrix
- IDH:
-
Isocitrate dehydrogenase
- LrGG:
-
Lower-grade gliomas
- NGDTM:
-
Neighborhood gray-tone difference matrix
- T1 C:
-
Contrast-enhanced T1-weighted imaging
- T E :
-
Echo time
- T R :
-
Repetition time
References
Gittleman H, Sloan AE, Barnholtz-Sloan JS (2020) An independently validated survival nomogram for lower-grade glioma. Neuro Oncol 22:665–674
Suzuki H, Aoki K, Chiba K et al (2015) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 47:458–468
Cancer Genome Atlas Research Network, Brat DJ, Verhaak RG et al (2015) Comprehensive, Integrative Genomic Analysis of Diffuse Lower-Grade Gliomas. N Engl J Med 372:2481–2498
Wang Q, Zhang L, Cui Y et al (2020) Increased RLIP76 expression in IDH1 wild-type glioblastoma multiforme is associated with worse prognosis. Oncol Rep 43:188–200
Cancer Genome Atlas Research Network (2015) Comprehensive, integrative genomic analysis of diffuse lower-grade gliomas. N Engl J Med 372:2481–2498
Beiko J, Suki D, Hess KR et al (2014) IDH1 mutant malignant astrocytomas are more amenable to surgical resection and have a survival benefit associated with maximal surgical resection. Neuro Oncol 16:81–91
Kizilbash SH, Giannini C, Voss JS et al (2014) The impact of concurrent temozolomide with adjuvant radiation and IDH mutation status among patients with anaplastic astrocytoma. J Neurooncol 120:85–93
Grasso CS, Tang Y, Truffaux N et al (2015) Functionally defined therapeutic targets in diffuse intrinsic pontine glioma. Nat Med 21:555–559
Bleeker FE, Atai NA, Lamba S et al (2010) The prognostic IDH1R132 mutation is associated with reduced NADP+-dependent IDH activity in glioblastoma. Acta Neuropathol 119:487–494
Li S, Chou AP, Chen W et al (2013) Overexpression of isocitrate dehydrogenase mutant proteins renders glioma cells more sensitive to radiation. Neuro Oncol 15:57–68
Van Den Bent MJ, Dubbink HJ, Marie Y et al (2010) IDH1 and IDH2 mutations are prognostic but not predictive for outcome in anaplastic oligodendroglial tumors: a report of the European Organization for Research and Treatment of Cancer Brain Tumor Group. Clin Cancer Res 16:1597–1604
Schumacher T, Bunse L, Pusch S et al (2014) A vaccine targeting mutant IDH1 induces antitumour immunity. Nature 512:324–327
Delfanti RL, Piccioni DE, Handwerker J et al (2017) Imaging correlates for the 2016 update on WHO classification of grade II/III gliomas: implications for IDH, 1p/19q and ATRX status. J Neurooncol 135:601–609
Li F, Deng Z, Zhang L et al (2019) ATRX loss induces telomere dysfunction and necessitates induction of alternative lengthening of telomeres during human cell immortalization. EMBO J. 38:e96659
Vigneswaran K, Neill S, Hadjipanayis CG (2015) Beyond the World Health Organization grading of infiltrating gliomas: advances in the molecular genetics of glioma classification. Ann Transl Med 3:95
Weller M, Reifenberger G (2020) Beyond the World Health Organization classification of central nervous system tumors 2016: what are the new developments for gliomas from a clinician’s perspective? Curr Opin Neurol 33:701–706
Wiestler B, Capper D, Holland-Letz T et al (2013) ATRX loss refines the classification of anaplastic gliomas and identifies a subgroup of IDH mutant astrocytic tumors with better prognosis. Acta Neuropathol 126:443–451
Fack F, Tardito S, Hochart G et al (2017) Altered metabolic landscape in IDH -mutant gliomas affects phospholipid, energy, and oxidative stress pathways. EMBO Mol Med 9:1681–1695
Gillies RJ, Kinahan PE, Hricak H (2016) Radiomics: images are more than pictures, they are data. Radiology 278:563–577
Zhou M, Chaudhury B, Hall LO et al (2019) A multi-sequence and habitat-based MRI radiomics signature for preoperative prediction of MGMT promoter methylation in astrocytomas with prognostic implication. Eur Radiol 9:1–9
Ren Y, Zhang X, Rui W et al (2019) Noninvasive prediction of IDH1 mutation and ATRX expression loss in low-grade gliomas using multiparametric MR radiomic features. J Magn Reson Imaging 49:808–817
Liang S, Zhang R, Liang D et al (2018) Multimodal 3D densenet for IDH genotype prediction in gliomas. Genes (Basel) 9:382
Su C, Jiang J, Zhang S et al (2019) Radiomics based on multicontrast MRI can precisely differentiate among glioma subtypes and predict tumour-proliferative behaviour. Eur Radiol 29:1986–1996. https://doi.org/10.1007/s00330-018-5704-8
Tian Q, Yan LF, Zhang X et al (2018) Radiomics strategy for glioma grading using texture features from multiparametric MRI. J Magn Reson Imaging 48:1518–1528
Das D, Yoon B, Golden L et al (2017) Nimg-37. Correlation of Vasari-based Mri phenotypes with Mgmt and Idh status across glioma grades: a statistical analysis in 372 patients. Neuro Oncol 19:vi150–vi150
Stephen SFY, Hugo JWLA (2016) Applications and limitations of radiomics. Phys Med Biol 61:R150
Balachandran VP, Gonen M, Smith JJ, DeMatteo RP (2015) Nomograms in oncology: more than meets the eye. Lancet Oncol 16:e173–e180
Xu X, Wang H, Du P et al (2019) A predictive nomogram for individualized recurrence stratification of bladder cancer using multiparametric MRI and clinical risk factors. J Magn Reson Imaging 50:1893–1904
Zhang X, Lu H, Tian Q et al (2019) A radiomics nomogram based on multiparametric MRI might stratify glioblastoma patients according to survival. Eur Radiol 29:5528–5538
Zhang S, Zhang B, Tian J et al (2017) Radiomics features of multiparametric MRI as novel prognostic factors in advanced nasopharyngeal carcinoma. Clin Cancer Res 23:4259
Zhao L, Gong J, Xi Y et al (2020) MRI-based radiomics nomogram may predict the response to induction chemotherapy and survival in locally advanced nasopharyngeal carcinoma. Eur Radiol 30:537–546
Gittleman H, Lim D, Kattan MW et al (2017) An independently validated nomogram for individualized estimation of survival among patients with newly diagnosed glioblastoma: NRG Oncology RTOG 0525 and 0825. Neuro Oncol 19:669–677
Liu Z, Zhang T, Jiang H et al (2019) Conventional MR-based preoperative nomograms for prediction of IDH/1p19q subtype in low-grade glioma. Acad Radiol 26:1062–1070
Wang Y, Xin S, Zhang K et al (2019) Low GAS5 levels as a predictor of poor survival in patients with lower-grade gliomas. J Oncol 2019:1785042
Gorlia MT, Wu W, Wang M et al (2013) New validated prognostic models and prognostic calculators in patients with low-grade gliomas diagnosed by central pathology review: a pooled analysis of EORTC/RTOG/NCCTG phase III clinical trials. Neuro Oncol 15:1568–1579
Zhang M, Wang X, Chen X et al (2020) Novel immune-related gene signature for risk stratification and prognosis of survival in lower-grade glioma. Front Genet 11:363
Wang C, Qiu J, Chen S et al (2021) Prognostic model and nomogram construction based on autophagy signatures in lower grade glioma. J Cell Physiol 236:235–248
Braganza MZ, Kitahara CM, Berrington De González A et al (2012) Ionizing radiation and the risk of brain and central nervous system tumors: a systematic review. Neuro Oncol 14:1316–1324
Sun H, Wang S (2012) Penalized logistic regression for high-dimensional DNA methylation data with case-control studies. Bioinformatics 28:1368–1375
Yerukala Sathipati S, Ho SY (2017) Identifying the miRNA signature associated with survival time in patients with lung adenocarcinoma using miRNA expression profiles. Sci Rep 7:7507
Lee MK, Park JE, Jo Y et al (2020) Advanced imaging parameters improve the prediction of diffuse lower-grade gliomas subtype, IDH mutant with no 1p19q codeletion: added value to the T2/FLAIR mismatch sign. Eur Radiol 30:844–854
Kim M, Jung SY, Park JE et al (2020) Diffusion- and perfusion-weighted MRI radiomics model may predict isocitrate dehydrogenase (IDH) mutation and tumor aggressiveness in diffuse lower grade glioma. Eur Radiol 30:2142–2151
Choi YS, Ahn SS, Chang JH et al (2020) Machine learning and radiomic phenotyping of lower grade gliomas: improving survival prediction. Eur Radiol 30:3834–3842
Morshed RA, Han SJ, Hervey-Jumper SL et al (2019) Molecular features and clinical outcomes in surgically treated low-grade diffuse gliomas in patients over the age of 60. J Neurooncol 141:383–391. https://doi.org/10.1007/s11060-018-03044-4
Hyare H, Rice L, Thust S et al (2019) Modelling MR and clinical features in grade II/III astrocytomas to predict IDH mutation status. Eur J Radiol 114:120–127
Ebrahimi A, Skardelly M, Bonzheim I et al (2016) ATRX immunostaining predicts IDH and H3F3A status in gliomas. Acta Neuropathol Commun 4:60
Claus EB, Cannataro VL, Gaffney SG, Townsend JP (2021) Environmental and sex-specific molecular signatures of glioma causation. Neuro Oncol. https://doi.org/10.1093/neuonc/noab103
Ichimura S, Ohara K, Kono M et al (2021) Molecular investigation of brain tumors progressing during pregnancy or postpartum period: the association between tumor type, their receptors, and the timing of presentation. Clin Neurol Neurosurg. 207:106720
Cowppli-Bony A, Bouvier G, Rué M et al (2011) Brain tumors and hormonal factors: review of the epidemiological literature. Cancer Causes Control 22:697–714
Acknowledgements
This study is supported by Shanghai Science and Technology Commission (No. 18411967300).
Funding
This study has received funding from Shanghai Science and Technology Commission (No. 18411967300).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is Yan Ren.
Conflict of Interest
One of the authors (Yong Zhang) is an employee of GE Healthcare. The remaining authors declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and Biometry
No complex statistical methods were necessary for this paper.
Informed Consent
Written informed consent was waived by the Institutional Review Board.
Ethical Approval
Institutional Review Board approval was obtained.
Study subjects or cohorts overlap
Fifty-seven study subjects have been previously reported in Journal of Magnetic Resonance Imaging (2019, 49(3):808–817).
Methodology
• retrospective.
• diagnostic or prognostic study.
• performed at one institution.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, S., Zhang, X., Rui, W. et al. A nomogram strategy for identifying the subclassification of IDH mutation and ATRX expression loss in lower-grade gliomas. Eur Radiol 32, 3187–3198 (2022). https://doi.org/10.1007/s00330-021-08444-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-021-08444-1