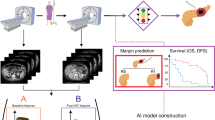
Abstract
Objectives
In resectable pancreatic ductal adenocarcinoma (PDAC), few pre-operative prognostic biomarkers are available. Radiomics has demonstrated potential but lacks external validation. We aimed to develop and externally validate a pre-operative clinical-radiomic prognostic model.
Methods
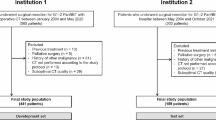
Retrospective international, multi-center study in resectable PDAC. The training cohort included 352 patients (pre-operative CTs from five Canadian hospitals). Cox models incorporated (a) pre-operative clinical variables (clinical), (b) clinical plus CT-radiomics, and (c) post-operative TNM model, which served as the reference. Outcomes were overall (OS)/disease-free survival (DFS). Models were assessed in the validation cohort from Ireland (n = 215, CTs from 34 hospitals), using C-statistic, calibration, and decision curve analyses.
Results
The radiomic signature was predictive of OS/DFS in the validation cohort, with adjusted hazard ratios (HR) 2.87 (95% CI: 1.40–5.87, p < 0.001)/5.28 (95% CI 2.35–11.86, p < 0.001), respectively, along with age 1.02 (1.01–1.04, p = 0.01)/1.02 (1.00–1.04, p = 0.03). In the validation cohort, median OS was 22.9/37 months (p = 0.0092) and DFS 14.2/29.8 (p = 0.0023) for high-/low-risk groups and calibration was moderate (mean absolute errors 7%/13% for OS at 3/5 years). The clinical-radiomic model discrimination (C = 0.545, 95%: 0.543–0.546) was higher than the clinical model alone (C = 0.497, 95% CI 0.496–0.499, p < 0.001) or TNM (C = 0.525, 95% CI: 0.524–0.526, p < 0.001). Despite superior net benefit compared to the clinical model, the clinical-radiomic model was not clinically useful for most threshold probabilities.
Conclusion
A multi-institutional pre-operative clinical-radiomic model for resectable PDAC prognostication demonstrated superior net benefit compared to a clinical model but limited clinical utility at external validation. This reflects inherent limitations of radiomics for PDAC prognostication, when deployed in real-world settings.
Key Points
• At external validation, a pre-operative clinical-radiomics prognostic model for pancreatic ductal adenocarcinoma (PDAC) outperformed pre-operative clinical variables alone or pathological TNM staging.
• Discrimination and clinical utility of the clinical-radiomic model for treatment decisions remained low, likely due to heterogeneity of CT acquisition parameters.
• Despite small improvements, prognosis in PDAC using state-of-the-art radiomics methodology remains challenging, mostly owing to its low discriminative ability. Future research should focus on standardization of CT protocols and acquisition parameters.





Similar content being viewed by others
Abbreviations
- AI:
-
Artificial intelligence
- AJCC:
-
American Joint Committee on Cancer
- CI:
-
Confidence intervals
- CPH:
-
Cox proportional hazard
- CT:
-
Computed tomography
- DCA:
-
Decision curve analysis
- DICOM:
-
Digital Imaging and Communications in Medicine
- GLCM:
-
Gray-level co-occurrence matrix
- HR:
-
Hazard ratio
- IBSI:
-
Image Biomarker Standardization Initiative
- ISI:
-
Image-to-surgery time interval in days
- LASSO:
-
Least absolute shrinkage and selection operator
- MVA:
-
Multivariate analysis
- OS:
-
Overall survival
- PACS:
-
Picture archiving and communication system
- PDAC:
-
Pancreatic ductal adenocarcinoma
- PV:
-
Portal venous
- QIBA:
-
Quantitative Imaging Biomarker Alliance
- RIS:
-
Radiology information system
- TNM:
-
Tumor Node Metastasis staging system
- TRIPOD:
-
Transparent Reporting of multivariable prediction model for Individual Prognosis or Diagnosis statement
References
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. https://doi.org/10.3322/caac.21660
National Cancer Registry Ireland. Pancreas Factsheet. Available via https://www.ncri.ie/sites/ncri/files/factsheets/Factsheet%20pancreas.pdf. Accessed February 3rd 2021
(2017) Canadian Cancer Statistics 2017. Special topic: pancreatic cancer. Available via https://www.cancer.ca/~/media/cancer.ca/CW/cancer%20information/cancer%20101/Canadian%20cancer%20statistics/Canadian-Cancer-Statistics-2017-EN.pdf. Accessed February 3rd 2021Accessed February 3rd 2021
Sun H, Ma H, Hong G, Sun H, Wang J (2014) Survival improvement in patients with pancreatic cancer by decade: a period analysis of the SEER database, 1981–2010. Sci Rep 4:6747
Huang L, Jansen L, Balavarca Y et al (2019) Resection of pancreatic cancer in Europe and USA: an international large-scale study highlighting large variations. Gut 68:130–139
Kim JR, Kim H, Kwon W, Jang JY, Kim SW (2021) Pattern of local recurrence after curative resection in pancreatic ductal adenocarcinoma according to the initial location of the tumor. J Hepatobiliary Pancreat Sci 28:105–114
Gaskill CE, Maxwell J, Ikoma N et al (2021) History of preoperative therapy for pancreatic cancer and the MD Anderson experience. J Surg Oncol 123:1414–1422
Schneider M, Neoptolemos JP, Büchler MW (2020) Commentary: Neoadjuvant treatment of resectable pancreatic cancer: lack of level III evidence. Surgery 168:1015–1016
Versteijne E, Suker M, Groothuis K et al (2020) Preoperative chemoradiotherapy versus immediate surgery for resectable and borderline resectable pancreatic cancer: results of the Dutch Randomized Phase III PREOPANC Trial. J Clin Oncol 38:1763–1773
O’Reilly EM, Ferrone C (2020) Neoadjuvant or adjuvant therapy for resectable or borderline resectable pancreatic cancer: which is preferred? J Clin Oncol 38:1757–1759
Haab BB, Huang Y, Balasenthil S et al (2015) Definitive characterization of CA 19–9 in resectable pancreatic cancer using a reference set of serum and plasma specimens. PLoS One 10:e0139049
Xie T, Wang X, Li M, Tong T, Yu X, Zhou Z (2020) Pancreatic ductal adenocarcinoma: a radiomics nomogram outperforms clinical model and TNM staging for survival estimation after curative resection. Eur Radiol 30:2513–2524
Cassinotto C, Chong J, Zogopoulos G et al (2017) Resectable pancreatic adenocarcinoma: role of CT quantitative imaging biomarkers for predicting pathology and patient outcomes. Eur J Radiol 90:152–158
Park JK, Paik WH, Ryu JK et al (2013) Clinical significance and revisiting the meaning of CA 19–9 blood level before and after the treatment of pancreatic ductal adenocarcinoma: analysis of 1,446 patients from the pancreatic cancer cohort in a single institution. PLoS One 8:e78977
Xu D, Zhang K, Li M et al (2020) Prognostic nomogram for resected pancreatic adenocarcinoma: a TRIPOD-compliant retrospective long-term survival analysis. World J Surg 44:1260–1269
Brennan MF, Kattan MW, Klimstra D, Conlon K (2004) Prognostic nomogram for patients undergoing resection for adenocarcinoma of the pancreas. Ann Surg 240:293–298
Grossmann P, Stringfield O, El-Hachem N et al (2017) Defining the biological basis of radiomic phenotypes in lung cancer. Elife 6:e23421. https://doi.org/10.7554/eLife.23421
Li H, Zhu Y, Burnside ES et al (2016) Quantitative MRI radiomics in the prediction of molecular classifications of breast cancer subtypes in the TCGA/TCIA data set. NPJ Breast Cancer 2:16012
Zwanenburg A, Vallières M, Abdalah MA et al (2020) The Image Biomarker Standardization Initiative: standardized quantitative radiomics for high-throughput image-based phenotyping. Radiology 295:328–338
Abunahel BM, Pontre B, Kumar H, Petrov MS (2020) Pancreas image mining: a systematic review of radiomics. Eur Radiol. https://doi.org/10.1007/s00330-020-07376-6
Li K, Xiao J, Yang J et al (2019) Association of radiomic imaging features and gene expression profile as prognostic factors in pancreatic ductal adenocarcinoma. Am J Transl Res 11:4491–4499
Shi H, Wei Y, Cheng S et al (2021) Survival prediction after upfront surgery in patients with pancreatic ductal adenocarcinoma: radiomic, clinic-pathologic and body composition analysis. Pancreatology. https://doi.org/10.1016/j.pan.2021.02.009
Attiyeh MA, Chakraborty J, Doussot A et al (2018) Survival prediction in pancreatic ductal adenocarcinoma by quantitative computed tomography image analysis. Ann Surg Oncol 25:1034–1042
Eilaghi A, Baig S, Zhang Y et al (2017) CT texture features are associated with overall survival in pancreatic ductal adenocarcinoma - a quantitative analysis. BMC Med Imaging 17:38
Yun G, Kim YH, Lee YJ, Kim B, Hwang JH, Choi DJ (2018) Tumor heterogeneity of pancreas head cancer assessed by CT texture analysis: association with survival outcomes after curative resection. Sci Rep 8:7226
Kim HS, Kim YJ, Kim KG, Park JS (2019) Preoperative CT texture features predict prognosis after curative resection in pancreatic cancer. Sci Rep 9:17389
Zhang H, Dullerud N, Seyyed-Kalantari L, Morris Q, Joshi S, Ghassemi M (2021) An empirical framework for domain generalization in clinical settings. Proceedings of the Conference on Health, Inference, and Learning, pp 279–290. https://doi.org/10.1145/3450439.3451878
Khalvati F, Zhang Y, Baig S et al (2019) Prognostic value of CT radiomic features in resectable pancreatic ductal adenocarcinoma. Sci Rep 9:5449
Zhang Y, Lobo-Mueller EM, Karanicolas P, Gallinger S, Haider MA, Khalvati F (2020) CNN-based survival model for pancreatic ductal adenocarcinoma in medical imaging. BMC Med Imaging 20:11
Zhang Y, Lobo-Mueller EM, Karanicolas P, Gallinger S, Haider MA, Khalvati F (2021) Improving prognostic performance in resectable pancreatic ductal adenocarcinoma using radiomics and deep learning features fusion in CT images. Sci Rep 11:1378
Lambin P, Leijenaar RTH, Deist TM et al (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14:749–762
Collins GS, Reitsma JB, Altman DG, Moons KG (2015) Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD): the TRIPOD Statement. Br J Surg 102:148–158
Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A, editors. (2010) AJCC Cancer Staging Manual (7th ed). Springer, New York, NY
Fedorov A, Beichel R, Kalpathy-Cramer J et al (2012) 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magn Reson Imaging 30:1323–1341
van Griethuysen JJM, Fedorov A, Parmar C et al (2017) Computational radiomics system to decode the radiographic phenotype. Cancer Res 77:e104–e107
The image biomarker standardisation initiative. Available via https://ibsi.readthedocs.io/en/latest/index.html
Fornacon-Wood I, Mistry H, Ackermann CJ et al (2020) Reliability and prognostic value of radiomic features are highly dependent on choice of feature extraction platform. Eur Radiol. https://doi.org/10.1007/s00330-020-06957-9
Mackin D, Fave X, Zhang L et al (2017) Harmonizing the pixel size in retrospective computed tomography radiomics studies. PLoS One 12:e0178524
Shafiq-Ul-Hassan M, Latifi K, Zhang G, Ullah G, Gillies R, Moros E (2018) Voxel size and gray level normalization of CT radiomic features in lung cancer. Sci Rep 8:10545
Ligero M, Jordi-Ollero O, Bernatowicz K et al (2021) Minimizing acquisition-related radiomics variability by image resampling and batch effect correction to allow for large-scale data analysis. Eur Radiol 31:1460–1470
Berenguer R, Pastor-Juan MDR, Canales-Vázquez J et al (2018) Radiomics of CT features may be nonreproducible and redundant: influence of CT acquisition parameters. Radiology 288:407–415
Fortin JP, Parker D, Tunç B et al (2017) Harmonization of multi-site diffusion tensor imaging data. Neuroimage 161:149–170
Orlhac F, Frouin F, Nioche C, Ayache N, Buvat I (2019) Validation of A method to compensate multicenter effects affecting CT radiomics. Radiology 291:53–59
Little R (1988) Missing-data adjustments in large surveys. J Bus Econ Stat 6:287–296
Steyerberg E (2009) Clinical prediction models: a practical approach to development, validation and updating, 2nd edn. Springer
Eid M, Ostřížková L, Kunovský L et al (2021) Current view of neoadjuvant chemotherapy in primarily resectable pancreatic adenocarcinoma. Neoplasma 68:1–9
Tempero MA, Malafa MP, Al-Hawary M et al (2021) Pancreatic Adenocarcinoma, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 19:439-457
Roalsø M, Aunan JR, Søreide K (2020) Refined TNM-staging for pancreatic adenocarcinoma – real progress or much ado about nothing? Eur J Surg Oncol 46:1554–1557
Chang JC, Kundranda M (2017) Novel Diagnostic and Predictive Biomarkers in Pancreatic Adenocarcinoma. Int J Mol Sci 18:667
Guo SW, Shen J, Gao JH et al (2020) A preoperative risk model for early recurrence after radical resection may facilitate initial treatment decisions concerning the use of neoadjuvant therapy for patients with pancreatic ductal adenocarcinoma. Surgery 168:1003–1014
Vickers AJ, Cronin AM (2010) Everything you always wanted to know about evaluating prediction models (but were too afraid to ask). Urology 76:1298–1301
He L, Huang Y, Ma Z, Liang C, Liang C, Liu Z (2016) Effects of contrast-enhancement, reconstruction slice thickness and convolution kernel on the diagnostic performance of radiomics signature in solitary pulmonary nodule. Sci Rep 6:34921
Chu LC, Solmaz B, Park S et al (2020) Diagnostic performance of commercially available vs. in-house radiomics software in classification of CT images from patients with pancreatic ductal adenocarcinoma vs. healthy controls. Abdom Radiol (NY) 45:2469–2475
Koay EJ, Truty MJ, Cristini V et al (2014) Transport properties of pancreatic cancer describe gemcitabine delivery and response. J Clin Invest 124:1525–1536
Fukukura Y, Takumi K, Higashi M et al (2014) Contrast-enhanced CT and diffusion-weighted MR imaging: performance as a prognostic factor in patients with pancreatic ductal adenocarcinoma. Eur J Radiol 83:612–619
Ganeshan B, Panayiotou E, Burnand K, Dizdarevic S, Miles K (2012) Tumour heterogeneity in non-small cell lung carcinoma assessed by CT texture analysis: a potential marker of survival. Eur Radiol 22:796–802
Larue R, Klaassen R, Jochems A et al (2018) Pre-treatment CT radiomics to predict 3-year overall survival following chemoradiotherapy of esophageal cancer. Acta Oncol 57:1475–1481
Shim KY, Chung SW, Jeong JH et al (2021) Radiomics-based neural network predicts recurrence patterns in glioblastoma using dynamic susceptibility contrast-enhanced MRI. Sci Rep 11:9974–9974
Kulkarni A, Carrion-Martinez I, Dhindsa K, Alaref AA, Rozenberg R, van der Pol CB (2020) Pancreas adenocarcinoma CT texture analysis: comparison of 3D and 2D tumor segmentation techniques. Abdom Radiol (NY). https://doi.org/10.1007/s00261-020-02759-1
Ng F, Kozarski R, Ganeshan B, Goh V (2013) Assessment of tumor heterogeneity by CT texture analysis: can the largest cross-sectional area be used as an alternative to whole tumor analysis? Eur J Radiol 82:342–348
Lubner MG, Smith AD, Sandrasegaran K, Sahani DV, Pickhardt PJ (2017) CT texture analysis: definitions, applications, biologic correlates, and challenges. Radiographics 37:1483–1503
Yamashita R, Perrin T, Chakraborty J et al (2020) Radiomic feature reproducibility in contrast-enhanced CT of the pancreas is affected by variabilities in scan parameters and manual segmentation. Eur Radiol 30:195–205
Harrell FE Jr, Lee KL, Mark DB (1996) Multivariable prognostic models: issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat Med 15:361–387
Funding
M.A.H receives funding from the Ontario Institute for Cancer Research (OICR) Translational Research Initiative in Pancreatic Cancer and the Clinical Investigator Program. GMH is a Clinical Research Fellow who is funded by a research grant from the Faculty of Radiologists, Royal College of Surgeons in Ireland. DD is a research fellow who is funded by a DFG (Deutsche Forschungsgemeinschaft) Fellowship DE 3207/1–1 (DD).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Dr Masoom Haider
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
Xin Dong and Rahi Jain (both co-authors) kindly provided statistical advice for this manuscript.
Informed consent
The need for informed consent was waived by research ethics boards.
Ethical approval
Institutional Review Board approval was obtained.
Study subjects or cohorts overlap
Fifty-five patients within the North-American cohort have been previously used in a PDAC Radiomics prognostication study which generated three publications.
Methodology
• Retrospective
• Diagnostic or prognostic study
• Performed at multiple institutions
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Healy, G.M., Salinas-Miranda, E., Jain, R. et al. Pre-operative radiomics model for prognostication in resectable pancreatic adenocarcinoma with external validation. Eur Radiol 32, 2492–2505 (2022). https://doi.org/10.1007/s00330-021-08314-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-021-08314-w