Abstract
Objectives
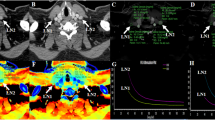
To explore the importance of quantitative characteristics of dual-energy CT (DECT) between pulmonary metastasis and benign lung nodules in thyroid cancer.
Methods
In this retrospective study, we identified 63 patients from our institution’s database with pathologically proven thyroid cancer who underwent DECT to assess pulmonary metastasis. Among these patients, 22 had 55 pulmonary metastases, and 41 had 97 benign nodules. If nodules showed increased iodine uptake on I-131 single-photon emission computed tomography-computed tomography or increased size in follow-up CT, they were considered metastatic. We compared the clinical findings and DECT parameters of both groups and performed a receiver operating characteristic analysis to evaluate the optimal cutoff values of the DECT parameters.
Results
Patients with metastases were significantly older than patients with benign nodules (p = 0.048). The DECT parameters of the metastatic nodules were significantly higher than those of the benign nodules (iodine concentration [IC], 5.61 ± 2.02 mg/mL vs. 1.61 ± 0.98 mg/mL; normalized IC [NIC], 0.60 ± 0.20 vs. 0.16 ± 0.11; NIC using pulmonary artery [NICPA], 0.60 ± 0.44 vs. 0.15 ± 0.11; slope of the spectral attenuation curves [λHU], 5.18 ± 2.54 vs. 2.12 ± 1.39; and Z-effective value [Zeff], 10.0 ± 0.94 vs. 8.79 ± 0.75; all p < 0.001). In the subgroup analysis according to nodule size, all DECT parameters of the metastatic nodules in all subgroups were significantly higher than those of the benign nodules (all p < 0.05). The cutoff values for IC, NIC, λHU, NICPA, and Zeff for diagnosing metastases were 3.10, 0.29, 3.57, 0.28, and 9.34, respectively (all p < 0.001).
Conclusions
DECT parameters can help to differentiate metastatic and benign lung nodules in thyroid cancer.
Key Points
• DECT parameters can help to differentiate metastatic and benign lung nodules in patients with thyroid cancer.
• DECT parameters showed a significant difference between benign lung nodules and lung metastases, even for nodules with diameters ≥ 3 mm and < 5 mm.
• Among the DECT parameters, the highest diagnostic accuracy for differentiating pulmonary metastases from benign lung nodules was achieved with the NIC and IC, followed by the NIC PA and λHU, and their cutoff values were 0.29, 3.10, 0.28, and 3.57, respectively.



Similar content being viewed by others
Abbreviations
- DECT:
-
Dual-energy CT
- IC:
-
Iodine concentration
- LN:
-
Lymph node
- NIC:
-
Normalized iodine concentration
- Tg:
-
Thyroglobulin
- TSH:
-
Thyroid-stimulating hormone
- VMI:
-
Virtual monochromatic image
- Z eff :
-
Z-effective value
- λHU:
-
Slope of the spectral attenuation curve
References
Kim H, Kim HI, Kim SW et al (2018) Prognosis of differentiated thyroid carcinoma with initial distant metastasis: a multicenter study in Korea. Endocrinol Metab (Seoul) 33:287–295
Kim M, Kim WG, Park S et al (2017) Initial size of metastatic lesions is best prognostic factor in patients with metastatic differentiated thyroid carcinoma confined to the lung. Thyroid 27:49–58
Sabra MM, Ghossein R, Tuttle RM (2016) Time course and predictors of structural disease progression in pulmonary metastases arising from follicular cell-derived thyroid cancer. Thyroid 26:518–524
Cho SW, Choi HS, Yeom GJ et al (2014) Long-term prognosis of differentiated thyroid cancer with lung metastasis in Korea and its prognostic factors. Thyroid 24:277–286
Hirsch D, Levy S, Tsvetov G et al (2017) Long-term outcomes and prognostic factors in patietns with differentiated thyroid cancer and distant metastases. Endocr Pract 23:1193–1200
Shoup M, Stojadinovic A, Nissan A et al (2003) Prognostic indicators of outcomes in patients with distant metastases from differentiated thyroid carcinoma. J Am Coll Surg 197:191–197
Giraudet AL, Vanel D, Leboulleux S et al (2007) Imaging medullary thyroid carcinoma with persistent elevated calcitonin levels. J Clin Endocrinol Metab 92:4185–4190
Schlumberger M, Bastholt L, Dralle H, Jarzab B, Pacini F, Smit J (2012) 2012 European Thyroid Association guidelines for metastatic medullary thyroid cancer. Eur Thyroid J 1:5–14
Ibrahimpasic T, Ghossein R, Carlson DL et al (2013) Poorly differentiated thyroid carcinoma presenting with gross extrathyroidal extension: 1986–2009 Memorial Sloan-Kettering Cancer Center experience. Thyroid 23:997–1002
Kim C, Kim W, Park SJ et al (2020) Application of dual-energy spectral computed tomography to thoracic oncology imaging. Korean J Radiol 21:838–850
Deniffel D, Sauter A, Dangelmaier J, Fingerle A, Rummeny EJ, Pfeiffer D (2019) Differentiating intrapulmonary metastases from different primary tumors via quantitative dual-energy CT based iodine concentration and conventional CT attenuation. Eur J Radiol 111:6–13
Wen Q, Yue Y, Shang J, Lu X, Gao L, Hou Y (2021) The application of dual-layer spectral detector computed tomography in solitary pulmonary nodule identification. Quant Imaging Med Surg 11:521–532
Zhang Y, Cheng J, Hua X et al (2016) Can spectral CT imaging improve the differentiation between malignant and benign solitary pulmonary nodules? PLoS One 11:e0147537
Chen ML, Li XT, Wei YY, Qi LP, Sun YS (2019) Can spectral computed tomography imaging improve the differentiation between malignant and benign pulmonary lesions manifesting as solitary pure ground glass, mixed ground glass, and solid nodules? Thoracic Cancer 10:234–242
Wu L, Cao G, Zhao L et al (2018) Spectral CT analysis of solitary pulmonary nodules for differentiating malignancy from benignancy: the value of iodine concentration spatial distribution difference. Biomed Res Int 2018:4830659
Hou WS, Wu HW, Yin Y, Cheng JJ, Zhang Q, Xu JR (2015) Differentiation of lung cancers from inflammatory masses with dual-energy spectral CT imaging. Acad Radiol 22:337–344
Bankier AA, MacMahon H, Goo JM, Rubin GD, Schaefer-Prokop CM, Naidich DP (2017) Recommendations for measuring pulmonary nodules at CT: a statement from the Fleischner Society. Radiology 285:584–600
MacMahon H, Naidich DP, Goo JM et al (2017) Guidelines for management of incidental pulmonary nodules detected on CT images: from the Fleischner Society 2017. Radiology 284:228–243
Casara D, Rubello D, Saladini G et al (1993) Different features of pulmonary metastases in differentiated thyroid cancer: natural history and multivariate statistical analysis of prognostic variables. J Nucl Med 34:1626–1631
Degroot LJ, Kaplan EL, McCormick M, Straus FH (1990) Natural history, treatment, and course of papillary thyroid carcinoma. J Clin Endocrinol Metab 71:414–424
Thoresen S, Akslen L, Glattre E, Haldorsen T, Lund E, Schoultz M (1989) Survival and prognostic factors in differentiated thyroid cancer–a multivariate analysis of 1,055 cases. Br J Cancer 59:231–235
Martin S, Maurice T, Vathaire FD et al (1986) Long-term results of treatment of 283 patients with lung and bone metastases from differentiated thyroid carcinoma. J Clin Endocrinol Metab 63:960–967
Küçük NO, Külak HA, Aras G (2006) Clinical importance of technetium-99m-methoxyisobutylisonitrile (MIBI) scintigraphy in differentiated thyroid carcinoma patients with elevated thyroglobulin levels and negative I-131 scanning results. Ann Nucl Med 20:393–397
Hung MC, Wu HS, Kao CH, Chen WK, Changlai SP (2003) F18-fluorodeoxyglucose positron emission tomography in detecting metastatic papillary thyroid carcinoma with elevated human serum thyroglobulin levels but negative I-131 whole body scan. Endocr Res 29:169–175
Vural GU, Akkas BE, Ercakmak N, Basu S, Alavi A (2012) Prognostic significance of FDG PET/CT on the follow-up of patients of differentiated thyroid carcinoma with negative 131I whole-body scan and elevated thyroglobulin levels: Correlation with clinical and histopathologic characteristics and long-term follow-up data. Clin Nucl Med 37:953–959
Liu X, Ouyang D, Li H et al (2015) Papillary thyroid cancer: Dual-energy spectral CT quantitative parameters for preoperative diagnosis of metastasis to the cervical lymph nodes. Radiology 275:167–176
Zhao Y, Li X, Li L et al (2017) Preliminary study on the diagnostic value of single-source dual-energy CT in diagnosing cervical lymph node metastasis of thyroid carcinoma. J Thorac Dis 9:4758–4766
He M, Lin C, Yin L, Lin Y, Zhang S, Ma M (2019) Value of dual-energy computed tomography for diagnosing cervical lymph node metastasis in patients with papillary thyroid cancer. J Comput Assist Tomogr 43:970–975
Gao SY, Zhang XY, Wei W et al (2016) Identification of benign and malignant thyroid nodules by in vivo iodine concentration measurement using single-source dual-energy CT: A retrospective diagnostic accuracy study. Medicine (Baltimore) 95:e4816
Wu F, Zhou H, Li F, Wang JT, Ai T (2018) Spectral CT imaging of lung cancer: Quantitative analysis of spectral parameters and their correlation with tumor characteristics. Acad Radiol 25:1398–1404
Jia Y, Xiao X, Sun Q, Jiang H (2018) CT spectral parameters and serum tumour markers to differentiate histological types of cancer histology. Clin Radiol 73:1033–1040
Seo JB, Im JG, Goo JM, Chung MJ, Kim MY (2001) Atypical pulmonary metastases: spectrum of radiologic findings. Radiographics 21:403–417
Hung WW, Wang CS, Tsai KB, Ou-Yang F, Shin SJ, Hsiao PJ (2009) Medullary thyroid carcinoma with poor differentiation and atypical radiographic pattern of metastasis. Pathol Int 59:660–663
Jimenez JM, Casey SO, Citron M, Khan A (1995) Calcified pulmonary metastases from medullary carcinoma of the thyroid. Comput Med Imaging Graph 19:325–328
Maile CW, Rodan BA, Godwin JD, Chen JT, Ravin CE (1982) Calcification in pulmonary metastases. Br J Radiol 55:108–113
Lennartz S, Mager A, Große Hokamp N et al (2021) Texture analysis of iodine maps and conventional images for k-nearest neighbor classification of benign and metastatic lung nodules. Cancer Imaging 21:17
Lennartz S, Parakh A, Cao J, Zopfs D, Große Hokamp N, Kambadakone A (2021) Inter-scan and inter-scanner variation of quantitative dual-energy CT: evaluation with three different scanner types. Eur Radiol. https://doi.org/10.1007/s00330-020-07611-0
Zopfs D, Reimer RP, Sonnabend K et al (2021) Intraindividual consistency of iodine concentration in dual-energy computed tomography of the chest and abdomen. Invest Radiol 56:181–187
Zopfs D, Graffe J, Reimer RP et al (2021) Quantitative distribution of iodinated contrast media in body computed tomography: data from a large reference cohort. Eur Radiol 31:2340–2348
Funding
This research was funded by the National Research Foundation of Korea (NRF) (NRF-2018R1D1A1B07049989), Korea University Ansan Hospital research grant (O1903581), and Korea University grant (K2008271).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Cherry Kim.
Conflict of interest
The authors declare no competing interests.
Statistics and biometry
Jaehyung Cha, one of the authors, has contributed to the statistical analysis.
Informed consent
Written informed consent was waived by the institutional review board.
Ethical approval
Institutional review board approval was obtained.
Methodology
• retrospective.
• case–control study.
• performed at one institution.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ha, T., Kim, W., Cha, J. et al. Differentiating pulmonary metastasis from benign lung nodules in thyroid cancer patients using dual-energy CT parameters. Eur Radiol 32, 1902–1911 (2022). https://doi.org/10.1007/s00330-021-08278-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-021-08278-x