Abstract
Objectives
To develop a simplified MRI-based model to predict the risk for positive surgical margins (PSMs) after radical prostatectomy (RP) in patients with prostate cancer (PCa).
Methods
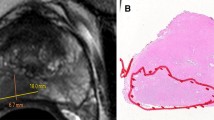
Consecutive patients who underwent RP for PCa were retrospectively identified from a tertiary referral hospital. Patients who underwent RP between January 2014 and June 2014 were assigned as derivation cohort (n = 330) and those between January 2018 and February 2018 were assigned as validation cohort (n = 100). MRI-based predictors associated with PSM were assessed: tumor size, tumor-capsule contact length, the Prostate Imaging Reporting and Data System (PI-RADS) category, tumor location (tumor contact to the apex or posterolateral side near the neurovascular bundle), apical depth, and prostate volume. A prediction model was developed by using multivariable logistic regression, and then it was transformed into a scoring system. The prediction and calibration performance of this scoring system was evaluated using the C statistics and Hosmer-Lemeshow goodness-of-fit test.
Results
A total of 121 (36.7%) and 32 (32.0%) of patients in the derivation and validation cohorts had PSMs after RP. The scoring system consisted of the following variables: tumor-capsule contact length, PI-RADS category, tumor located at the apex and/or posterolateral side. This scoring system provided good prediction performance for PSM in the derivation (C statistics, 0.80 [95% CI: 0.76, 0.85]) and validation (C statistics, 0.77 [95% CI: 0.68, 0.87]) cohorts, and also showed good calibration in both cohorts (p = 0.83 and 0.86, respectively).
Conclusions
An MRI-based scoring system can help estimate the risk of PSM after RP.
Key Points
• An MRI-based scoring system served as a tool to estimate the risk of positive surgical margin (C statistics, 0.80 and 0.77 in the derivation and validation cohorts, respectively) after radical prostatectomy.
• Tumor with contact to the apex or posterolateral aspect, the tumor contact length to capsule, and higher PI-RADS category were independent predictors for the presence of positive resection margins after radical prostatectomy in men with prostate cancer.
• High-risk patients as determined by the scoring system demonstrated adverse post-surgical outcomes compared with low- or intermediate-risk patients, in regard to longer length (mean length, 13.0 mm versus 3.9 mm in low risk or 6.2 mm in intermediate risk; p ≤ 0.001) and higher Gleason grade at the margin (grades 4 and 5 in 69.4% and 20.4% versus 16.7% and 16.7% in low risk or 46.7% and 5.4% in intermediate risk; p < 0.001).






Similar content being viewed by others
Abbreviations
- ADC:
-
Apparent diffusion coefficient
- DWI:
-
Diffusion-weighted image
- EPE:
-
Extraprostatic extension
- mpMRI:
-
Multiparametric magnetic resonance imaging
- PI-RADS:
-
Prostate Imaging Reporting and Data System
- PSA:
-
Prostate-specific antigen
- PSM:
-
Positive surgical margin
- RP:
-
Radical prostatectomy
- TCL:
-
Tumor-capsule contact length
- T2WI:
-
T2-weighted image
References
Novara G, Ficarra V, Mocellin S et al (2012) Systematic review and meta-analysis of studies reporting oncologic outcome after robot-assisted radical prostatectomy. Eur Urol 62:382–404
Yossepowitch O, Briganti A, Eastham JA et al (2014) Positive surgical margins after radical prostatectomy: a systematic review and contemporary update. Eur Urol 65:303–313
Tian X-J, Wang Z-L, Li G et al (2019) Development and validation of a preoperative nomogram for predicting positive surgical margins after laparoscopic radical prostatectomy. Chin Med J (Engl) 132:928–934
Cheng L, Slezak J, Bergstralh EJ, Myers RP, Zincke H, Bostwick DG (2000) Preoperative prediction of surgical margin status in patients with prostate cancer treated by radical prostatectomy. J Clin Oncol 18:2862–2868
Patel VR, Coelho RF, Rocco B et al (2011) Positive surgical margins after robotic assisted radical prostatectomy: a multi-institutional study. J Urol 186:511–516
McEvoy SH, Raeside MC, Chaim J, Ehdaie B, Akin O (2018) Preoperative prostate MRI: a road map for surgery. AJR Am J Roentgenol 211:383–391
Matikainen MP, Von Bodman CJ, Secin FP et al (2010) The depth of the prostatic apex is an independent predictor of positive apical margins at radical prostatectomy. BJU Int 106:622–626
Mehralivand S, Shih JH, Harmon S et al (2019) A grading system for the assessment of risk of extraprostatic extension of prostate cancer at multiparametric MRI. Radiology 290:709–719
Park KJ, Kim MH, Kim JK (2020) Extraprostatic tumor extension: comparison of preoperative multiparametric MRI criteria and histopathologic correlation after radical prostatectomy. Radiology. https://doi.org/10.1148/radiol.2020192133:192133
Petralia G, Musi G, Padhani AR et al (2015) Robot-assisted radical prostatectomy: multiparametric MR imaging-directed intraoperative frozen-section analysis to reduce the rate of positive surgical margins. Radiology 274:434–444
Alessi S, Pricolo P, Summers P et al (2019) Low PI-RADS assessment category excludes extraprostatic extension (≥pT3a) of prostate cancer: a histology-validated study including 301 operated patients. Eur Radiol. https://doi.org/10.1007/s00330-019-06092-0
Baco E, Rud E, Vlatkovic L et al (2015) Predictive value of magnetic resonance imaging determined tumor contact length for extracapsular extension of prostate cancer. J Urol 193:466–472
Turkbey B, Rosenkrantz AB, Haider MA et al (2019) Prostate Imaging Reporting and Data System version 2.1: 2019 update of Prostate Imaging Reporting and Data System version 2. Eur Urol. https://doi.org/10.1016/j.eururo.2019.02.033
Han K, Song K, Choi BW (2016) How to develop, validate, and compare clinical prediction models involving radiological parameters: study design and statistical methods. Korean J Radiol 17:339–350
Harrell FE Jr, Lee KL, Mark DB (1996) Multivariable prognostic models: issues in developing models, evaluating assumptions and adequacy, and measuring and reducing errors. Stat Med 15:361–387
de Rooij M, Hamoen EHJ, Witjes JA, Barentsz JO, Rovers MM (2016) Accuracy of magnetic resonance imaging for local staging of prostate cancer: a diagnostic meta-analysis. Eur Urol 70:233–245
Lee IJ, Oh JJ, Kim TJ et al (2016) Clinical significance of positive surgical margin after radical prostatectomy according to pathological stage. Korean J Urol Oncol 14:159–164
Roethke MC, Lichy MP, Kniess M et al (2013) Accuracy of preoperative endorectal MRI in predicting extracapsular extension and influence on neurovascular bundle sparing in radical prostatectomy. World J Urol 31:1111–1116
Partin AW, Borland RN, Epstein JI, Brendler CB (1993) Influence of wide excision of the neurovascular bundle(s) on prognosis in men with clinically localized prostate cancer with established capsular penetration. J Urol 150:142–146 discussion 146-148
Ahn H, Hwang SI, Lee HJ et al (2020) Prediction of extraprostatic extension on multi-parametric magnetic resonance imaging in patients with anterior prostate cancer. Eur Radiol 30:26–37
Rosenkrantz AB, Shanbhogue AK, Wang A, Kong MX, Babb JS, Taneja SS (2016) Length of capsular contact for diagnosing extraprostatic extension on prostate MRI: assessment at an optimal threshold. J Magn Reson Imaging 43:990–997
Chuang AY, Nielsen ME, Hernandez DJ, Walsh PC, Epstein JI (2007) The significance of positive surgical margin in areas of capsular incision in otherwise organ confined disease at radical prostatectomy. J Urol 178:1306–1310
Kang YJ, Abalajon MJ, Jang WS et al (2016) Association of anterior and lateral extraprostatic extensions with base-positive resection margins in prostate cancer. PLoS One 11:e0158922
Park S-W, Readal N, Jeong BC et al (2015) Risk factors for intraprostatic incision into malignant glands at radical prostatectomy. Eur Urol 68:311–316
Van den Broeck T, van den Bergh RCN, Briers E et al (2019) Biochemical recurrence in prostate cancer: the European Association of Urology Prostate Cancer guidelines panel recommendations. Eur Urol Focus. https://doi.org/10.1016/j.euf.2019.06.004
Philippou Y, Harriss E, Davies L et al (2018) Prostatic capsular incision during radical prostatectomy has important oncological implications: a systematic review and meta-analysis. BJU Int. https://doi.org/10.1111/bju.14522
Kwak KW, Lee HM, Choi HY (2010) Impact of capsular incision on biochemical recurrence after radical perineal prostatectomy. Prostate Cancer Prostatic Dis 13:28–33
Brembilla G, Dell’Oglio P, Stabile A et al (2020) Interreader variability in prostate MRI reporting using Prostate Imaging Reporting and Data System version 2.1. Eur Radiol. https://doi.org/10.1007/s00330-019-06654-2
Kim T-H, Woo S, Han S et al (2020) The diagnostic performance of the length of tumor capsular contact on MRI for detecting prostate cancer extraprostatic extension: a systematic review and meta-analysis. Korean J Radiol 21(6):684–694
Lee S, Kim KB, Jo JK et al (2016) Prognostic value of focal positive surgical margins after radical prostatectomy. Clin Genitourin Cancer 14:e313–e319
Koskas Y, Lannes F, Branger N et al (2019) Extent of positive surgical margins following radical prostatectomy: impact on biochemical recurrence with long-term follow-up. BMC Urol 19:37
van Oort IM, Bruins HM, Kiemeney LA, Knipscheer BC, Witjes JA, Hulsbergen-van de Kaa CA (2010) The length of positive surgical margins correlates with biochemical recurrence after radical prostatectomy. Histopathology 56:464–471
Kates M, Sopko NA, Han M, Partin AW, Epstein JI (2016) Importance of reporting the Gleason score at the positive surgical margin site: analysis of 4,082 consecutive radical prostatectomy cases. J Urol 195:337–342
Lim CS, McInnes MD, Lim RS et al (2016) Prognostic value of Prostate Imaging and Data Reporting System (PI-RADS) v. 2 assessment categories 4 and 5 compared to histopathological outcomes after radical prostatectomy. J Magn Reson Imaging 46(1):257–266
Park SY, Oh YT, Jung DC et al (2016) Prediction of biochemical recurrence after radical prostatectomy with PI-RADS version 2 in prostate cancers: initial results. Eur Radiol 26:2502–2509
Acknowledgments
We thank Seo Young Park for her assistance with statistical analysis which improved the manuscript.
Funding
None
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Jeong Kon Kim, M.D., Ph.D.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
Seo Young Park, the statistician of Asan Medical Center, kindly provided statistical advice for this manuscript.
Informed consent
The requirement for written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• Retrospective
• Diagnostic performance study
• Performed at a single institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 194 kb)
Rights and permissions
About this article
Cite this article
Park, M.Y., Park, K.J., Kim, Mh. et al. Preoperative MRI-based estimation of risk for positive resection margin after radical prostatectomy in patients with prostate cancer: development and validation of a simple scoring system. Eur Radiol 31, 4898–4907 (2021). https://doi.org/10.1007/s00330-020-07569-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-07569-z