Abstract
Objectives
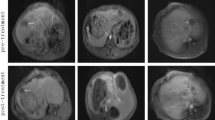
In this study, a treatment combining ethanol ablation (EA) and focused ultrasound (FUS) was performed to investigate its synergistic ablation effect on normal liver and VX2 liver tumours in rabbits.
Methods
A total of 59 healthy New Zealand white rabbits were included. For normal liver ablation, 39 animals were treated with FUS alone (n = 12), EA alone (n = 12), EA+FUS combination treatment (n = 12), or the control treatment (n = 3). The other 20 rabbits with implanted VX2 liver tumours were treated with EA alone (n = 10) or EA+FUS (n = 10). For FUS, the liver was exposed to 1 MHz FUS with an intensity of 33.0 W/cm2 (ISPTA) for 20 s. The EA group received an injection of absolute ethanol in the liver or liver tumours. For EA+FUS combination therapy, FUS was focused at the EA injection site, and both methods were carried out at the same time.
Results
In normal liver tissues, the ablated volume treated by FUS combined with EA (1.46 ± 0.30 cm3) was approximately 3 times larger than that of EA alone (0.51 ± 0.17 cm3); in VX2 liver tumours, the tumour necrosis rate of the combination therapy was 90.27%, which was much higher than that of EA treatment (63.55%).
Conclusion
The combination of EA and FUS could effectively increase the liver ablation volume and induce more complete tumour necrosis.
Key Points
• This study demonstrated a novel method for enhancing ethanol ablation and elucidated its potential to enhance percutaneous ethanol ablation (PEA) in a simple non-invasive way.
• Ethanol excited by focused ultrasound (FUS) exposure tended to accumulate at the injection site, which could prevent ethanol from being washed out by the bloodstream.
• The combination of EA and FUS could effectively increase the liver ablation volume and induce more complete tumour necrosis.






Similar content being viewed by others
Abbreviations
- 2-D:
-
Two-dimensional
- ALT:
-
Alanine aminotransferase
- ANOVA:
-
Analysis of variance
- AST:
-
Aspartate aminotransferase
- CEUS:
-
Contrast-enhanced US
- FUS:
-
Focused ultrasound
- H&E:
-
Haematoxylin and eosin
- HCC:
-
Hepatocellular carcinoma
- HIFU:
-
High-intensity focused ultrasound
- MEUS:
-
Microbubble-enhanced ultrasound
- MWA:
-
Microwave ablation
- PEA:
-
Percutaneous ethanol ablation
- RFA:
-
Radiofrequency ablation
References
Livraghi T, Festi D, Monti F, Salmi A, Vettori C (1986) US-guided percutaneous alcohol injection of small hepatic and abdominal tumors. Radiology 161:309–312
Ebara M, Okabe S, Kita K et al (2005) Percutaneous ethanol injection for small hepatocellular carcinoma: therapeutic efficacy based on 20-year observation. J Hepatol 43:458–464
Willatt JM, Francis IR, Novelli PM, Vellody R, Pandya A, Krishnamurthy VN (2012) Interventional therapies for hepatocellular carcinoma. Cancer Imaging 12:79–88
Yu J, Liang P, Yu XL et al (2015) Local tumour progression after ultrasound-guided microwave ablation of liver malignancies: risk factors analysis of 2529 tumours. Eur Radiol 25:1119–1126
Lencioni R, Llovet JM (2005) Percutaneous ethanol injection for hepatocellular carcinoma: alive or dead? J Hepatol 43:377–380
Zhang F, Wu G, Sun H et al (2014) Radiofrequency ablation of hepatocellular carcinoma in elderly patients fitting the Milan criteria: a single centre with 13 years experience. Int J Hyperthermia 30:471–479
Kirikoshi H, Saito S, Yoneda M et al (2009) Outcome of transarterial chemoembolization monotherapy, and in combination with percutaneous ethanol injection, or radiofrequency ablation therapy for hepatocellular carcinoma. Hepatol Res 39:553–562
Gao W, Qiao L, Gao Y et al (2016) Effect of microbubble-enhanced ultrasound on percutaneous ethanol ablation of rat walker-256 tumour. Eur Radiol 26:3017–3025
Wang S, Zhuang L, Meng Z (2013) Hepatocellular carcinoma more than 3 cm in diameter: a systematic review of transcatheter arterial chemoembolization plus percutaneous ethanol injection versus transcatheter arterial chemoembolization alone. ISRN Gastroenterol 2013:526024
Hoang NH, Murad HY, Ratnayaka SH, Chen C, Khismatullin DB (2014) Synergistic ablation of liver tissue and liver cancer cells with high-intensity focused ultrasound and ethanol. Ultrasound Med Biol 40:1869–1881
Oosterhof GO, Cornel EB, Smits GA, Debruyne FM, Schalken JA (1997) Influence of high-intensity focused ultrasound on the development of metastases. Eur Urol 32:91–95
Rabkin BA, Zderic V, Vaezy S (2005) Hyperecho in ultrasound images of HIFU therapy: involvement of cavitation. Ultrasound Med Biol 31:947–956
Zhou Y, Gao XW (2016) Effect of hydrodynamic cavitation in the tissue erosion by pulsed high-intensity focused ultrasound (pHIFU). Phys Med Biol 61:6651–6667
Coussios CC, Farny CH, Haar GT, Roy RA (2007) Role of acoustic cavitation in the delivery and monitoring of cancer treatment by high-intensity focused ultrasound (HIFU). Int J Hyperthermia 23:105–120
Carling U, Barkhatov L, Reims HM et al (2019) Can we ablate liver lesions close to large portal and hepatic veins with MR-guided HIFU? An experimental study in a porcine model. Eur Radiol 29:5013–5021
Napoli A, Anzidei M, Marincola BC et al (2013) MR imaging-guided focused ultrasound for treatment of bone metastasis. Radiographics 33:1555–1568
Phenix CP, Togtema M, Pichardo S, Zehbe I, Curiel L (2014) High intensity focused ultrasound technology, its scope and applications in therapy and drug delivery. J Pharm Pharm Sci 17:136–153
Mitsunobu M, Toyosaka A, Oriyama T, Okamoto E, Nakao N (1996) Intrahepatic metastases in hepatocellular carcinoma: the role of the portal vein as an efferent vessel. Clin Exp Metastasis 14:520–529
Francica G, Meloni MF, Riccardi L et al (2018) Ablation treatment of primary and secondary liver tumors under contrast-enhanced ultrasound guidance in field practice of interventional ultrasound centers. A multicenter study. Eur J Radiol 105:96–101
Chen S, Peng Z, Lin M et al (2018) Combined percutaneous radiofrequency ablation and ethanol injection versus hepatic resection for 2.1-5.0 cm solitary hepatocellular carcinoma: a retrospective comparative multicentre study. Eur Radiol 28:3651–3660
Funding
This study has received funding by the National Natural Science Foundation of China (Grant No. 81601511).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Dr. Liu Zheng.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Approval from the institutional animal care committee was obtained.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• Experimental
• Performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary Figure
Photographs of haematoxylin and eosin staining. The ablation zone was seen around the central vein area (black arrow), surrounded by normal liver tissue in the EA-treated liver (A). A visible boundary was present between the necrotic liver and live liver tissues (black arrows) with infiltration of the inflammatory cells into the periphery of the necrotic area 48 h after treatment (B). Necrotic tumour cells showed marked karyolysis and karyopyknosis with wider intercellular interspaces than live tumour cells (C). A visible boundary was present between the live tumour cells and normal liver tissues (black arrows) (D). (DOCX 900 kb)
Rights and permissions
About this article
Cite this article
Qiao, W., Yu, Y., Huang, Y. et al. Impact of focused ultrasound on the ethanol ablation of VX2 liver tumours in rabbits. Eur Radiol 30, 5862–5870 (2020). https://doi.org/10.1007/s00330-020-06941-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-06941-3