Abstract
Objectives
The purpose of this study was to investigate the association of relaxation-compensated chemical exchange saturation transfer (CEST) MRI with overall survival (OS) and progression-free survival (PFS) in newly diagnosed high-grade glioma (HGG) patients.
Methods
Twenty-six patients with newly diagnosed high-grade glioma (WHO grades III–IV) were included in this prospective IRB-approved study. CEST MRI was performed on a 7.0-T whole-body scanner. Association of patient OS/PFS with relaxation-compensated CEST MRI (amide proton transfer (APT), relayed nuclear Overhauser effect (rNOE)/NOE, downfield-rNOE-suppressed APT (dns-APT)) and diffusion-weighted imaging (apparent diffusion coefficient) were assessed using the univariate Cox proportional hazards regression model. Hazard ratios (HRs) and corresponding 95% confidence intervals were calculated. Furthermore, OS/PFS association with clinical parameters (age, gender, O6-methylguanine-DNA methyltransferase (MGMT) promotor methylation status, and therapy: biopsy + radio-chemotherapy vs. debulking surgery + radio-chemotherapy) were tested accordingly.
Results
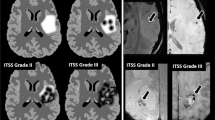
Relaxation-compensated APT MRI was significantly correlated with patient OS (HR = 3.15, p = 0.02) and PFS (HR = 1.83, p = 0.009). The strongest association with PFS was found for the dns-APT metric (HR = 2.61, p = 0.002). These results still stand for the relaxation-compensated APT contrasts in a homogenous subcohort of n = 22 glioblastoma patients with isocitrate dehydrogenase (IDH) wild-type status. Among the tested clinical parameters, patient age (HR = 1.1, p = 0.001) and therapy (HR = 3.68, p = 0.026) were significant for OS; age additionally for PFS (HR = 1.04, p = 0.048).
Conclusion
Relaxation-compensated APT MRI signal intensity is associated with overall survival and progression-free survival in newly diagnosed, previously untreated glioma patients and may, therefore, help to customize treatment and response monitoring in the future.
Key Points
• Amide proton transfer (APT) MRI signal intensity is associated with overall survival and progression in glioma patients.
• Relaxation compensation enhances the information value of APT MRI in tumors.
• Chemical exchange saturation transfer (CEST) MRI may serve as a non-invasive biomarker to predict prognosis and customize treatment.


Similar content being viewed by others
Abbreviations
- APT:
-
Amide proton transfer
- APTAREX :
-
APT contrast calculated with the AREX metric
- APTLD :
-
APT contrast calculated with the LD metric
- AREX:
-
Apparent exchange-dependent relaxation
- CEST:
-
Chemical exchange saturation transfer
- dns-APT:
-
Downfield relayed nuclear Overhauser effect suppressed APT
- FLAIR:
-
Fluid-attenuated inversion recovery
- FoV:
-
Field of view
- GBCA:
-
Gadolinium-based contrast agents
- GBM:
-
Glioblastoma multiforme
- gdce-T1:
-
T1-weighted gadolinium contrast-enhanced MRI
- GRE:
-
Gradient echo
- HGG:
-
High-grade glioma
- IDH:
-
Isocitrate dehydrogenase
- IQR:
-
Interquartile range
- KPS:
-
Karnofsky performance scale
- LD:
-
Lorentzian difference
- MGMT:
-
O6-Methylguanine-DNA methyltransferase
- MITK:
-
Medical Imaging Interaction Toolkit
- MPRAGE:
-
Magnetization-prepared rapid gradient echo
- MTRasym :
-
Magnetization transfer ratio asymmetry
- MTRLD :
-
Magnetization transfer Lorentzian difference
- NOE:
-
Nuclear Overhauser effect
- NOEAREX :
-
NOE contrast calculated with the AREX metric
- NOELD :
-
NOE contrast calculated with the LD metric
- OS:
-
Overall survival
- PFS:
-
Progression-free survival
- RANO:
-
Response assessment in neuro-oncology
- rCBV:
-
Relative cerebral blood volume
- RCT:
-
Radio-chemotherapy
- rNOE:
-
Relayed nuclear Overhauser effect
- T1-w:
-
T1-weighted
- T2-w:
-
T2-weighted
- TE:
-
Echo time
- TR:
-
Repetition time
- TSE:
-
Turbo spin echo
- WASABI:
-
Simultaneous mapping of water shift and B1
- WHO:
-
World Health Organization
- Zlab :
-
Label Z-spectrum
- Zref :
-
Reference Z-spectrum
References
Kim KB (2014) PFS as a surrogate for overall survival in metastatic melanoma. Lancet Oncol 15:246–248
Porter KR, McCarthy BJ, Freels S, Kim Y, Davis FG (2010) Prevalence estimates for primary brain tumors in the United States by age, gender, behavior, and histology. Neuro Oncol 12:520–527
Stupp R, Roila F (2009) Malignant glioma: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol 20(Suppl 4):126–128
Yan H, Parsons DW, Jin G et al (2009) IDH1 and IDH2 mutations in gliomas. N Engl J Med 360:765–773
Hegi ME, Diserens AC, Gorlia T et al (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352:997–1003
Wen PY, Kesari S (2008) Malignant gliomas in adults. N Engl J Med 359:492–507
Cerqua R, Balestrini S, Perozzi C et al (2016) Diagnostic delay and prognosis in primary central nervous system lymphoma compared with glioblastoma multiforme. Neurol Sci 37:23–29
Henson JW, Gaviani P, Gonzalez RG (2005) MRI in treatment of adult gliomas. Lancet Oncol 6:167–175
Ellingson BM, Chung C, Pope WB, Boxerman JL, Kaufmann TJ (2017) Pseudoprogression, radionecrosis, inflammation or true tumor progression? Challenges associated with glioblastoma response assessment in an evolving therapeutic landscape. J Neurooncol 134(3):495–504
Pope WB, Qiao XJ, Kim HJ et al (2012) Apparent diffusion coefficient histogram analysis stratifies progression-free and overall survival in patients with recurrent GBM treated with bevacizumab: a multi-center study. J Neurooncol 108:491–498
Oh J, Henry RG, Pirzkall A et al (2004) Survival analysis in patients with glioblastoma multiforme: predictive value of choline-to-n-acetylaspartate index, apparent diffusion coefficient, and relative cerebral blood volume. J Magn Reson Imaging 19:546–554
Ellingson BM, Cloughesy TF, Lai A et al (2011) Graded functional diffusion map–defined characteristics of apparent diffusion coefficients predict overall survival in recurrent glioblastoma treated with bevacizumab. Neuro Oncol 13:1151–1161
Higano S, Yun X, Kumabe T et al (2006) Malignant astrocytic tumors: clinical importance of apparent diffusion coefficient in prediction of grade and prognosis. Radiology 241:839–846
Law M, Young RJ, Babb JS et al (2008) Gliomas: predicting time to progression or survival with cerebral blood volume measurements at dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging. Radiology 247:490–498
Hamstra DA, Chenevert TL, Moffat BA et al (2005) Evaluation of the functional diffusion map as an early biomarker of time-to-progression and overall survival in high-grade glioma. Proc Natl Acad Sci U S A 102:16759–16764
Bonekamp D, Deike K, Wiestler B et al (2015) Association of overall survival in patients with newly diagnosed glioblastoma with contrast-enhanced perfusion MRI: comparison of intraindividually matched T1 - and T2 (*) -based bolus techniques. J Magn Reson Imaging 42:87–96
Burth S, Kickingereder P, Eidel O et al (2016) Clinical parameters outweigh diffusion- and perfusion-derived MRI parameters in predicting survival in newly diagnosed glioblastoma. Neuro Oncol 18:1673–1679
Wiestler B, Kluge A, Lukas M et al (2016) Multiparametric MRI-based differentiation of WHO grade II/III glioma and WHO grade IV glioblastoma. Sci Rep 6:35142
Kickingereder P, Götz M, Muschelli J et al (2016) Large-scale radiomic profiling of recurrent glioblastoma identifies an imaging predictor for stratifying anti-angiogenic treatment response. Clin Cancer Res 22:5765–5771
Lao J, Chen Y, Li ZC et al (2017) A deep learning-based radiomics model for prediction of survival in glioblastoma multiforme. Sci Rep 7:10353
Jones CK, Huang A, Xu J et al (2013) Nuclear Overhauser enhancement (NOE) imaging in the human brain at 7T. Neuroimage 77:114–124
Jin T, Wang P, Zong X, Kim SG (2013) MR imaging of the amide-proton transfer effect and the pH-insensitive nuclear overhauser effect at 9.4 T. Magn Reson Med 69:760–770
Zaiss M, Kunz P, Goerke S, Radbruch A, Bachert P (2013) MR imaging of protein folding in vitro employing nuclear-Overhauser-mediated saturation transfer. NMR Biomed 26:1815–1822
Goerke S, Zaiss M, Kunz P et al (2015) Signature of protein unfolding in chemical exchange saturation transfer imaging. NMR Biomed 28:906–913
Longo DL, Di Gregorio E, Abategiovanni R et al (2014) Chemical exchange saturation transfer (CEST): an efficient tool for detecting molecular information on proteins’ behaviour. Analyst 139:2687–2690
Zhou J, Payen JF, Wilson DA, Traystman RJ, van Zijl PC (2003) Using the amide proton signals of intracellular proteins and peptides to detect pH effects in MRI. Nat Med 9:1085–1090
Sun PZ, Benner T, Copen WA, Sorensen AG (2010) Early experience of translating pH-weighted MRI to image human subjects at 3 Tesla. Stroke 41:S147–S151
Zaiss M, Xu J, Goerke S et al (2014) Inverse Z-spectrum analysis for spillover-, MT-, and T1-corrected steady-state pulsed CEST-MRI--application to pH-weighted MRI of acute stroke. NMR Biomed 27:240–252
Zhou J, Lal B, Wilson DA, Laterra J, van Zijl PC (2003) Amide proton transfer (APT) contrast for imaging of brain tumors. Magn Reson Med 50:1120–1126
Zhou J, Blakeley JO, Hua J et al (2008) Practical data acquisition method for human brain tumor amide proton transfer (APT) imaging. Magn Reson Med 60:842–849
Togao O, Yoshiura T, Keupp J et al (2014) Amide proton transfer imaging of adult diffuse gliomas: correlation with histopathological grades. Neuro Oncol 16:441–448
Paech D, Windschuh J, Oberhollenzer J et al (2018) Assessing the predictability of IDH mutation and MGMT methylation status in glioma patients using relaxation-compensated multi-pool CEST MRI at 7.0 Tesla. Neuro Oncol 20(12):1661–1671
Zaiss M, Windschuh J, Paech D et al (2015) Relaxation-compensated CEST-MRI of the human brain at 7 T: unbiased insight into NOE and amide signal changes in human glioblastoma. Neuroimage 112:180–188
Louis DN, Perry A, Reifenberger G et al (2016) The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol 131:803–820
Regnery S, Adeberg S, Dreher C et al (2018) Chemical exchange saturation transfer MRI serves as predictor of early progression in glioblastoma patients. Oncotarget 9:28772–28783
Zaiss M, Windschuh J, Goerke S et al (2017) Downfield-NOE-suppressed amide-CEST-MRI at 7 Tesla provides a unique contrast in human glioblastoma. Magn Reson Med 77:196–208
Wen PY, Macdonald DR, Reardon DA et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28:1963–1972
Zaiss M, Zu Z, Xu J et al (2015) A combined analytical solution for chemical exchange saturation transfer and semi-solid magnetization transfer. NMR Biomed 28:217–230
Schuenke P, Windschuh J, Roeloffs V, Ladd ME, Bachert P, Zaiss M (2017) Simultaneous mapping of water shift and B1 (WASABI)—application to field-inhomogeneity correction of CESTMRI data. Magn Reson Med 77:571–580
Windschuh J, Zaiss M, Meissner JE et al (2015) Correction of B1-inhomogeneities for relaxation-compensated CEST imaging at 7 T. NMR Biomed 28:529–537
Nolden M, Zelzer S, Seitel A et al (2013) The Medical Imaging Interaction Toolkit: challenges and advances: 10 years of open-source development. Int J Comput Assist Radiol Surg 8:607–620
Shanshan J, Tianyu Z, Eberhart GC et al (2017) Predicting IDH mutation status in grade II gliomas using amide proton transfer-weighted (APTw) MRI. Magn Reson Med 78:1100–1109
Paech D, Zaiss M, Meissner JE et al (2014) Nuclear Overhauser enhancement mediated chemical exchange saturation transfer imaging at 7 tesla in glioblastoma patients. PLoS One 9:e104181
Desmond KL, Mehrabian H, Chavez S et al (2017) Chemical exchange saturation transfer for predicting response to stereotactic radiosurgery in human brain metastasis. Magn Reson Med 78:1110–1120
Paech D, Burth S, Windschuh J et al (2015) Nuclear Overhauser enhancement imaging of glioblastoma at 7 Tesla: region specific correlation with apparent diffusion coefficient and histology. PLoS One 10:e0121220
Choi YS, Ahn SS, Lee SK et al (2017) Amide proton transfer imaging to discriminate between low- and high-grade gliomas: added value to apparent diffusion coefficient and relative cerebral blood volume. Eur Radiol 27:3181–3189
Sakata A, Okada T, Yamamoto A et al (2015) Grading glial tumors with amide proton transfer MR imaging: different analytical approaches. J Neurooncol 122:339–348
Jiang S, Rui Q, Wang Y et al (2017) Discriminating MGMT promoter methylation status in patients with glioblastoma employing amide proton transfer-weighted MRI metrics. Eur Radiol 28(5):2115–2123
Heo H-Y, Zhang Y, Jiang S, Lee DH, Zhou J (2016) Quantitative assessment of amide proton transfer (APT) and nuclear overhauser enhancement (NOE) imaging with extrapolated semisolid magnetization transfer reference (EMR) signals: II. Comparison of three EMR models and application to human brain glioma at 3 Tesla. Magn Reson Med 75:1630–1639
Xu J, Yadav NN, Bar-Shir A et al (2014) Variable delay multi-pulse train for fast chemical exchange saturation transfer and relayed-nuclear overhauser enhancement MRI. Magn Reson Med 71:1798–1812
Acknowledgements
The authors would like to thank Prof. Dr. Annette Kopp-Schneider for her invaluable help with the statistical analyses and Joseph Weygand, M.S., for carefully proof reading and reviewing of the manuscript.
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Dr. Daniel Paech.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
Prof. Dr. Annette Kopp-Schneider (Division of Biostatistics, German Cancer Research Center, Heidelberg, Germany) kindly provided statistical advice for this manuscript.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Study subjects or cohorts overlap
The study cohort has previously been reported (Paech et al. Neuro Oncol, 2018, noy073 and Regnery et al. Oncotarget, 2018, 9:28772–28783) and a subcohort of eleven patients has been included in methodical publications (Zaiss et al. Neuroimage, 2015, 112:180–188 and Zaiss et al. MRM, 2017, 77(1):196–208). However, no investigations of overall survival and progression-free survival have previously been performed.
Methodology
• prospective
• diagnostic or prognostic
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 2813 kb)
Rights and permissions
About this article
Cite this article
Paech, D., Dreher, C., Regnery, S. et al. Relaxation-compensated amide proton transfer (APT) MRI signal intensity is associated with survival and progression in high-grade glioma patients. Eur Radiol 29, 4957–4967 (2019). https://doi.org/10.1007/s00330-019-06066-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-019-06066-2