Abstract
Objective
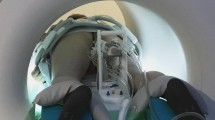
The aim of our study was (1) to establish an in-bore targeted biopsy of suspicious prostate lesions, avoiding bowel penetration using a transgluteal approach and (2) to assess operator setup, patient comfort and safety aspects in the clinical setting for freehand real-time MR-guidance established for percutaneous procedures in an open MR-scanner.
Material and methods
30 patients with suspect prostate lesions were biopsied in a cylindrical 3T-MRI system using a transgluteal approach in freehand technique. One to three biopsies were sampled using continuous dynamic imaging. Size, location and visibility of the lesion, intervention time, needle artefact size, interventional complications and histopathological diagnosis were recorded.
Results
All biopsies were technically successful. Nineteen patients showed evidence of prostate carcinoma. Cancer detection rate was 50 % in patients with previously negative TRUS-biopsy. The average intervention time was 26 min including a learning curve as the time was 13 min by the end of the study. No antibiotic prophylaxis was performed as none of the patients showed signs of infection.
Conclusions
MR-guided targeted freehand biopsies of prostate lesions using a transgluteal approach are both technically feasible and time efficient in a standard closed-bore 3T-MR scanner as well as safe for the individual patient.
Key Points
• Open-bore freehand interventional principles were adapted to closed-bore systems.
• Prostate MR-guided freehand biopsies were feasible in a clinical setting.
• A transgluteal approach provides a short and simplified work flow.
• An inoculation of the prostate with bowel flora is avoided.
• The intervention time is comparable to the stereotactic approach.






Similar content being viewed by others
References
Bastian PJ, Bellmunt J, Bolla M et al (2014) European Association of Urology. EAU guidelines on prostate cancer. part 1: screening, diagnosis, and local treatment with curative intent-update. Eur Urol 65:124–137
Walz J, Graefen M, Chun FK et al (2006) High incidence of prostate cancer detected by saturation biopsy after previous negative biopsy series. Eur Urol 50:498–505
Haas GP, Delongchamps NB, Jones RF et al (2007) Needle biopsies on autopsy prostates: sensitivity of cancer detection based on true prevalence. J Natl Cancer Inst 99:1484–1489
Fernandes ET, Sundaram CP, Long R et al (1997) Biopsy Gleason score: how does it correlate with the final pathological diagnosis in prostate cancer? Br J Urol 79:615–617
Chun FK, Steuber T, Erbersdobler A et al (2006) Development and internal validation of a nomogram predicting the probability of prostate cancer Gleason sum upgrading between biopsy and radical prostatectomy pathology. Eur Urol 49:820–826
Schoots IG, Roobol MJ, Nieboer D et al (2015) Magnetic resonance imaging-targeted biopsy may enhance the diagnostic accuracy of significant prostate cancer detection compared to standard transrectal ultrasound-guided biopsy: a systematic review and meta-analysis. Eur Urol 68:438–450
Mariotti GC, Falsarella PM, Garcia RG et al (2017) Incremental diagnostic value of targeted biopsy using mpMRI-TRUS fusion versus 14-fragments prostatic biopsy: a prospective controlled study. Eur Radiol. https://doi.org/10.1007/s00330-017-4939-0
Wegelin O, van Melick HH, Hooft L et al (2017) Comparing Three Different Techniques for Magnetic Resonance Imaging-targeted Prostate Biopsies: A Systematic Review of In-bore versus Magnetic Resonance Imaging-transrectal Ultrasound fusion versus Cognitive Registration. Is There a Preferred Technique? Eur Urol 71:517–531
Arsov C, Rabenalt R, Blondin D et al (2015) Prospective randomized trial comparing magnetic resonance imaging (MRI)-guided in-bore biopsy to MRI-ultrasound fusion and transrectal ultrasound-guided prostate biopsy in patients with prior negative biopsies. Eur Urol 68:713–720
Loeb S, van den Heuvel S, Zhu X et al (2012) Infectious complications and hospital admissions after prostate biopsy in a European randomized trial. Eur Urol 61:1110–1114
Fischbach F, Eggemann H, Bunke J et al (2012) MR-guided freehand biopsy of breast lesions in a 1.0-T open MR imager with a near-real-time interactive platform: preliminary experience. Radiology 265:359–370
Fischbach F, Porsch M, Krenzien F et al (2011) MR imaging guided percutaneous nephrostomy using a 1.0 Tesla open MR scanner. Cardiovasc Intervent Radiol 34:857–863
Fischbach F, Bunke J, Thormann M et al (2011) MR-guided freehand biopsy of liver lesions with fast continuous imaging using a 1.0-T open MRI scanner: experience in 50 patients. Cardiovasc Intervent Radiol 34:188–192
Moore CM, Kasivisvanathan V, Eggener S et al (2013) START Consortium. Standards of reporting for MRI-targeted biopsy studies (START) of the prostate: recommendations from an International Working Group. Eur Urol 64:544–552
Vargas HA, Hötker AM, Goldman DA et al (2016) Updated prostate imaging reporting and data system (PIRADS v2) recommendations for the detection of clinically significant prostate cancer using multiparametric MRI: critical evaluation using whole-mount pathology as standard of reference. Eur Radiol 26:1606–1612
Beyersdorff D, Winkel A, Hamm B et al (2005) MR imaging-guided prostate biopsy with a closed MR unit at 1.5 T: initial results. Radiology 234:576–581
Hambrock T, Fütterer JJ, Huisman HJ et al (2008) Thirty-two-channel coil 3T magnetic resonance-guided biopsies of prostate tumor suspicious regions identified on multimodality 3T magnetic resonance imaging: technique and feasibility. Invest Radiol 43:686–694
Zangos S, Melzer A, Eichler K et al (2011) MR-compatible assistance system for biopsy in a high-field-strength system: initial results in patients with suspicious prostate lesions. Radiology 259:903–910
Gupta S, Nguyen HL, Morello FA Jr et al (2004) Various approaches for CT-guided percutaneous biopsy of deep pelvic lesions: anatomic and technical considerations. Radiographics 24:175–189
Papanicolaou N, Eisenberg PJ, Silverman SG et al (1996) Prostatic biopsy after proctocolectomy: a transgluteal, CT-guided approach. Am J Roentgenol 166:1332–1334
Rodríguez LV, Terris MK (1998) Risks and complications of transrectal ultrasound guided prostate needle biopsy: a prospective study and review of the literature. J Urol 160:2115–2120
Valerio M, Ahmed HU, Emberton M et al (2014) The role of focal therapy in the management of localised prostate cancer: a systematic review. Eur Urol 66:732–751
Overduin CG, Jenniskens SF, Sedelaar JP, et al (2017) Percutaneous MR-guided focal cryoablation for recurrent prostate cancer following radiation therapy: retrospective analysis of iceball margins and outcomes. Eur Radiol 27:4828–36
Acknowledgments
This manuscript is dedicated to our friend and co-author Lukas Wien, who suddenly passed away. Lukas was an extraordinary and kind person and is missed sadly by all of us.
Funding
This study received funding by Philips Medical Healthcare.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Frank Fischbach, MD.
Conflict of interest
The authors of this manuscript declare relationships with the following companies:
Sascha Kruger and Bernhard Schnackenburg are employees of Philips Medical Healthcare.
Frank Fischbach, Katharina Fischbach and Jens Ricke received research grants from Philips Medical Healthcare.
The other authors declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• prospective
• performed at one institution
Rights and permissions
About this article
Cite this article
Fischbach, F., Wien, L., Krueger, S. et al. Feasibility study of MR-guided transgluteal targeted in-bore biopsy for suspicious lesions of the prostate at 3 Tesla using a freehand approach. Eur Radiol 28, 2690–2699 (2018). https://doi.org/10.1007/s00330-017-5187-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-5187-z