Abstract
Objectives
Assess liver stiffness using ultrasound point shear wave elastography (US P-SWE) in children before and after the Fontan operation.
Methods
Eighteen children undergoing the Fontan operation were prospectively enrolled. Eight US P-SWE measurements were obtained from the right hepatic lobe before surgery, and at multiple postoperative time points. Temporally related inferior vena cava pressure (IVC) data was collected from medical records, when available. Changes in mean liver shear wave speed (SWS) were assessed using a mixed-effect model with post hoc Tukey correction. Changes in IVC pressure were evaluated using the Wilcoxon signed-rank test. A p value less than 0.05 was considered significant.
Results
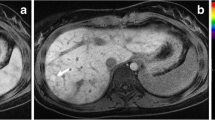
Mean age at enrolment was 33.5 ± 10.5 months. Baseline mean liver SWS was normal at 1.18 ± 0.29 m/s, increased to 2.28 ± 0.31 m/s at 2.5 ± 1.2 days (p < 0.0001) and to 2.22 ± 0.38 m/s at 7.5 ± 1.4 days (p < 0.0001). Five subjects returned at a mean of 185 ± 28 days, and mean liver SWS remained elevated at 2.08 ± 0.24 m/s (p < 0.0001). Mean IVC pressure increased from 7.2 ± 2.6 mmHg at baseline to 16.44 ± 3.3 mmHg at 2.2 ± 0.8 days post-surgery (p = 0.004).
Conclusion
The Fontan operation immediately and chronically increases liver stiffness and IVC pressure. Our study provides further evidence that congestion is a key driver of Fontan-associated liver disease.
Key points
• The Fontan operation triggers immediate hepatic congestion and marked liver stiffening.
• Congestion, not fibrosis, drives early increased liver stiffness in Fontan patients.
• Hepatic congestion persists chronically for months after the Fontan operation.
• Congestion confounds shear wave elastography as a post-Fontan liver fibrosis biomarker.






Similar content being viewed by others
Abbreviations
- FALD:
-
Fontan-associated liver disease
- IVC:
-
Inferior vena cava
- MR:
-
Magnetic resonance
- P-SWE:
-
Point shear wave elastography
- SWE:
-
Shear wave elastography
- SWS:
-
Shear wave speed
- US:
-
Ultrasound
References
de Leval MR, Deanfield JE (2010) Four decades of Fontan palliation. Nat Rev Cardiol 7:520–527
Khairy P, Fernandes SM, Mayer JE Jr et al (2008) Long-term survival, modes of death, and predictors of mortality in patients with Fontan surgery. Circulation 117:85–92
d’Udekem Y, Iyengar AJ, Cochrane AD et al (2007) The Fontan procedure: contemporary techniques have improved long-term outcomes. Circulation 116:I157–I164
Rychik J, Veldtman G, Rand E et al (2012) The precarious state of the liver after a Fontan operation: summary of a multidisciplinary symposium. Pediatr Cardiol 33:1001–1012
Greenway SC, Crossland DS, Hudson M et al (2015) Fontan-associated liver disease: implications for heart transplantation. J Heart Lung Transplant. doi:10.1016/j.healun.2015.10.015
Kendall TJ, Stedman B, Hacking N et al (2008) Hepatic fibrosis and cirrhosis in the Fontan circulation: a detailed morphological study. J Clin Pathol 61:504–508
Kiesewetter CH, Sheron N, Vettukattill JJ et al (2007) Hepatic changes in the failing Fontan circulation. Heart 93:579–584
Ghaferi AA, Hutchins GM (2005) Progression of liver pathology in patients undergoing the Fontan procedure: chronic passive congestion, cardiac cirrhosis, hepatic adenoma, and hepatocellular carcinoma. J Thorac Cardiovasc Surg 129:1348–1352
Schwartz MC, Sullivan LM, Glatz AC et al (2013) Portal and sinusoidal fibrosis are common on liver biopsy after Fontan surgery. Pediatr Cardiol 34:135–142
Kutty SS, Peng Q, Danford DA et al (2014) Increased hepatic stiffness as consequence of high hepatic afterload in the Fontan circulation: a vascular Doppler and elastography study. Hepatology 59:251–260
Wallihan DB, Podberesky DJ (2013) Hepatic pathology after Fontan palliation: spectrum of imaging findings. Pediatr Radiol 43:330–338
Asrani SK, Warnes CA, Kamath PS (2013) Hepatocellular carcinoma after the Fontan procedure. N Engl J Med 368:1756–1757
Saliba T, Dorkhom S, O’Reilly EM, Ludwig E, Gansukh B, Abou-Alfa GK (2010) Hepatocellular carcinoma in two patients with cardiac cirrhosis. Eur J Gastroenterol Hepatol 22:889–891
Elder RW, Parekh S, Book WM (2013) More on hepatocellular carcinoma after the Fontan procedure. N Engl J Med 369:490
Baek JS, Bae EJ, Ko JS et al (2010) Late hepatic complications after Fontan operation; non-invasive markers of hepatic fibrosis and risk factors. Heart 96:1750–1755
Ginde S, Hohenwalter MD, Foley WD et al (2012) Noninvasive assessment of liver fibrosis in adult patients following the Fontan procedure. Congenit Heart Dis 7:235–242
Shah H, Kuehl K, Sherker AH (2010) Liver disease after the Fontan procedure: what the hepatologist needs to know. J Clin Gastroenterol 44:428–431
Bulut OP, Romero R, Mahle WT et al (2013) Magnetic resonance imaging identifies unsuspected liver abnormalities in patients after the Fontan procedure. J Pediatr 163:201–206
Serai SD, Wallihan DB, Venkatesh SK et al (2014) Magnetic resonance elastography of the liver in patients status-post fontan procedure: feasibility and preliminary results. Congenit Heart Dis 9:7–14
Wallihan DB, Podberesky DJ, Marino BS, Sticka JS, Serai S (2014) Relationship of MR elastography determined liver stiffness with cardiac function after Fontan palliation. J Magn Reson Imaging 40:1328–1335
Deorsola L, Aidala E, Cascarano MT, Valori A, Agnoletti G, Napoleone CP (2016) Liver stiffness modifications shortly after total cavopulmonary connection. Interact Cardiovasc Thorac Surg. doi:10.1093/icvts/ivw186
Ferraioli G, Tinelli C, Dal Bello B et al (2012) Accuracy of real-time shear wave elastography for assessing liver fibrosis in chronic hepatitis C: a pilot study. Hepatology 56:2125–2133
Ferraioli G, Parekh P, Levitov AB, Filice C (2014) Shear wave elastography for evaluation of liver fibrosis. J Ultrasound Med 33:197–203
Castera L (2012) Noninvasive methods to assess liver disease in patients with hepatitis B or C. Gastroenterology 142:1293–1302 e1294
Dillman JR, Chen S, Davenport MS et al (2015) Superficial ultrasound shear wave speed measurements in soft and hard elasticity phantoms: repeatability and reproducibility using two ultrasound systems. Pediatr Radiol 45:376–385
Muller M, Gennisson JL, Deffieux T, Tanter M, Fink M (2009) Quantitative viscoelasticity mapping of human liver using supersonic shear imaging: preliminary in vivo feasibility study. Ultrasound Med Biol 35:219–229
Ferraioli G, Tinelli C, Zicchetti M et al (2012) Reproducibility of real-time shear wave elastography in the evaluation of liver elasticity. Eur J Radiol 81:3102–3106
Woo H, Lee JY, Yoon JH, Kim W, Cho B, Choi BI (2015) Comparison of the reliability of acoustic radiation force impulse imaging and supersonic shear imaging in measurement of liver stiffness. Radiology 277:881–886
Trout AT, Serai S, Mahley AD et al (2016) Liver stiffness measurements with MR elastography: agreement and repeatability across imaging systems, field strengths, and pulse sequences. Radiology. doi:10.1148/radiol.2016160209:160209
Eiler J, Kleinholdermann U, Albers D et al (2012) Standard value of ultrasound elastography using acoustic radiation force impulse imaging (ARFI) in healthy liver tissue of children and adolescents. Ultraschall Med 33:474–479
Hanquinet S, Courvoisier D, Kanavaki A, Dhouib A, Anooshiravani M (2013) Acoustic radiation force impulse imaging-normal values of liver stiffness in healthy children. Pediatr Radiol 43:539–544
Lee MJ, Kim MJ, Han KH, Yoon CS (2013) Age-related changes in liver, kidney, and spleen stiffness in healthy children measured with acoustic radiation force impulse imaging. Eur J Radiol 82:e290–e294
Matos H, Trindade A, Noruegas MJ (2014) Acoustic radiation force impulse imaging in paediatric patients: normal liver values. J Pediatr Gastroenterol Nutr 59:684–688
Schwartz MC, Sullivan L, Cohen MS et al (2012) Hepatic pathology may develop before the Fontan operation in children with functional single ventricle: an autopsy study. J Thorac Cardiovasc Surg 143:904–909
Barr RG, Ferraioli G, Palmeri ML et al (2015) Elastography assessment of liver fibrosis: Society of Radiologists in Ultrasound Consensus Conference Statement. Radiology 276:845–861
Dillman JR, Heider A, Bilhartz JL et al (2015) Ultrasound shear wave speed measurements correlate with liver fibrosis in children. Pediatr Radiol 45:1480–1488
Acknowledgments
The scientific guarantor of this publication is Frank W. DiPaola, MD. The authors of this manuscript declare relationships with the following companies: Dr. Dillman discloses that he receives unrelated, unrestricted, investigator-initiated research funding from Siemens Medical Solutions USA, Inc. The authors have no other conflicts of interest to disclose. This research was funded by a pilot grant provided by the Michigan Institute for Clinical and Health Research (MICHR), University of Michigan, Ann Arbor, MI (CTSA 2UL1TR000433).
Aishwarya Parameswaran, MS, of the Michigan Institute for Clinical and Health Research (MICHR) and one of the authors, has significant statistical expertise and kindly provided statistical advice for this manuscript. Institutional review board approval was obtained. Written informed consent was obtained from all subjects (patients) in this study. Methodology: prospective, cohort study, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
Diagrams of cardiac anatomy (using the example of hypoplastic left heart syndrome in which the right ventricle is the single functional ventricle) and direction of hepatic venous return before and after the Fontan operation. a Cardiac anatomy prior to the Fontan operation demonstrating that hepatic venous return (via the inferior vena cava) is routed through the right atrium and to the right ventricle and is not exposed to the pressure within the pulmonary circulation (PA), since this pathway is blocked with a surgically placed patch. There is lower pressure within the inferior vena cava and normal liver stiffness. b Cardiac anatomy following the Fontan operation, in which a surgically placed baffle now routes hepatic venous return directly to the pulmonary circulation. The inferior vena cava and liver are now exposed to the pressure within the pulmonary circulation resulting in higher pressure within the inferior vena cava and increased liver stiffness. IVC inferior vena cava, Ao aorta, LA left atrium, PA pulmonary artery, RA right atrium, RV right ventricle (GIF 61 kb)
(GIF 62 kb)
Supplementary Fig. 2
Pressure curve demonstrating changes in blood pressure across the circulation in a patient before and after the Fontan operation. Blood pressure increases dramatically within the right ventricle (RV) as blood is ejected into the aorta (Ao), and then blood pressure falls as blood traverses the systemic circulation. In the pre-Fontan circulation, blood pressure falls to comparatively lower values within the inferior vena cava (IVC) and right atrium (RA) because these structures are not in direct communication with the pressure within the pulmonary circulation (P). In contrast, in the post-Fontan circulation, there is an elevated pressure within the inferior vena cava and right atrium (which results in increased liver stiffness) because these structures communicate directly with the pulmonary circulation. Blue deoxygenated blood, purple partially oxygenated blood, red fully oxygenated blood. (Figure elements adapted from Gewillig M and Goldberg DJ. Failure of the Fontan circulation. Heart Fail Clin. 2014;10(1):105-116) (GIF 29 kb)
Rights and permissions
About this article
Cite this article
DiPaola, F.W., Schumacher, K.R., Goldberg, C.S. et al. Effect of Fontan operation on liver stiffness in children with single ventricle physiology. Eur Radiol 27, 2434–2442 (2017). https://doi.org/10.1007/s00330-016-4614-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-016-4614-x