Abstract
Objectives
To evaluate the efficacy and safety of percutaneous laser ablation (PLA) for unifocal T1N0M0 papillary thyroid microcarcinoma(PTMC).
Methods
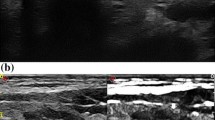
A retrospective study was conducted in 30 patients with single PTMC which was diagnosed by fine-needle aspiration biopsy (FNAB). PLA was performed for proper time to achieve adequate ablation zones. The extent of ablation was assessed by contrast-enhanced ultrasound (CEUS) immediately and 7 days after PLA. Complications were recorded. The size and volume of the ablation area and thyroid hormones were measured, and clinical evaluations were performed at 1, 3, 6 and12 months, and every half year thereafter.
Results
The procedure was well tolerated without serious complications. Of the 30 patients, 29 were treated successfully under local anaesthesia in a single session. Only one incomplete ablation was detected by immediate CEUS after PLA, and a second ablation was performed. At the last follow-up, ten (33.3 %) ablation zones had disappeared, and 20 (66.67 %) ablation zones remained as scar-like lesions. No regrowth of treated tumours, local recurrence or distant metastases were detected.
Conclusions
During the short-term follow-up period, ultrasound-guided PLA appears to be effective and safe for treating solitary T1N0M0 PTMC in selected patients who are ineligible for surgery.
Key Points
• Ultrasound is a useful tool in percutaneous laser ablation (PLA).
• PLA is safe for treating papillary thyroid microcarcinoma.
• PLA is effective for single papillary thyroid microcarcinoma.





Similar content being viewed by others
Abbreviations
- CEUS:
-
Contrast-enhanced ultrasound
- FNAB:
-
Fine needle aspiration biopsy
- PLA:
-
Percutaneous laser ablation
- PTMC:
-
Papillary thyroid microcarcinoma
References
Cooper DS, Doherty GM, Haugen BR et al (2009) Revised American thyroid association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 19:1167–1214
Bernet V (2010) Approach to the patient with incidental papillary microcarcinoma. J Clin Endocrinol Metab 95:3586–3592
Cramer JD, Fu P, Harth KC et al (2010) Analysis of the rising incidence of thyroid cancer using the surveillance, epidemiology and end results national cancer data registry. Surgery 148:1147–1153
Chen AY, Jemal A, Ward EM (2009) Increasing incidence of differentiated thyroid cancer in the United States, 1988-2005. Cancer 115:3801–3807
Ito Y, Miyauchi A, Inoue H et al (2010) An observational trial for papillary thyroid microcarcinoma in Japanese patients. World J Surg 34:28–35
Yu XM, Wan Y, Sippel RS et al (2011) Should all papillary thyroid microcarcinomas be aggressively treated? An analysis of 18,445 cases. Ann Surg 254:653–660
Ross DS, Litofsky D, Ain KB et al (2009) Recurrence after treatment of micropapillary thyroid cancer. Thyroid 19:1043–1048
Muntean V, Domsa I, Zolog A et al (2013) Incidental papillary thyroid microcarcinoma: is completion surgery required? Chirurgia (Bucur) 108:490–497
Yue W, Wang S, Yu S et al (2014) Ultrasound-guided percutaneous microwave ablation of solitary T1N0M0 papillary thyroid microcarcinoma: initial experience. Int J Hyperthermia 30:150–157
Valcavi R, Riganti F, Bertani A et al (2010) Percutaneous laser ablation of cold benign thyroid nodules: a 3-year follow-up study in 122 patients. Thyroid 20:1253–1261
Dossing H, Bennedbaek FN, Hegedus L (2005) Effect of ultrasound-guided interstitial laser photocoagulation on benign solitary solid cold thyroid nodules-a randomised study. Eur J Endocrinol 152:341–345
Pacella CM, Bizzarri G, Spiezia S et al (2004) Thyroid tissue: US-guided percutaneous laser thermal ablation. Radiology 232:272–280
Papini E, Guglielmi R, Bizzarri G et al (2007) Treatment of benign cold thyroid nodules: a randomized clinical trial of percutaneous laser ablation versus levothyroxine therapy or follow-up. Thyroid 17:229–235
Papini E, Guglielmi R, Gharib H et al (2011) Ultrasound-guided laser ablation of incidental papillary thyroid microcarcinoma: a potential therapeutic approach in patients at surgical risk. Thyroid 21:917–920
Valcavi R, Piana S, Bortolan GS et al (2013) Ultrasound-guided percutaneous laser ablation of papillary thyroid microcarcinoma: a feasibility study on three cases with pathological and immunohistochemical evaluation. Thyroid 23:1578–1582
Greene FL, Sobin LH (2009) A worldwide approach to the TNM staging system: collaborative efforts of the AJCC and UICC. J Surg Oncol 99:269–272
Hoang JK, Lee WK, Lee M et al (2007) US Features of thyroid malignancy: pearls and pitfalls. Radiographics 27:847–860, discussion 861-865
Spiezia S, Vitale G, Di Somma C et al (2003) Ultrasound-guided laser thermal ablation in the treatment of autonomous hyperfunctioning thyroid nodules and compressive nontoxic nodular goiter. Thyroid 13:941–947
Pellegriti G, Scollo C, Lumera G et al (2004) Clinical behavior and outcome of papillary thyroid cancers smaller than 1.5cm in diameter: study of 299 cases. J Clin Endocrinol Metab 89:3713–3720
Baek JH, Moon WJ, Kim YS et al (2009) Radiofrequency ablation for the treatment of autonomously functioning thyroid nodules. World J Surg 33:1971–1977
Kim HY, Ryu WS, Woo SU et al (2010) Primary papillary thyroid carcinoma previously treated incompletely with radiofrequency ablation. J Cancer Res Ther 6:310–312
Papini E, Bizzarri G, Bianchini A et al (2013) Percutaneous ultrasound-guided laser ablation is effective for treating selected nodal metastases in papillary thyroid cancer. J Clin Endocrinol Metab 98:E92–E97
Monchik JM, Donatini G, Iannuccilli J et al (2006) Radiofrequency ablation and percutaneous ethanol injection treatment for recurrent local and distant well-differentiated thyroid carcinoma. Ann Surg 244:296–304
Gambelunghe G, Fede R, Bini V et al (2013) Ultrasound-guided interstitial laser ablation for thyroid nodules is effective only at high total amounts of energy: results from a three-year pilot study. Surgery 20:345–350
Acknowledgments
The scientific guarantor of this publication is Zhan. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article. This study has received funding by the national clinical key specialty construction projects of China and Science and Technology Commission Foundation of Shanghai. No complex statistical methods were necessary for this paper. Institutional Review Board approval was obtained. Written informed consent was obtained from all subjects (patients) in this study. Our study subjects or cohorts have not been previously reported. Methodology: retrospective, diagnostic or prognostic study, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhou, W., Jiang, S., Zhan, W. et al. Ultrasound-guided percutaneous laser ablation of unifocal T1N0M0 papillary thyroid microcarcinoma: Preliminary results. Eur Radiol 27, 2934–2940 (2017). https://doi.org/10.1007/s00330-016-4610-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-016-4610-1