Abstract
Objectives
Evaluation of the technical success, patient safety and technical effectiveness of magnetic resonance (MR)-guided microwave ablation of hepatic malignancies.
Methods
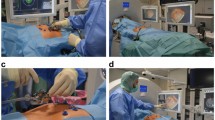
Institutional review board approval and informed patient consent were obtained. Fifteen patients (59.8 years ± 9.5) with 18 hepatic malignancies (7 hepatocellular carcinomas, 11 metastases) underwent MR-guided microwave ablation using a 1.5-T MR system. Mean tumour size was 15.4 mm ± 7.7 (7-37 mm). Technical success and ablation zone diameters were assessed by post-ablative MR imaging. Technique effectiveness was assessed after 1 month. Complications were classified according to the Common Terminology Criteria for Adverse Events (CTCAE). Mean follow-up was 5.8 months ± 2.6 (1-10 months).
Results
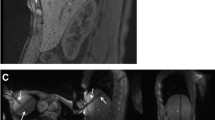
Technical success and technique effectiveness were achieved in all lesions. Lesions were treated using 2.5 ± 1.2 applicator positions. Mean energy and ablation duration per tumour were 37.6 kJ ± 21.7 (9-87 kJ) and 24.7 min ± 11.1 (7-49 min), respectively. Coagulation zone short- and long-axis diameters were 31.5 mm ± 10.5 (16-65 mm) and 52.7 mm ± 15.4 (27-94 mm), respectively. Two CTCAE-2-complications occurred (pneumothorax, pleural effusion). Seven patients developed new tumour manifestations in the untreated liver. Local tumour progression was not observed.
Conclusions
Microwave ablation is feasible under near real-time MR guidance and provides effective treatment of hepatic malignancies in one session.
Key Points
• Planning, applicator placement and therapy monitoring are possible without using contrast enhancement
• Energy transmission from the generator to the scanner room is safely possible
• MR-guided microwave ablation provides effective treatment of hepatic malignancies in one session
• Therapy monitoring is possible without applicator retraction from the ablation site





Similar content being viewed by others
References
Solbiati L, Ahmed M, Cova L, Ierace T, Brioschi M, Goldberg SN (2012) Small liver colorectal metastases treated with percutaneous radiofrequency ablation: local response rate and long-term survival with up to 10-year follow-up. Radiology 265:958–968
Gillams A, Goldberg N, Ahmed M et al (2015) Thermal ablation of colorectal liver metastases: a position paper by an international panel of ablation experts, the interventional oncology sans frontieres meeting 2013. Eur Radiol 25:3438–3454
Frericks BB, Ritz JP, Albrecht T et al (2008) Influence of intrahepatic vessels on volume and shape of percutaneous thermal ablation zones: in vivo evaluation in a porcine model. Invest Radiol 43:211–218
Lee HY, Rhim H, Lee MW et al (2013) Early diffuse recurrence of hepatocellular carcinoma after percutaneous radiofrequency ablation: analysis of risk factors. Eur Radiol 23:190–197
Fan W, Li X, Zhang L, Jiang H, Zhang J (2012) Comparison of microwave ablation and multipolar radiofrequency ablation in vivo using two internally cooled probes. AJR Am J Roentgenol 198:W46–W50
Laeseke PF, Lee FT Jr, Sampson LA, van der Weide DW, Brace CL (2009) Microwave ablation versus radiofrequency ablation in the kidney: high-power triaxial antennas create larger ablation zones than similarly sized internally cooled electrodes. J Vasc Interv Radiol 20:1224–1229
Alexander ES, Wolf FJ, Machan JT et al (2015) Microwave ablation of focal hepatic malignancies regardless of size: a 9-year retrospective study of 64 patients. Eur J Radiol 84:1083–1090
Andreano A, Brace CL (2013) A comparison of direct heating during radiofrequency and microwave ablation in ex vivo liver. Cardiovasc Intervent Radiol 36:505–511
Huang S, Yu J, Liang P et al (2014) Percutaneous microwave ablation for hepatocellular carcinoma adjacent to large vessels: a long-term follow-up. Eur J Radiol 83:552–558
Chinnaratha MA, Chuang MA, Fraser RJ, Woodman RJ, Wigg AJ (2016) Percutaneous thermal ablation for primary hepatocellular carcinoma: a systematic review and meta-analysis. J Gastroenterol Hepatol 31:294–301
Leyendecker JR, Dodd GD 3rd, Halff GA et al (2002) Sonographically observed echogenic response during intraoperative radiofrequency ablation of cirrhotic livers: pathologic correlation. AJR Am J Roentgenol 178:1147–1151
Yu J, Liang P, Yu XL et al (2015) Local tumour progression after ultrasound-guided microwave ablation of liver malignancies: risk factors analysis of 2529 tumours. Eur Radiol 25:1119–1126
Park MH, Rhim H, Kim YS, Choi D, Lim HK, Lee WJ (2008) Spectrum of CT findings after radiofrequency ablation of hepatic tumors. Radiographics 28:379–390
Rempp H, Loh H, Hoffmann R et al (2014) Liver lesion conspicuity during real-time MR-guided radiofrequency applicator placement using spoiled gradient echo and balanced steady-state free precession imaging. J Magn Reson Imaging 40:432–439
Anzidei M, Napoli A, Sandolo F et al (2014) Magnetic resonance-guided focused ultrasound ablation in abdominal moving organs: a feasibility study in selected cases of pancreatic and liver cancer. Cardiovasc Intervent Radiol 37:1611–1617
Hoffmann R, Thomas C, Rempp H et al (2012) Performing MR-guided biopsies in clinical routine: factors that influence accuracy and procedure time. Eur Radiol 22:663–671
Maurer MH, Schreiter N, de Bucourt M et al (2013) Cost comparison of nerve root infiltration of the lumbar spine under MRI and CT guidance. Eur Radiol 23:1487–1494
Kierans AS, Elazzazi M, Braga L et al (2010) Thermoablative treatments for malignant liver lesions: 10-year experience of MRI appearances of treatment response. AJR Am J Roentgenol 194:523–529
Ahmed M, Solbiati L, Brace CL et al (2014) Image-guided tumor ablation: standardization of terminology and reporting criteria—a 10-year update. Radiology 273:241–260
Institute NC (June 14, 2010) Common Terminology Criteria for Adverse Events (CTCAE). Version 4.03. Available at: http://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference_5x7.pdf. Accessed 16 January 2016
Rempp H, Waibel L, Hoffmann R, Claussen CD, Pereira PL, Clasen S (2012) MR-guided radiofrequency ablation using a wide-bore 1.5-T MR system: clinical results of 213 treated liver lesions. Eur Radiol 22:1972–1982
Mindjuk I, Trumm CG, Herzog P, Stahl R, Matzko M (2015) MRI predictors of clinical success in MR-guided focused ultrasound (MRgFUS) treatments of uterine fibroids: results from a single centre. Eur Radiol 25:1317–1328
Ringe KI, Lutat C, Rieder C, Schenk A, Wacker F, Raatschen HJ (2015) Experimental evaluation of the heat sink effect in hepatic microwave ablation. PLoS One 10:e0134301
Qian GJ, Wang N, Shen Q et al (2012) Efficacy of microwave versus radiofrequency ablation for treatment of small hepatocellular carcinoma: experimental and clinical studies. Eur Radiol 22:1983–1990
Clasen S, Rempp H, Hoffmann R, Graf H, Pereira PL, Claussen CD (2014) Image-guided radiofrequency ablation of hepatocellular carcinoma (HCC): is MR guidance more effective than CT guidance? Eur J Radiol 83:111–116
Kim YS, Rhim H, Lim HK, Choi D, Lee MW, Park MJ (2011) Coagulation necrosis induced by radiofrequency ablation in the liver: histopathologic and radiologic review of usual to extremely rare changes. Radiographics 31:377–390
Holzapfel K, Reiser-Erkan C, Fingerle AA et al (2011) Comparison of diffusion-weighted MR imaging and multidetector-row CT in the detection of liver metastases in patients operated for pancreatic cancer. Abdom Imaging 36:179–184
Kim HS, Choi D, Kim SH et al (2013) Changes in the signal- and contrast-to-noise ratios of hepatocellular carcinomas on gadoxetic acid-enhanced dynamic MR imaging. Eur J Radiol 82:62–68
Fischbach F, Lohfink K, Gaffke G et al (2013) Magnetic resonance-guided freehand radiofrequency ablation of malignant liver lesions: a new simplified and time-efficient approach using an interactive open magnetic resonance scan platform and hepatocyte-specific contrast agent. Invest Radiol 48:422–428
Rempp H, Clasen S, Pereira PL (2012) Image-based monitoring of magnetic resonance-guided thermoablative therapies for liver tumors. Cardiovasc Intervent Radiol 35:1281–1294
Aube C, Schmidt D, Brieger J et al (2004) Magnetic resonance imaging characteristics of six radiofrequency electrodes in a phantom study. J Vasc Interv Radiol 15:385–392
Brace CL, Diaz TA, Hinshaw JL, Lee FT Jr (2010) Tissue contraction caused by radiofrequency and microwave ablation: a laboratory study in liver and lung. J Vasc Interv Radiol 21:1280–1286
Acknowledgments
Parts of this manuscript were presented at ECR 2016. The scientific guarantor of this publication is Prof. Stephan Clasen. The authors of this manuscript declare relationships with the following companies: Prof. Philippe L Pereira: consultant for Angiodynamics, Terumo, Covidien, BSD. All other authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article. The authors state that this work has not received any funding. No complex statistical methods were necessary for this paper. Institutional Review Board approval was obtained. Written informed consent was obtained from all subjects (patients) in this study. Approval from the institutional animal care committee was not required. No study subjects or cohorts have been previously reported. Methodology: prospective, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hoffmann, R., Rempp, H., Keßler, DE. et al. MR-guided microwave ablation in hepatic tumours: initial results in clinical routine. Eur Radiol 27, 1467–1476 (2017). https://doi.org/10.1007/s00330-016-4517-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-016-4517-x