Abstract
Objective
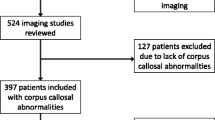
To describe the MRI and structural features of a peculiar malformation of the corpus callosum (CC) in a group of young patients with intellectual disability.
Methods
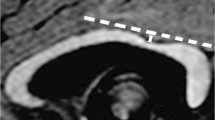
We studied with conventional MRI and DTI a group of subjects showing an aberrant supracallosal bundle, characterized by the presence of a triangle-shaped bulging above the dorsal surface of CC on the midline. Clinical evaluations, CGH-array and instrumental analysis were also collected.
Results
Among 85 patients with malformed CC, we identified 15 subjects that showed the supracallosal bundle. The CC was thickened in five cases, long and thinned in three cases, short and thinned in three cases and it had a “ribbon-like” appearance in four subjects. Additional brain anomalies were present in eight cases. DTI colour maps and tractography showed that the bundle had an antero-posterior longitudinal orientation and that the tract bifurcated posteriorly, ending in the posterior hippocampi. Patients had different combinations of neurological symptoms, but all showed mild or severe intellectual disability.
Conclusions
Combining radiological and genetic data with embryological knowledge of the development of cerebral commissures, we hypothesize that the supracallosal bundle represents a vestigial structure, the dorsal fornix, present during fetal life. Its persistence is associated with intellectual disability.
Key Points
• An aberrant longitudinal bundle can be detected above corpus callosum.
• The presence of the supracallosal bundle is associated with intellectual disability.
• The supracallosal bundle may represent a persistent dorsal fornix.





Similar content being viewed by others
Abbreviations
- CC:
-
Corpus callosum
- DTI:
-
Diffusion tensor imaging
- VF:
-
Volume fraction
- FA:
-
Fractional anisotropy
- AD:
-
Axial diffusivity
- RD:
-
Radial diffusivity
- MD:
-
Mean diffusivity
- ROI:
-
Region of interest
- IG:
-
Indusium griseum
References
Edwards TJ, Sherr EH, Barkovich AJ, Richards LJ (2014) Clinical, genetic and imaging findings identify new causes for corpus callosum development syndromes. Brain 137:1579–1613
Rakic P, Yakovlev PI (1968) Development of the corpus callosum and cavum septi in man. J Comp Neurol 132:45–72
Raybaud C (2010) The corpus callosum, the other great forebrain commissures, and the septum pellucidum: anatomy, development, and malformation. Neuroradiology 52:447–477
Rollins NK (2013) Diffusion imaging of the congenitally thickened corpus callosum. AJNR Am J Neuroradiol 34:660–665
Friede RL, Briner J (1978) Midline hyperplasia with malformation of the fornical system. Neurology 28:1302–1305
Malobabic S, Puskas L, Vela A (1988) Enlarged medial longitudinal (supracallosal) stria in one human brain. Acta Biol Med Exp 13:87–90
Reinhold S, Laas R (1989) Longitudinal supracallosal bundles without callosal defect. Clin Neuropathol 8:246–247
Sy J, Alturkustani M, Ang LC (2010) Intracallosal longitudinal fiber bundle: an unexpected finding mimicking demyelination in a patient with turner syndrome. Acta Neuropathol 120:545–547
Hori A, Stan AC (2004) Supracallosal longitudinal fiber bundle: heterotopic cingulum, dorsal fornix or probst bundle? Neuropathology 24:56–59
Poretti A, Meoded A, Rossi A, Raybaud C, Huisman TA (2013) Diffusion tensor imaging and fiber tractography in brain malformations. Pediatr Radiol 43:28–54
Garel C, Cont I, Alberti C, Josserand E, Moutard ML, Ducou le Pointe H (2011) Biometry of the corpus callosum in children: MR imaging reference data. AJNR Am J Neuroradiol 32:1436–1443
Wahl M, Strominger Z, Jeremy RJ et al (2009) Variability of homotopic and heterotopic callosal connectivity in partial agenesis of the corpus callosum: a 3T diffusion tensor imaging and Q-ball tractography study. AJNR Am J Neuroradiol 30:282–289
Koob M, Weingertner AS, Gasser B, Oubel E, Dietemann JL (2012) Thick corpus callosum: a clue to the diagnosis of fetal septopreoptic holoprosencephaly? Pediatr Radiol 42:886–890
Lerman-Sagie T, Ben-Sira L, Achiron R et al (2009) Thick fetal corpus callosum: an ominous sign? Ultrasound Obstet Gynecol 34:55–61
Rypens F, Sonigo P, Aubry MC, Delezoide AL, Cessot F, Brunelle F (1996) Prenatal MR diagnosis of a thick corpus callosum. AJNR Am J Neuroradiol 17:1918–1920
Vachha B, Adams RC, Rollins NK (2006) Limbic tract anomalies in pediatric myelomeningocele and chiari II malformation: anatomic correlations with memory and learning--initial investigation. Radiology 240:194–202
Fish PA (1893) The indusium of the callosum. J Comp Neurol 3:61–68
Smith GE (1897) The morphology of the indusium and striae lancisii. Anat Anz 13:23–27
Pavlovic S, Stefanovic N, Malobabic S, Babic Z, Kostic A, Pavlovic M (2009) Longitudinal striae of the human fornix: shape, relations and variations. Surg Radiol Anat 31:501–506
Tubbs RS, Prekupec M, Loukas M, Hattab EM, Cohen-Gadol AA (2013) The indusium griseum: anatomic study with potential application to callosotomy. Neurosurgery 73:312–316
Ren T, Anderson A, Shen WB et al (2006) Imaging, anatomical, and molecular analysis of callosal formation in the developing human fetal brain. Anat Rec A: Discov Mol Cell Evol Biol 288:191–204
Shu T, Richards LJ (2001) Cortical axon guidance by the glial wedge during the development of the corpus callosum. J Neurosci 21:2749–2758
Nakada T (1999) High-field, high-resolution MR imaging of the human indusium griseum. AJNR Am J Neuroradiol 20:524–525
Di Ieva A, Tschabitscher M, Rodriguez y Baena R (2007) Lancisi's nerves and the seat of the soul. Neurosurgery 60:563–568
Di Ieva A, Fathalla H, Cusimano MD, Tschabitscher M (2015) The indusium griseum and the longitudinal striae of the corpus callosum. Cortex 62:34–40
Sidman RL, Rakic P (1982) Development of the human central nervous system. In: Haymaker W, Adams RD (eds). Histology and histopathology of the nervous system. Thomas, Springfield, Ill, pp 3-145
Smith GE (1897) The relation of the fornix to the margin of the cerebral cortex. J Anat Physiol 32:23–58
Kier EL, Fulbright RK, Bronen RA (1995) Limbic lobe embryology and anatomy: dissection and MR of the medial surface of the fetal cerebral hemisphere. AJNR Am J Neuroradiol 16:1847–1853
Bayer S, Altman J (2007) Atlas of the human central nervous system development. CRC press
Li LX, Zhou WB, Tao Z et al (2010) Effect of FAM172A protein on apoptosis and proliferation in HEK293 cells. Zhonghua Yi Xue Za Zhi 90:2424–2427
Olah J, Vincze O, Virok D et al (2011) Interactions of pathological hallmark proteins: tubulin polymerization promoting protein/p25, beta-amyloid, and alpha-synuclein. J Biol Chem 286:34088–34100
Suzuki N, Ando S, Sumida K, Horie N, Saito K (2011) Analysis of altered gene expression specific to embryotoxic chemical treatment during embryonic stem cell differentiation into myocardiac and neural cells. J Toxicol Sci 36:569–585
Fan Y, Chen J, Ye J, Yan H, Cai Y (2014) Proteinase-activated receptor 2 modulates corticotropin releasing hormone-induced brain-derived neurotrophic factor release from microglial cells. Cell Biol Int 38:92–96
Lee SL, Thomas P, Fenech M (2014) Extracellular amyloid beta 42 causes necrosis, inhibition of nuclear division, and mitotic disruption under both folate deficient and folate replete conditions as measured by the cytokinesis-block micronucleus cytome assay. Environ Mol Mutagen 55:1–14
Sun P, Li X, Chen C et al (2014) Activating transcription factor 4 modulates BDNF release from microglial cells. J Mol Neurosci 52:225–230
Zhang F, Yao SY, Whetsell WO Jr, Sriram S (2013) Astrogliopathy and oligodendrogliopathy are early events in CNS demyelination. Glia 61:1261–1273
Acknowledgments
The scientific guarantor of this publication is Dr. Fabio Triulzi. The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article. This study has received funding by the Italian Ministry of Health to Dr. Arrigoni (RC2012) and Dr. Borgatti. No complex statistical methods were necessary for this paper. Institutional Review Board approval was obtained. Written informed consent was obtained from all subjects (patients) in this study. Methodology: retrospective, cross sectional study, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 946 kb)
Rights and permissions
About this article
Cite this article
Arrigoni, F., Romaniello, R., Peruzzo, D. et al. Aberrant supracallosal longitudinal bundle: MR features, pathogenesis and associated clinical phenotype. Eur Radiol 26, 2587–2596 (2016). https://doi.org/10.1007/s00330-015-4084-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-015-4084-6