Abstract
Objectives
To date, PROPELLER MRI, a breathing-motion-insensitive technique, has not been assessed for cystic fibrosis (CF) lung disease. We compared this technique to CT for assessing CF lung disease in children and adults.
Methods
Thirty-eight stable CF patients (median 21 years, range 6-51 years, 22 female) underwent MRI and CT on the same day. Study protocol included respiratory-triggered PROPELLER MRI and volumetric CT end-inspiratory and -expiratory acquisitions. Two observers scored the images using the CF-MRI and CF-CT systems. Scores were compared with intra-class correlation coefficient (ICC) and Bland-Altman plots. The sensitivity and specificity of MRI versus CT were calculated.
Results
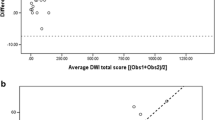
MRI sensitivity for detecting severe CF bronchiectasis was 0.33 (CI 0.09-0.57), while specificity was 100 % (CI 0.88-1). ICCs for bronchiectasis and trapped air were as follows: MRI-bronchiectasis (0.79); CT-bronchiectasis (0.85); MRI-trapped air (0.51); CT-trapped air (0.87). Bland-Altman plots showed an MRI tendency to overestimate the severity of bronchiectasis in mild CF disease and underestimate bronchiectasis in severe disease.
Conclusions
Motion correction in PROPELLER MRI does not improve assessment of CF lung disease compared to CT. However, the good inter- and intra-observer agreement and the high specificity suggest that MRI might play a role in the short-term follow-up of CF lung disease (i.e. pulmonary exacerbations).
Key Points
• PROPELLER MRI does not match CT sensitivity to assess CF lung disease.
• PROPELLER MRI has lower sensitivity than CT to detect severe bronchiectasis.
• PROPELLER MRI has good to very good intra- and inter-observer variability.
• PROPELLER MRI can be used for short-term follow-up studies in CF.





Similar content being viewed by others
Abbreviations
- CF:
-
Cystic fibrosis
- CT:
-
Computed tomography
- CXR:
-
Chest X-ray
- MRI:
-
Magnetic resonance imaging
- PFT:
-
Pulmonary function tests
- PROPELLER:
-
Periodically Rotated Overlapping ParallEL Lines with Enhanced Reconstruction
- TE:
-
Echo time
- TR:
-
Repetition time
References
Cohen-Cymberknoh M, Shoseyov D, Kerem E (2011) Managing cystic fibrosis: strategies that increase life expectancy and improve quality of life. Am J Respir Crit Care Med 183:1463–1471
Tiddens HAWM, Rosenow T (2014) What did we learn from two decades of chest computed tomography in cystic fibrosis? Pediatr Radiol 44:1490–1495
Tepper LA, Utens EMWJ, Caudri D et al (2013) Impact of bronchiectasis and trapped air on quality of life and exacerbations in cystic fibrosis. Eur Respir J 42:371–379
Tiddens HAWM, Donaldson SH, Rosenfeld M, Paré PD (2010) Cystic fibrosis lung disease starts in the small airways: can we treat it more effectively? Pediatr Pulmonol 45:107–117
Eichinger M, Heussel C-P, Kauczor H-U et al (2010) Computed tomography and magnetic resonance imaging in cystic fibrosis lung disease. J Magn Reson Imaging 32:1370–1378
De Jong PA, Lindblad A, Rubin L et al (2006) Progression of lung disease on computed tomography and pulmonary function tests in children and adults with cystic fibrosis. Thorax 61:80–85
Loeve M, Lequin MH, de Bruijne M et al (2009) Cystic fibrosis: are volumetric ultra-low-dose expiratory CT scans sufficient for monitoring related lung disease? Radiology 253:223–229
O’Connor OJ, Vandeleur M, McGarrigle AM et al (2010) Development of low-dose protocols for thin-section CT assessment of cystic fibrosis in pediatric patients. Radiology 257:820–829
Miglioretti DL, Johnson E, Williams A et al (2013) The use of computed tomography in pediatrics and the associated radiation exposure and estimated cancer risk. JAMA Pediatr 167:700–707
Macdougall RD, Strauss KJ, Lee EY (2013) Managing radiation dose from thoracic multidetector computed tomography in pediatric patients: background, current issues, and recommendations. Radiol Clin N Am 51:743–760
Puderbach M, Eichinger M, Gahr J et al (2007) Proton MRI appearance of cystic fibrosis: comparison to CT. Eur Radiol 17:716–724
Puderbach M, Kauczor H-U (2008) Can lung MR replace lung CT? Pediatr Radiol 38:S439–S451
Puderbach M, Eichinger M, Haeselbarth J et al (2007) Assessment of morphological MRI for pulmonary changes in cystic fibrosis (CF) patients: comparison to thin-section CT and chest x-ray. Investig Radiol 42:715–725
Failo R, Wielopolski PA, Tiddens HAWM et al (2009) Lung morphology assessment using MRI: a robust ultra-short TR/TE 2D steady state free precession sequence used in cystic fibrosis patients. Magn Reson Med 61:299–306
Rajaram S, Swift AJ, Capener D et al (2012) Lung morphology assessment with balanced steady-state free precession MR imaging compared with CT. Radiology 263:569–577
Hirokawa Y, Isoda H, Maetani YS et al (2008) Evaluation of motion correction effect and image quality with the periodically rotated overlapping parallel lines with enhanced reconstruction (PROPELLER) (BLADE) and parallel imaging acquisition technique in the upper abdomen. J Magn Reson Imaging 28:957–962
Tamhane AA, Arfanakis K (2009) Motion correction in periodically-rotated overlapping parallel lines with enhanced reconstruction (PROPELLER) and turboprop MRI. Magn Reson Med 62:174–182
Biederer J, Beer M, Hirsch W et al (2012) MRI of the lung (2/3). Why … when … how? Insights Imaging 3:355–371
Laszlo G (2006) Standardisation of lung function testing: helpful guidance from the ATS/ERS Task Force. Thorax 61:744–746
Wainwright CE, Armstrong DS, Byrnes CA et al (2011) Effect of bronchoalveolar lavage – directed therapy on pseudomonas aeruginosa infection and structural lung injury in children with cystic fibrosis. JAMA 306:163–171
Portney LG WM (2000) Foundations of clinical research: applications to practice. Prentice Hall 560–567
Loeve M, van Hal PTW, Robinson P et al (2009) The spectrum of structural abnormalities on CT scans from patients with CF with severe advanced lung disease. Thorax 64:876–882
Contoli M, Kraft M, Hamid Q et al (2012) Do small airway abnormalities characterize asthma phenotypes? In search of proof. Clin Exp Allergy 42:1150–1160
Lederlin M, Bauman G, Eichinger M et al (2013) Functional MRI using Fourier decomposition of lung signal: reproducibility of ventilation- and perfusion-weighted imaging in healthy volunteers. Eur J Radiol 82:1015–1022
Brodsky EK, Bultman EM, Johnson KM et al (2013) High-spatial and high-temporal resolution dynamic contrast-enhanced perfusion imaging of the liver with time-resolved three-dimensional radial MRI. Magn Reson Med. doi:10.1002/mrm.24727
Serra G, Milito C, Mitrevski M et al (2011) Lung MRI as a possible alternative to CT scan for patients with primary immune deficiencies and increased radiosensitivity. Chest 140:1581–1589
Pandit P, Qi Y, King KF, Johnson GA (2011) Reduction of artifacts in T2 -weighted PROPELLER in high-field preclinical imaging. Magn Reson Med 65:538–543
Bonnel A-S, Song SM-H, Kesavarju K et al (2004) Quantitative air-trapping analysis in children with mild cystic fibrosis lung disease. Pediatr Pulmonol 38:396–405
Acknowledgments
P.C. acknowledges financial support from the Italian Cystic Fibrosis League (Lega Italiana Fibrosi Cistica, LIFC). The researchers also wish to express their deepest gratitude to all CF patients who participated in the study. The scientific guarantor of this publication is Dr Giovanni Morana. The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article. Sandra Spronk, statistician, kindly provided statistical advice for this manuscript. Institutional Review Board approval was obtained from all participating centres. Written informed consent was obtained from all subjects (patients) in this study. Methodology: prospective, comparative cross-sectional, multicentre study.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
Bland-Altman plot of MRImean obs 1+2 vs. CTmean obs 1+2. All scoring data are numerical and scores are expressed as percentage of the maximal possible score. Horizontal axis: average air wall thickening subscore [(MRImean + CTmean)/2]; vertical axis = difference in the air wall thickening sub-score (MRImean -CTmean). Red and green lines represent mean and ±2 standard deviations (SD) respectively. (GIF 156 kb)
ESM 2
Bland-Altman plot of MRImean obs 1+2 vs. CTmean obs 1+2. All scoring data are numerical and scores are expressed as percentage of the maximal possible score. Horizontal axis: average mucus sub-score [(MRImean + CTmean)/2]; vertical axis = difference in the mucus sub-score (MRImean - CTmean). Red and green lines represent mean and ±2 standard deviations (SD) respectively. (GIF 156 kb)
ESM 3
Bland-Altman plot of MRImean obs 1+2 vs. CTmean obs 1+2. All scoring data are numerical and scores are expressed as percentage of the maximal possible score. Horizontal axis: average parenchyma sub-score [(MRImean + CTmean)/2]; vertical axis = difference in the parenchyma sub-score (MRImean - CTmean). Red and green lines represent mean and ±2 standard deviations (SD) respectively. (GIF 160 kb)
ESM 4
Bland-Altman plot of MRImean obs 1+2 vs. CTmean obs 1+2. All scoring data are numerical and scores are expressed as percentage of the maximal possible score. Horizontal axis: average total-score [(MRImean + CTmean)/2]; vertical axis = difference in the total score (MRImean - CTmean). Red and green lines represent mean and ±2 standard deviations (SD) respectively. (GIF 148 kb)
ESM 5
Bland-Altman plot of MRImean obs 1+2 vs. CTmean obs 1+2. All scoring data are numerical and scores are expressed as percentage of the maximal possible score. Horizontal axis: average trapped air sub-score [(MRImean + CTmean)/2]; vertical axis = difference in the trapped-air sub-score (MRImean - CTmean). Red and green lines represent mean and ±2 standard deviations (SD) respectively. (GIF 149 kb)
Rights and permissions
About this article
Cite this article
Ciet, P., Serra, G., Bertolo, S. et al. Assessment of CF lung disease using motion corrected PROPELLER MRI: a comparison with CT. Eur Radiol 26, 780–787 (2016). https://doi.org/10.1007/s00330-015-3850-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-015-3850-9