Abstract
Objectives
To assess the interchangeability of zone-specific (peripheral-zone (PZ) and transition-zone (TZ)) multiparametric-MRI (mp-MRI) logistic-regression (LR) models for classification of prostate cancer.
Methods
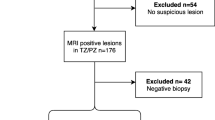
Two hundred and thirty-one patients (70 TZ training-cohort; 76 PZ training-cohort; 85 TZ temporal validation-cohort) underwent mp-MRI and transperineal-template-prostate-mapping biopsy. PZ and TZ uni/multi-variate mp-MRI LR-models for classification of significant cancer (any cancer-core-length (CCL) with Gleason > 3 + 3 or any grade with CCL ≥ 4 mm) were derived from the respective cohorts and validated within the same zone by leave-one-out analysis. Inter-zonal performance was tested by applying TZ models to the PZ training-cohort and vice-versa. Classification performance of TZ models for TZ cancer was further assessed in the TZ validation-cohort. ROC area-under-curve (ROC-AUC) analysis was used to compare models.
Results
The univariate parameters with the best classification performance were the normalised T2 signal (T2nSI) within the TZ (ROC-AUC = 0.77) and normalized early contrast-enhanced T1 signal (DCE-nSI) within the PZ (ROC-AUC = 0.79). Performance was not significantly improved by bi-variate/tri-variate modelling. PZ models that contained DCE-nSI performed poorly in classification of TZ cancer. The TZ model based solely on maximum-enhancement poorly classified PZ cancer.
Conclusion
LR-models dependent on DCE-MRI parameters alone are not interchangable between prostatic zones; however, models based exclusively on T2 and/or ADC are more robust for inter-zonal application.
Key points
• The ADC and T2-nSI of benign/cancer PZ are higher than benign/cancer TZ.
• DCE parameters are significantly different between benign PZ and TZ, but not between cancerous PZ and TZ.
• Diagnostic models containing contrast enhancement parameters have reduced performance when applied across zones.






Similar content being viewed by others
Abbreviations
- PSA:
-
prostate specific antigen
- TRUS:
-
transrectal ultrasound guided biopsy
- CAD:
-
computer assisted diagnosis
- LR:
-
logistic regression
- TPM:
-
template mapping biopsy
- DWI:
-
diffusion weighted imaging
- DCE:
-
dynamic contrast enhanced
- mp:
-
multi-parametric
- SI:
-
signal intensity
- T2-nSI:
-
normalized T2 signal intensity
- DCE-nSI:
-
early contrast enhanced T1 signal intensity
- SoE:
-
slope of enhancement
- ME:
-
maximum enhancement
- Etype:
-
curve type
- AUCtot:
-
total area under the dynamic contrast enhanced signal intensity time curve
- ROC:
-
receiver operator characteristic
- ROI:
-
region of interest
- AUC:
-
area under curve
References
Prostate cancer: diagnosis and treatment. NICE clinical guideline 175 Issued: January 2014 guidance.nice.org.uk/cg175
Barentsz JO, Richenberg J, Clements R et al (2012) ESUR prostate MR guidelines 2012. Eur Radiol 22:746–757
Dickinson L, Ahmed HU, Allen C et al (2011) Magnetic resonance imaging for the detection, localisation, and characterisation of prostate cancer: recommendations from a European consensus meeting. Eur Urol 59(4):477–494
Kirkham AP, Haslam P, Keanie JY et al (2013) Prostate MRI: who, when, and how? Report from a UK consensus meeting. Clin Radiol 68(10):1016–1023
Hoeks CM, Barentsz JO, Hambrock T et al (2011) Prostate cancer: multiparametric MR imaging for detection, localization, and staging. Radiology 261:46–66
Ruprecht O, Weisser P, Bodelle B et al (2012) MRI of the prostate: interobserver agreement compared with histopathologic outcome after radical prostatectomy. Eur J Radiol 81(3):456–460
Fütterer JJ (2007) MR imaging in local staging of prostate cancer. Eur J Radiol 63:328–334
Puech P, Betrouni N, Makni N et al (2009) Computer-assisted diagnosis of prostate cancer using DCE-MRI data: design, implementation and preliminary results. Int J Comput Assist Radiol Surg 4(1):1–10
Hambrock T, Vos PC (2013) Hulsbergen-van de Kaa CA, et al. Prostate cancer: computer-aided diagnosis with multiparametric 3-T MR imaging–effect on observer performance. Radiology 266(2):521–530
Sung YS, Kwon HJ, Park BW et al (2011) Prostate cancer detection on dynamic contrast-enhanced MRI: computer-aided diagnosis versus single perfusion parameter maps. AJR Am J Roentgenol 197(5):1122–1129
Niaf E, Rouvière O, Mège-Lechevallier F et al (2012) Computer-aided diagnosis of prostate cancer in the peripheral zone using multiparametric MRI. Phys Med Biol 57(12):3833–3851
Yoshizako T, Wada A, Hayashi T et al (2008) Usefulness of diffusion-weighted imaging and dynamic contrast-enhanced magnetic resonance imaging in the diagnosis of prostate transition-zone cancer. Acta Radiol 49:1207–1213
Hoeks CM, Hambrock T, Yakar D et al (2013) Transition zone prostate cancer: detection and localization with 3-T multiparametric MR imaging. Radiology 266(1):207–217
Engelbrecht MR, Huisman HJ, Laheij RJ et al (2003) Discrimination of prostate cancer from normal peripheral zone and central gland tissue by using dynamic contrast-enhanced MR imaging. Radiology 229:248–254
Greene DR, Wheeler TM, Egawa S et al (1991) A comparison of the morphological features of cancer arising in the transition zone and in the peripheral zone of the prostate. J Urol 146(4):1069–1076
Akin O, Sala E, Moskowitz CS et al (2006) Transition zone prostate cancers: features, detection, localization, and staging at endorectal MR imaging. Radiology 239(3):784–792
Erbersdobler A, Fritz H, Schnöger S et al (2002) Tumour grade, proliferation, apoptosis, microvessel density, p53, and bcl-2 in prostate cancers: differences between tumours located in the transition zone and in the peripheral zone. Eur Urol 41(1):40–46
Buckley DL, Roberts C, Parker GJ et al (2004) Prostate cancer: evaluation of vascular characteristics with dynamic contrast-enhanced T1-weighted MR imaging–initial experience. Radiology 233(3):709–715
Langer DL, van der Kwast TH, Evans AJ et al (2009) Prostate cancer detection with multi-parametric MRI: logistic regression analysis of quantitative T2, diffusion-weighted imaging, and dynamic contrast-enhanced MRI. J Magn Reson Imaging 30:327–334
Choi YJ, Kim JK, Kim N et al (2007) Functional MR imaging of prostate cancer. Radiographics 27(1):63–75
van Niekerk CG, Witjes JA, Barentsz JO et al (2013) Microvascularity in transition zone prostate tumors resembles normal prostatic tissue. Prostate 73(5):467–475
Onik G, Barzell W (2008) Transperineal 3D mapping biopsy of the prostate: an essential tool in selecting patients for focal prostate cancer therapy. Urol Oncol 26:506–510
Taira AV, Merrick GS, Galbreath RW et al (2010) Performance of transperineal template-guided mapping biopsy in detecting prostate cancer in the initial and repeat biopsy setting. Prostate Cancer Prostatic Dis 13:71–77
Barzell WE, Melamed MR (2007) Appropriate patient selection in the focal treatment of prostate cancer: the role of transperineal 3-dimensional pathologic mapping of the prostate–a 4-year experience. Urology 70:27–35
Ahmed HU, Hu Y, Carter T et al (2011) Characterizing clinically significant prostate cancer using template prostate mapping biopsy. J Urol 186:458–464
Zelhof B, Lowry M, Rodrigues G et al (2009) Description of magnetic resonance imaging-derived enhancement variables in pathologically confirmed prostate cancer and normal peripheral zone regions. BJU Int 104:621–627
Tofts PS (1997) Modeling tracer kinetics in dynamic Gd-DTPA MR imaging. J Magn Reson Imaging 7(1):91–101
S M Applied logistic regression analysis Sage University Paper Series on Quantitative Applications in the Social Sciences. 1995: 7–106
Fukunaga K, Hummels DM (1989) Leave-one-out procedures for nonparametric error estimates. IEEE Trans Pattern Anal Mach Intell 11:421–423
Ahmed HU, Emberton M, Kepner G et al (2012) A biomedical engineering approach to mitigate the errors of prostate biopsy. Nat Rev Urol 9:227–231
Roy C, Foudi F, Charton J et al (2013) Comparative sensitivities of functional MRI sequences in detection of local recurrence of prostate carcinoma after radical prostatectomy or external-beam radiotherapy. AJR Am J Roentgenol 200:361–368
Turkbey B, Pinto PA, Mani H et al (2010) Prostate cancer: value of multiparametric MR imaging at 3 T for detection–histopathologic correlation. Radiology 255(1):89–99
Kim CK, Park BK, Han JJ et al (2007) Diffusion-weighted imaging of the prostate at 3 T for differentiation of malignant and benign tissue in transition and peripheral zones: preliminary results. J Comput Assist Tomogr 31:449–454
Chesnais AL, Niaf E, Bratan F et al (2013) Differentiation of transitional zone prostate cancer from benign hyperplasia nodules: evaluation of discriminant criteria at multiparametric MRI. Clin Radiol 68(6):e323–e330
Oto A, Kayhan A, Jiang Y et al (2010) Prostate cancer: differentiation of central gland cancer from benign prostatic hyperplasia by using diffusion-weighted and dynamic contrast-enhanced MR imaging. Radiology 257:715–723
Acknowledgments
The scientific guarantor of this publication is Shonit Punwani. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article. This work was undertaken at the NIHR UCLH/UCL Biomedical Research Centre, which received a portion of the funding from the National Institute for Health Research. The views expressed in this publication are those of the authors and not necessarily those of the UK Department of Health.
This work was supported by the CRUK/EPSRC KCL/UCL Comprehensive Cancer Imaging Centre. ND was supported by UK EPSRC grants EP/I018700/1 and EP/H046410/1.
No complex statistical methods were necessary for this paper. Institutional Review Board approval was obtained. Written informed consent was waived by the Institutional Review Board. Methodology: retrospective, experimental, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dikaios, N., Alkalbani, J., Abd-Alazeez, M. et al. Zone-specific logistic regression models improve classification of prostate cancer on multi-parametric MRI. Eur Radiol 25, 2727–2737 (2015). https://doi.org/10.1007/s00330-015-3636-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-015-3636-0