Abstract
Objectives
We aimed to determine whether initial tumour responses measured during short-term follow-up computed tomography (CT) examinations after baseline examinations would correlate with clinical outcomes in patients with non-small cell lung cancer (NSCLC) who received epidermal growth factor receptor (EGFR)-targeted therapy.
Methods
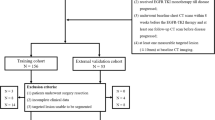
A total of 86 gefitinib-treated patients with advanced adenocarcinoma of the lung were retrospectively reviewed. All patients underwent baseline and short-term follow-up CT examinations. The new response criteria (NRC) by Lee et al. were used for the response evaluations. A Cox proportional hazards multiple regression model and Kaplan–Meier survival analyses were used to evaluate correlations between the initial tumour changes and progression-free and overall survival (PFS, OS).
Results
Better separation and smaller p values were observed for both PFS and OS when good and poor disease responses (as defined by NRC) were compared after excluding tumours with characteristic morphologies. Early tumour changes correlated with PFS in a size-dependent manner. Moreover, a stronger association was observed between size changes and PFS when characteristic morphology was also considered.
Conclusions
Initial changes in tumour size during short-term post-treatment CT examinations could act as a potential prognostic imaging surrogate for PFS in gefitinib-treated patients with advanced adenocarcinoma of the lung.
Key points
• Initial responses to gefitinib on computed tomography significantly correlate with clinical outcomes.
• Regardless of morphology, size decrease greater than 30 % predicts prolonged progression-free and overall survival.
• Combination of size and morphological changes yields prognostic independence regarding progression-free survival.





Similar content being viewed by others
Abbreviations
- CECT:
-
contrast-enhanced CT
- CI:
-
confidence intervals
- CR:
-
complete response
- CT:
-
computed tomography
- ECOG PS:
-
Eastern Cooperative Oncology Group Performance Status
- EGFR:
-
epidermal growth factor receptor
- EMR:
-
electronic medical records
- HR:
-
hazard ratio
- LD:
-
longest dimension
- NRC:
-
new response criteria
- NSCLC:
-
non-small cell lung cancer
- OS:
-
overall survival
- PD:
-
progressive disease
- PFS:
-
progression-free survival
- PR:
-
partial response
- RECIST 1.1:
-
Response Evaluation Criteria in Solid Tumors, version 1.1
- SD:
-
stable disease
- TDR:
-
tumour decrease rate
- TKI:
-
tyrosine kinase inhibitor
- VEGF:
-
vascular endothelial growth factor
References
Shepherd FA, Rodrigues Pereira J et al (2005) Erlotinib in previously treated non-small-cell lung cancer. N Engl J Med 353:123–132
Cataldo VD, Gibbons DL, Pérez-Soler R, Quintás-Cardama A (2011) Treatment of non-small-cell lung cancer with erlotinib or gefitinib. N Engl J Med 364:947–955
Tsao MS, Sakurada A, Cutz JC et al (2005) Erlotinib in lung cancer—molecular and clinical predictors of outcome. N Engl J Med 353:133–144
Sequist LV, Bell DW, Lynch TJ, Haber DA (2007) Molecular predictors of response to epidermal growth factor receptor antagonists in non-small-cell lung cancer. J Clin Oncol 25:587–595
Rosell R, Molina MA, Costa C et al (2011) Pretreatment EGFR T790M mutation and BRCA1 mRNA expression in erlotinib-treated advanced non-small-cell lung cancer patients with EGFR mutations. Clin Cancer Res 17:1160–1168
Schaake EE, Kappers I, Codrington HE et al (2012) Tumor response and toxicity of neoadjuvant erlotinib in patients with early-stage non-small-cell lung cancer. J Clin Oncol 30:2731–2738
Lee HY, Lee KS, Ahn MJ et al (2011) New CT response criteria in non-small cell lung cancer: proposal and application in EGFR tyrosine kinase inhibitor therapy. Lung Cancer 73:63–69
Oken MM, Creech RH, Tormey DC et al (1982) Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 5:649–655
Lin LI (1989) A concordance correlation coefficient to evaluate reproducibility. Biometrics 45:255–268
Bland JM, Altman DG (1986) Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1:307–310
Lavin PT (1981) An alternative model for the evaluation of antitumor activity. Cancer Clin Trials 4:451–457
Karrison TG, Maitland ML, Stadler WM, Ratain MJ (2001) Design of phase II cancer trials using a continuous endpoint of change in tumor size: application to a study of sorafenib and erlotinib in non small-cell lung cancer. J Natl Cancer Inst 99:1455–1461
Smith AD, Shah SN, Rini BI, Lieber ML, Remer EM (2013) Utilizing pre-therapy clinical schema and initial CT changes to predict progression-free survival in patients with metastatic renal cell carcinoma on VEGF-targeted therapy: a preliminary analysis. Urol Oncol 31:1283–1291
Suzuki C, Blomqvist L, Sundin A et al (2012) The initial change in tumor size predicts response and survival in patients with metastatic colorectal cancer treated with combination chemotherapy. Ann Oncol 23:948–954
Jang KM, Kim SH, Lee SJ et al (2013) Added value of diffusion-weighted MR imaging in the diagnosis of ampullary carcinoma. Radiology 266:491–501
Somoye G, Harry V, Semple S et al (2012) Early diffusion weighted magnetic resonance imaging can predict survival in women with locally advanced cancer of the cervix treated with combined chemo-radiation. Eur Radiol 22:2319–2327
Wang J, Wu N, Cham MD et al (2009) Tumor response in patients with advanced non-small cell lung cancer: perfusion CT evaluation of chemotherapy and radiation therapy. AJR Am J Roentgenol 193:1090–1096
Therasse P, Arbuck SG, Eisenhauer EA et al (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumours: revised RECIST guidelines (version 1.1). Eur J Cancer 45:228–247
Stacchiotti S, Collini P, Messina A et al (2009) High-grade soft-tissue sarcomas: tumor response assessment–pilot study to assess the correlation between radiologic and pathologic response by using RECIST and Choi criteria. Radiology 251:447–456
Husband JE, Schwartz LH, Spencer J et al (2004) Evaluation of the response to treatment of solid tumours - a consensus statement of the International Cancer Imaging Society. Br J Cancer 90:2256–2260
Lee HY, Lee KS, Hwang HS et al (2010) Molecularly targeted therapy using bevacizumab for non-small cell lung cancer: a pilot study for the new CT response criteria. Korean J Radiol 11:618–626
Kim YN, Lee HY, Lee KS et al (2012) Dual-energy CT in patients treated with anti-angiogenic agents for non-small cell lung cancer: new method of monitoring tumor response? Korean J Radiol 13:702–710
Tsujino K, Kawaguchi T, Kubo A et al (2009) Response rate is associated with prolonged survival in patients with advanced non-small cell lung cancer treated with gefitinib or erlotinib. J Thorac Oncol 4:994–1001
Ratain MJ, Eckhardt SG (2004) Phase II studies of modern drugs directed against new targets: if you are fazed, too, then resist RECIST. J Clin Oncol 22:4442–4445
Choi H, Charnsangavej C, Faria SC et al (2007) Correlation of computed tomography and positron emission tomography in patients with metastatic gastrointestinal stromal tumor treated at a single institution with imatinib mesylate: proposal of new computed tomography response criteria. J Clin Oncol 25:1753–1759
Sathornsumetee S, Cao Y, Marcello JE et al (2008) Tumor angiogenic and hypoxic profiles predict radiographic response and survival in malignant astrocytoma patients treated with bevacizumab and irinotecan. J Clin Oncol 26:271–278
Grothey A, Hedrick EE, Mass RD et al (2008) Response-independent survival benefit in metastatic colorectal cancer: a comparative analysis of N9741 and AVF2107. J Clin Oncol 26:183–189
Junker K, Langner K, Klinke F, Bosse U, Thomas M (2001) Grading of tumor regression in non-small cell lung cancer : morphology and prognosis. Chest 120:1584–1591
Junker K, Thomas M, Schulmann K, Klinke F, Bosse U, Müller KM (1997) Tumour regression in non-small-cell lung cancer following neoadjuvant therapy. Histological assessment. J Cancer Res Clin Oncol 123:469–477
Park MJ, Lee J, Hong JY et al (2009) Prognostic model to predict outcomes in nonsmall cell lung cancer patients treated with gefitinib as a salvage treatment. Cancer 115:1518–1530
Buyse M, Burzykowski T, Carroll K et al (2007) Progression-free survival is a surrogate for survival in advanced colorectal cancer. J Clin Oncol 25:5218–5224
Tang PA, Bentzen SM, Chen EX, Siu LL (2007) Surrogate end points for median overall survival in metastatic colorectal cancer: literature-based analysis from 39 randomized controlled trials of first-line chemotherapy. J Clin Oncol 25:4562–4568
Carroll KJ (2007) Analysis of progression-free survival in oncology trials: some common statistical issues. Pharm Stat 6:99–113
Shi Q, Sargent DJ (2009) Meta-analysis for the evaluation of surrogate endpoints in cancer clinical trials. Int J Clin Oncol 14:102–111
Levin VA, Ictech S, Hess KR (2007) Impact of phase II trials with progression-free survival as end-points on survival-based phase III studies in patients with anaplastic gliomas. BMC Cancer 7:106
Methy N, Bedenne L, Bonnetain F (2009) Validation of surrogate endpoints in digestive oncology. Bull Cancer 96:591–595
Lee KS, Kim Y, Han J, Ko EJ, Park CK, Primack SL (1997) Bronchioloalveolar carcinoma: clinical, histopathologic, and radiologic findings. Radiographics 17:1345–1357
Acknowledgments
The authors thank the Cancer Registry Group of Tri-Service General Hospital for the help with the clinical data. The scientific guarantor of this publication is Hsian-He Hsu. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article. The authors state that this work has not received any funding. One of the authors has significant statistical expertise. Institutional review board approval was obtained. Written informed consent was waived by the institutional review board. No study subjects or cohorts have been previously reported. Methodology: retrospective, diagnostic or prognostic study, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, YC., Hsu, HH., Chang, WC. et al. Prognostic potential of initial CT changes for progression-free survival in gefitinib-treated patients with advanced adenocarcinoma of the lung: a preliminary analysis. Eur Radiol 25, 1801–1813 (2015). https://doi.org/10.1007/s00330-014-3579-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-014-3579-x