Abstract
Objectives
To implement a novel voxel-based technique to identify statistically significant local cartilage deformation and analyze in-vivo topographic knee cartilage deformation patterns using a voxel-based thickness map approach for high-flexion postures.
Methods
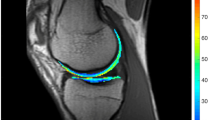
Sagittal 3T 3D-T1w-FLASH-WE-sequences of 10 healthy knees were acquired before and immediately after loading (kneeling/squatting/heel sitting/knee bends). After cartilage segmentation, 3D-reconstruction and 3D-registration, colour-coded deformation maps were generated by voxel-based subtraction of loaded from unloaded datasets to visualize cartilage thickness changes in all knee compartments.
Results
Compression areas were found bifocal at the peripheral medial/caudolateral patella, both posterior femoral condyles and both anterior/central tibiae. Local cartilage thickening were found adjacent to the compression areas. Significant local strain ranged from +13 to -15 %. Changes were most pronounced after squatting, least after knee bends. Shape and location of deformation areas varied slightly with the loading paradigm, but followed a similar pattern consistent between different individuals.
Conclusions
Voxel-based deformation maps identify individual in-vivo load-specific and posture-associated strain distribution in the articular cartilage. The data facilitate understanding individual knee loading properties and contribute to improve biomechanical 3 models. They lay a base to investigate the relationship between cartilage degeneration patterns in common osteoarthritis and areas at risk of cartilage wear due to mechanical loading in work-related activities.
Key points
• 3D MRI helps differentiate true knee-cartilage deformation from random measurement error
• 3D MRI maps depict in vivo topographic distribution of cartilage deformation after loading
• 3D MRI maps depict in vivo intensity of cartilage deformation after loading
• Locating cartilage contact areas might aid differentiating common and work-related osteoarthritis






Similar content being viewed by others
Abbreviations
- CBI:
-
Cartilage–bone interface
- CBIA:
-
Cartilage–bone interface area
- CUELA:
-
Computer-assisted recording and long-term analysis system for loading on the musculoskeletal system
- IFA:
-
Sankt Augustin, Germany
- FLASH:
-
Fast low-angle shot
- MRI:
-
Magnetic resonance imaging
- mTh:
-
Mean thickness
- OA:
-
Osteoarthritis
- Vol:
-
Volume
References
Ateshian GA, Lai WM, Zhu WB, Mow VC (1994) An asymptotic solution for the contact of two biphasic cartilage layers. J Biomech 27:1347–1360
Mow VC, Holmes MH, Lai WM (1984) Fluid transport and mechanical properties of articular cartilage: a review. J Biomech 17:377–394
Griffin TM, Guilak F (2005) The role of mechanical loading in the onset and progression of osteoarthritis. Exerc Sport Sci Rev 33:195–200
Guilak F (2012) Biomechanical factors in osteoarthritis. Best Pract Res Clin Rheumatol 25:815–823
Eckstein F, Cicuttini F, Raynauld JP, Waterton JC, Peterfy C (2006) Magnetic resonance imaging (MRI) of articular cartilage in knee osteoarthritis (OA): morphological assessment. Osteoarthr Cartil 14:A46–A75
Eckstein F, Hudelmaier M, Putz R (2006) The effects of exercise on human articular cartilage. J Anat 208:491–512
Eckstein F, Lemberger B, Gratzke C, Hudelmaier M, Glaser C, Englmeier KH et al (2005) In vivo cartilage deformation after different types of activity and its dependence on physical training status. Ann Rheum Dis 64:291–295
Eckstein F, Tieschky M, Faber SC, Haubner M, Kolem H, Englmeier KH et al (1998) Effect of physical exercise on cartilage volume and thickness in vivo: MR imaging study. Radiology 207:243–248
Eckstein F, Lemberger B, Stammberger T, Englmeier KH, Reiser M (2000) Patellar cartilage deformation in vivo after static versus dynamic loading. J Biomech 33:819–825
Hartmann B, Glitsch U, Görgens HW, Grosser V, Weber M, Schürmann J et al (2007) Ein belastungskonformes Schadensbild der Gonarthrose durch Knien oder vergleichbare Kniebelastung? Arbeitsmed Sozialmed Umweltmed 42:64–67
(2005) Berufskrankheiten-Verordnung - Bekanntmachung des BGMS. BArbBl (10):46
Ellegast R, Kupfer J, Reinert D (1997) Load weight determination during dynamic working procedures using the pedar foot pressure distribution measuring system. Clin Biomech (Bristol, Avon) 12:S10–S11
Glaser C, Faber S, Eckstein F, Fischer H, Springer V, Heudorfer L et al (2001) Optimization and validation of a rapid high-resolution T1-w 3D FLASH water excitation MRI sequence for the quantitative assessment of articular cartilage volume and thickness. Magn Reson Imaging 19:177–185
Glocker B, Komodakis N, Paragios N, Glaser C, Tziritas G, Navab N (2007) Primal/dual linear programming and statistical atlases for cartilage segmentation. Med Image Comput Comput Assist Interv 10:536–543
König L, Groher M, Keil A, Glaser C, Reiser M, Navab N (2007) Semi-automatic segmentation of the patellar cartilage in MRI. Bildverarbeitung Die Med :404–408
Raya JG, Horng A, Dietrich O, Weber J, Dinges J, Mutzel E et al (2009) Voxel-based reproducibility of T2 relaxation time in patellar cartilage at 1.5T with a new validated 3D rigid registration algorithm. MAGMA 22:229–239
Eckstein F, Charles HC, Buck RJ, Kraus VB, Remmers AE, Hudelmaier M et al (2005) Accuracy and precision of quantitative assessment of cartilage morphology by magnetic resonance imaging at 3.0T. Arthritis Rheum 52:3132–3136
Herberhold C, Faber S, Stammberger T, Steinlechner M, Putz R, Englmeier KH et al (1999) In situ measurement of articular cartilage deformation in intact femoropatellar joints under static loading. J Biomech 32:1287–1295
Bingham JT, Papannagari R, Van de Velde SK, Gross C, Gill TJ, Felson DT et al (2008) In vivo cartilage contact deformation in the healthy human tibiofemoral joint. Rheumatology (Oxford) 47:1622–1627
Hehne HJ (1990) Biomechanics of the patellofemoral joint and its clinical relevance. Clin Orthop Relat Res 258:73–85
Heegaard J, Leyvraz PF, Curnier A, Rakotomanana L, Huiskes R (1995) The biomechanics of the human patella during passive knee flexion. J Biomech 28:1265–1279
Hinterwimmer S, von Eisenhart-Rothe R, Siebert M, Welsch F, Vogl T, Graichen H (2004) Patella kinematics and patello-femoral contact areas in patients with genu varum and mild osteoarthritis. Clin Biomech (Bristol, Avon) 19:704–710
Nakagawa S, Kadoya Y, Kobayashi A, Tatsumi I, Nishida N, Yamano Y (2003) Kinematics of the patella in deep flexion. Analysis with magnetic resonance imaging. J Bone Joint Surg Am 85-A:1238–1242
Nakagawa S, Kadoya Y, Todo S, Kobayashi A, Sakamoto H, Freeman MA et al (2000) Tibiofemoral movement 3: full flexion in the living knee studied by MRI. J Bone Joint Surg (Br) 82:1199–1200
Freeman MA, Pinskerova V (2005) The movement of the normal tibio-femoral joint. J Biomech 38:197–208
Goodfellow J, Hungerford DS, Zindel M (1976) Patello-femoral joint mechanics and pathology. 1. Functional anatomy of the patello-femoral joint. J Bone Joint Surg (Br) 58:287–290
Horng A, Raya J, Zscharn M, Konig L, Notohamiprodjo M, Pietschmann M, et al (2011) Locoregional deformation pattern of the patellar cartilage after different loading types - high-resolution 3D-MRI volumetry at 3T in vivo. Fortschr Röntgenstr
Cicuttini F, Forbes A, Asbeutah A, Morris K, Stuckey S (2000) Comparison and reproducibility of fast and conventional spoiled gradient-echo magnetic resonance sequences in the determination of knee cartilage volume. J Orthop Res 18:580–584
Lee TQ, Morris G, Csintalan RP (2003) The influence of tibial and femoral rotation on patellofemoral contact area and pressure. J Orthop Sports Phys Ther 33:686–693
Li G, Park SE, DeFrate LE, Schutzer ME, Ji L, Gill TJ et al (2005) The cartilage thickness distribution in the tibiofemoral joint and its correlation with cartilage-to-cartilage contact. Clin Biomech (Bristol, Avon) 20:736–744
Liu F, Kozanek M, Hosseini A, Van de Velde SK, Gill TJ, Rubash HE et al In vivo tibiofemoral cartilage deformation during the stance phase of gait. J Biomech 43(4):658–665
Froimson MI, Ratcliffe A, Gardner TR, Mow VC (1997) Differences in patellofemoral joint cartilage material properties and their significance to the etiology of cartilage surface fibrillation. Osteoarthr Cartil 5:377–386
Athanasiou KA, Rosenwasser MP, Buckwalter JA, Malinin TI, Mow VC (1991) Interspecies comparisons of in situ intrinsic mechanical properties of distal femoral cartilage. J Orthop Res 9:330–340
Spahn G, Wittig R (2003) Biomechanical properties (compressive strength and compressive pressure at break) of hyaline cartilage under axial load. Zentralbl Chir 128:78–82
Walker PS, Arno S, Liang S, Yildirim G, Regatte R, Samuels J et al (2011) Medial osteoarthritis of the knee and its association with the medial meniscus. In. ORS. Long Beach, CA
Acknowledgement
The scientific guarantor of this publication is Prof. Dr. Christian Glaser. The authors declare no relationships with any companies, whose products or services may be related to the subject matter of the article. This study has received funding by the German Social Accident Insurance (DGUV; Deutsche Gesetzliche Unfallversicherung). One of the authors has significant statistical expertise. Institutional Review Board approval was obtained. Written informed consent was obtained from all subjects in this study. In this study no animals were examined. Some study subjects have been previously reported in: Horng A, Raya J, Zscharn M, et al. [Locoregional deformation pattern of the patellar cartilage after different loading types - high-resolution 3D-MRI volumetry at 3T in vivo]. Rofo. 2011 May;183 (5):432-40. German. Methodology: prospective, cross-sectional study / observational / experimental, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Horng, A., Raya, J.G., Stockinger, M. et al. Topographic deformation patterns of knee cartilage after exercises with high knee flexion: an in vivo 3D MRI study using voxel-based analysis at 3T. Eur Radiol 25, 1731–1741 (2015). https://doi.org/10.1007/s00330-014-3545-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-014-3545-7