Abstract
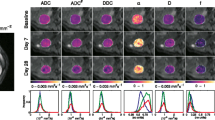
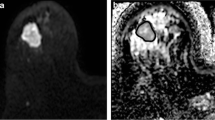
The purpose was to determine the reproducibility of apparent diffusion coefficient (ADC) measurements in a two-centre phase I clinical trial; and to track ADC changes in response to the sequential administration of the vascular disrupting agent, combretastatin A4 phosphate (CA4P), and the anti-angiogenic drug, bevacizumab. Sixteen patients with solid tumours received CA4P and bevacizumab treatment. Echo-planar diffusion-weighted MRI was performed using six b values (b = 0–750 s/mm2) before (×2), and at 3 and 72 h after a first dose of CA4P. Bevacizumab was given 4 h after a second dose of CA4P, and imaging performed 3 h post CA4P and 72 h after bevacizumab treatment. The coefficient of repeatability (r) of ADC total (all b values), ADC high (b = 100–750) and ADC low (b = 0–100) was calculated by Bland–Altman analysis. The ADC total and ADC high showed good measurement reproducibility (r% = 13.3, 14.1). There was poor reproducibility of the perfusion-sensitive ADC low (r% = 62.5). Significant increases in the median ADC total and ADC high occurred at 3 h after the second dose of CA4P (p < 0.05). ADC measurements were highly reproducible in a two-centre clinical trial setting and appear promising for evaluating the effects of drugs that target tumour vasculature.






Similar content being viewed by others
References
Koh DM, Collins DJ (2007) Diffusion-weighted MRI in the body: applications and challenges in oncology. AJR Am J Roentgenol 188:1622–1635
Kamel IR, Bluemke DA, Ramsey D et al (2003) Role of diffusion-weighted imaging in estimating tumor necrosis after chemoembolization of hepatocellular carcinoma. AJR Am J Roentgenol 181:708–710
Mardor Y, Pfeffer R, Spiegelmann R et al (2003) Early detection of response to radiation therapy in patients with brain malignancies using conventional and high b-value diffusion-weighted magnetic resonance imaging. J Clin Oncol 21:1094–1100
Hein PA, Kremser C, Judmaier W et al (2003) Diffusion-weighted magnetic resonance imaging for monitoring diffusion changes in rectal carcinoma during combined, preoperative chemoradiation: preliminary results of a prospective study. Eur J Radiol 45:214–222
Byun WM, Shin SO, Chang Y et al (2002) Diffusion-weighted MR imaging of metastatic disease of the spine: assessment of response to therapy. AJNR Am J Neuroradiol 23:906–912
Geschwind JF, Artemov D, Abraham S et al (2000) Chemoembolization of liver tumor in a rabbit model: assessment of tumor cell death with diffusion-weighted MR imaging and histologic analysis. J Vasc Interv Radiol 11:1245–1255
Koh DM, Scurr E, Collins D et al (2007) Predicting response of colorectal hepatic metastasis: value of pretreatment apparent diffusion coefficients. AJR Am J Roentgenol 188:1001–1008
Kwee TC, Takahara T, Koh DM et al (2008) Comparison and reproducibility of ADC measurements in breathhold, respiratory triggered, and free-breathing diffusion-weighted MR imaging of the liver. J Magn Reson Imaging 28:1141–1148
Thoeny HC, De Keyzer F, Chen F et al (2005) Diffusion-weighted magnetic resonance imaging allows noninvasive in vivo monitoring of the effects of combretastatin A-4 phosphate after repeated administration. Neoplasia 7:779–787
Thoeny HC, De Keyzer F, Vandecaveye V et al (2005) Effect of vascular targeting agent in rat tumor model: dynamic contrast-enhanced versus diffusion-weighted MR imaging. Radiology 237:492–499
Dowlati A, Robertson K, Cooney M et al (2002) A phase I pharmacokinetic and translational study of the novel vascular targeting agent combretastatin A-4 phosphate on a single-dose intravenous schedule in patients with advanced cancer. Cancer Res 62:3408–3416
Galbraith SM, Maxwell RJ, Lodge MA et al (2003) Combretastatin A4 phosphate has tumor antivascular activity in rat and man as demonstrated by dynamic magnetic resonance imaging. J Clin Oncol 21:2831–2842
Stevenson JP, Rosen M, Sun W et al (2003) Phase I trial of the antivascular agent combretastatin A4 phosphate on a 5-day schedule to patients with cancer: magnetic resonance imaging evidence for altered tumor blood flow. J Clin Oncol 21:4428–4438
Ellis LM (2005) Bevacizumab. Nat Rev Drug Discov Suppl:S8–9
Jain RK (2005) Antiangiogenic therapy for cancer: current and emerging concepts. Oncology (Williston Park) 19:7–16
Hormigo A, Gutin PH, Rafii S (2007) Tracking normalization of brain tumor vasculature by magnetic imaging and proangiogenic biomarkers. Cancer Cell 11:6–8
Batchelor TT, Sorensen AG, di Tomaso E et al (2007) AZD2171, a pan-VEGF receptor tyrosine kinase inhibitor, normalizes tumor vasculature and alleviates edema in glioblastoma patients. Cancer Cell 11:83–95
Delakis I, Moore EM, Leach MO et al (2004) Developing a quality control protocol for diffusion imaging on a clinical MRI system. Phys Med Biol 49:1409–1422
Bland JM, Altman DG (1999) Measuring agreement in method comparison studies. Stat Methods Med Res 8:135–160
Galbraith SM, Lodge MA, Taylor NJ et al (2002) Reproducibility of dynamic contrast-enhanced MRI in human muscle and tumours: comparison of quantitative and semi-quantitative analysis. NMR Biomed 15:132–142
Thoeny HC, De Keyzer F, Chen F et al (2005) Diffusion-weighted MR imaging in monitoring the effect of a vascular targeting agent on rhabdomyosarcoma in rats. Radiology 234:756–764
Malcontenti-Wilson C, Muralidharan V, Skinner S et al (2001) Combretastatin A4 prodrug study of effect on the growth and the microvasculature of colorectal liver metastases in a murine model. Clin Cancer Res 7:1052–1060
Tozer GM, Prise VE, Wilson J et al (2001) Mechanisms associated with tumor vascular shut-down induced by combretastatin A-4 phosphate: intravital microscopy and measurement of vascular permeability. Cancer Res 61:6413–6422
Tozer GM, Akerman S, Cross NA et al (2008) Blood vessel maturation and response to vascular-disrupting therapy in single vascular endothelial growth factor-A isoform-producing tumors. Cancer Res 68:2301–2311
Cui Y, Zhang XP, Sun YS et al (2008) Apparent diffusion coefficient: potential imaging biomarker for prediction and early detection of response to chemotherapy in hepatic metastases. Radiology 248:894–900
Dzik-Jurasz A, Domenig C, George M et al (2002) Diffusion MRI for prediction of response of rectal cancer to chemoradiation. Lancet 360:307–308
DeVries AF, Kremser C, Hein PA et al (2003) Tumor microcirculation and diffusion predict therapy outcome for primary rectal carcinoma. Int J Radiat Oncol Biol Phys 56:958–965
Hamstra DA, Galban CJ, Meyer CR et al (2008) Functional diffusion map as an early imaging biomarker for high-grade glioma: correlation with conventional radiologic response and overall survival. J Clin Oncol 26:3387–3394
Lee KC, Bradley DA, Hussain M et al (2007) A feasibility study evaluating the functional diffusion map as a predictive imaging biomarker for detection of treatment response in a patient with metastatic prostate cancer to the bone. Neoplasia 9:1003–1011
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was sponsored by Oxigene (Oxigene Inc, Waltham, MA, USA); and supported by Cancer Research UK Grant C1060/A5117 and NHS funding to the NIHR Biomedical Research Centre.
Appendix: details of Bland–Altman statistical analysis
Appendix: details of Bland–Altman statistical analysis
For each tumour, the distribution of the ADC pixel values was asymmetric, and the median values were thus used to summarise the distribution. However, the distribution of the median values of all tumours and the differences in the baseline median values of tumours conformed to a normal distribution (D’Angostino–Pearson test, p > 0.05), which allowed us to perform Bland–Altman analyses to determine measurement reproducibility of the ADC total, ADC high and ADC low. For each tumour, the difference between the two baseline median ADC measurements (d) was calculated. The mean squared difference was calculated by:
This was used to calculate the 95% confidence interval (CI) for changes in the study cohort of n individuals:
Thus, a change in the ADC value greater than this value in the patient cohort would be significant at the 5% level.
The within-patient coefficient of variance (wCV) was calculated by:
The coefficient of repeatability (r) was calculated by:
The value of the coefficient of repeatability indicates that the difference between the two baseline median ADC measurements for the same tumour will be less than this value for 95% of the pairs of observations. This value is usually expressed as an absolute value on the same scale as the ADC parameter, but may also be approximated as a percentage of the baseline values. For good measurement reproducibility on a per patient basis, both the within-patient coefficient of variance and the coefficient of repeatability should be low. Similarly, for good cohort measurement reproducibility, the group confidence interval should also be small.
Rights and permissions
About this article
Cite this article
Koh, DM., Blackledge, M., Collins, D.J. et al. Reproducibility and changes in the apparent diffusion coefficients of solid tumours treated with combretastatin A4 phosphate and bevacizumab in a two-centre phase I clinical trial. Eur Radiol 19, 2728–2738 (2009). https://doi.org/10.1007/s00330-009-1469-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-009-1469-4