Abstract
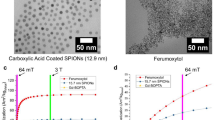
In this study we evaluated the effects of intracellular compartmentalization of the ultrasmall superparamagnetic iron oxide (USPIO) ferumoxtran-10 on its proton T1 and T2 relaxivities at 1.5 and 3T. Monocytes were labeled with ferumoxtran-10 by simple incubation. Decreasing quantities of ferumoxtran-10-labeled cells (2.5×107-0.3×107 cells/ml) and decreasing concentrations of free ferumoxtran-10 (without cells) in Ficoll solution were evaluated with 1.5 and 3T clinical magnetic resonance (MR) scanners. Pulse sequences comprised axial spin echo (SE) sequences with multiple TRs and fixed TE and SE sequences with fixed TR and increasing TEs. Signal intensity measurements were used to calculate T1 and T2 relaxation times of all samples, assuming a monoexponential signal decay. The iron content in all samples was determined by inductively coupled plasma atomic emission spectrometry and used for calculating relaxivities. Measurements at 1.5T and 3T showed higher T1 and T2 relaxivity values of free extracellular ferumoxtran-10 as opposed to intracellularly compartmentalized ferumoxtran-10, under the evaluated conditions of homogeneously dispersed contrast agents/cells in Ficoll solution and a cell density of up to 2.5×107 cells/ml. At 3T, differences in T1-relaxivities between intra- and extracellular USPIO were smaller, while differences in USPIO T2-relaxivities were similar compared with 1.5T. In conclusion, cellular compartmentalization of ferumoxtran-10 changes proton relaxivity.


Similar content being viewed by others
References
Tanimoto A, Oshio K, Suematsu M, Pouliquen D, Stark DD (2001) Relaxation effects of clustered particles. J Magn Reson Imaging 14:72–77
Ferrucci JT, Stark DD (1990) Iron oxide-enhanced MR imaging of the liver and spleen: review of the first 5 years. AJR Am J Roentgenol 155:943–950
Small WC, Nelson RC, Bernardino ME (1993) Dual contrast enhancement of both T1- and T2-weighted sequences using ultrasmall superparamagnetic iron oxide. Magn Reson Imaging 11:645–654
Li W, Tutton S, Vu AT, et al. (2005) First-pass contrast-enhanced magnetic resonance angiography in humans using ferumoxytol, a novel ultrasmall superparamagnetic iron oxide (USPIO)-based blood pool agent. J Magn Reson Imaging 21:46–52
Lutz AM, Weishaupt D, Persohn E, et al. (2005) Imaging of macrophages in soft-tissue infection in rats: relationship between ultrasmall superparamagnetic iron oxide dose and MR signal characteristics. Radiology 234:765–775
Tanimoto A, Pouliquen D, Kreft BP, Stark DD (1994) Effects of spatial distribution on proton relaxation enhancement by particulate iron oxide. J Magn Reson Imaging 4:653–657
Weisskoff RM, Zuo CS, Boxerman JL, Rosen BR (1994) Microscopic susceptibility variation and transverse relaxation: theory and experiment. Magn Reson Med 31:601–610
Bulte JM, Vymazal J, Brooks RA, Pierpaoli C, Frank JA (1993) Frequency dependence of MR relaxation times. II. Iron oxides. J Magn Reson Imaging 3:641–648
Metz S, Bonaterra G, Rudelius M, Settles M, Rummeny EJ, Daldrup-Link HE (2004) Capacity of human monocytes to phagocytose approved iron oxide MR contrast agents in vitro. Eur Radiol 14:1851–1858
Beckmann N, Falk R, Zurbrugg S, Dawson J, Engelhardt P (2003) Macrophage infiltration into the rat knee detected by MRI in a model of antigen-induced arthritis. Magn Reson Med 49:1047–1055
Lutz AM, Seemayer C, Corot C, et al. (2004) Detection of synovial macrophages in an experimental rabbit model of antigen-induced arthritis: ultrasmall superparamagnetic iron oxide-enhanced MR imaging. Radiology 233:149–157
Pirko I, Ciric B, Johnson AJ, Gamez J, Rodriguez M, Macura S (2003) Magnetic resonance imaging of immune cells in inflammation of central nervous system. Croat Med J 44:463–468
Pirko I, Johnson A, Ciric B, et al. (2004) In vivo magnetic resonance imaging of immune cells in the central nervous system with superparamagnetic antibodies. FASEB J 18:179–182
Ruehm SG, Corot C, Vogt P, Kolb S, Debatin JF (2001) Magnetic resonance imaging of atherosclerotic plaque with ultrasmall superparamagnetic particles of iron oxide in hyperlipidemic rabbits. Circulation 103:415–422
Ruehm SG, Corot C, Vogt P, Cristina H, Debatin JF (2002) Ultrasmall superparamagnetic iron oxide-enhanced MR imaging of atherosclerotic plaque in hyperlipidemic rabbits. Acad Radiol 9(suppl 1):S143–S144
Schmitz SA, Taupitz M, Wagner S, Wolf KJ, Beyersdorff D, Hamm B (2001) Magnetic resonance imaging of atherosclerotic plaques using superparamagnetic iron oxide particles. J Magn Reson Imaging 14:355–361
Saleh A, Wiedermann D, Schroeter M, Jonkmanns C, Jander S, Hoehn M (2004) Central nervous system inflammatory response after cerebral infarction as detected by magnetic resonance imaging. NMR Biomed 17:163–169
Saleh A, Schroeter M, Jonkmanns C, Hartung HP, Modder U, Jander S (2004) In vivo MRI of brain inflammation in human ischaemic stroke. Brain 127:1670–1677
Rausch M, Sauter A, Frohlich J, Neubacher U, Radu EW, Rudin M (2001) Dynamic patterns of USPIO enhancement can be observed in macrophages after ischemic brain damage. Magn Reson Med 46:1018–1022
Kraitchman DL, Heldman AW, Atalar E, et al. (2003) In vivo magnetic resonance imaging of mesenchymal stem cells in myocardial infarction. Circulation 107:2290–2293
Himes N, Min JY, Lee R, et al. (2004) In vivo MRI of embryonic stem cells in a mouse model of myocardial infarction. Magn Reson Med 52:1214–1219
Daldrup-Link HE, Meier R, Rudelius M, et al. (2005) In vivo tracking of genetically engineered, anti-HER2/neu directed natural killer cells to HER2/neu positive mammary tumors with magnetic resonance imaging. Eur Radiol 15:4–13
Daldrup-Link HE, Rudelius M, Piontek G, et al. (2005) Migration of iron oxide-labeled human hematopoietic progenitor cells in a mouse model: in vivo monitoring with 1.5-T MR imaging equipment. Radiology 234:197–205
Daldrup-Link HE, Rudelius M, Oostendorp RA, et al. (2005) Comparison of iron oxide labeling properties of hematopoietic progenitor cells from umbilical cord blood and from peripheral blood for subsequent in vivo tracking in a xenotransplant mouse model (1). Acad Radiol 12:502–510
Majumdar S, Zoghbi S, Pope CF, Gore JC (1989) A quantitative study of relaxation rate enhancement produced by iron oxide particles in polyacrylamide gels and tissue. Magn Reson Med 9:185–202
Billotey C, Wilhelm C, Devaud M, Bacri JC, Bittoun J, Gazeau F (2003) Cell internalization of anionic maghemite nanoparticles: quantitative effect on magnetic resonance imaging. Magn Reson Med 49:646–654
Sjogren CE, Johansson C, Naevestad A, Sontum PC, Briley-Saebo K, Fahlvik AK (1997) Crystal size and properties of superparamagnetic iron oxide (SPIO) particles. Magn Reson Imaging 15:55–67
Jung CW, Jacobs P (1995) Physical and chemical properties of superparamagnetic iron oxide MR contrast agents: ferumoxides, ferumoxtran, ferumoxsil. Magn Reson Imaging 13:661–674
Kaim AH, Wischer T, O’Reilly T, et al. (2002) MR imaging with ultrasmall superparamagnetic iron oxide particles in experimental soft-tissue infections in rats. Radiology 225:808–814
Graziani-Bowering GM, Graham JM, Filion LG, et al. (1997) A quick, easy and inexpensive method for the isolation of human peripheral blood monocytes. J Immunol Methods 207:157–168
Roch A, Muller RN (1999) Theory of proton relaxation induced by supraparamagnetic particles. J Chem Phys 110:5403–5411
Kellar KE, Fujii DK, Gunther WH, et al. (2002) Important considerations in the design of iron oxide nanoparticles as contrast agents for TI-weighted MRI and MRA. Acad Radiol 9(suppl 1):S34–S37
Majumdar S, Zoghbi SS, Gore JC, et al. (1989) The influence of pulse sequence on the relaxation effects of superparamagnetic iron oxide contrast agents. Magn Reson Med 10:289–301
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by a seed grant from the Department of Radiology, University of California of San Francisco.
Rights and permissions
About this article
Cite this article
Simon, G.H., Bauer, J., Saborovski, O. et al. T1 and T2 relaxivity of intracellular and extracellular USPIO at 1.5T and 3T clinical MR scanning. Eur Radiol 16, 738–745 (2006). https://doi.org/10.1007/s00330-005-0031-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-005-0031-2