Abstract
The cape petrel (Daption capense) is an abundant procellariform in seabird assemblages attending fishing vessels in the Southwestern Atlantic Ocean. The bulk of extant information comes from surveys conducted during the breeding season at Antarctic and sub-Antarctic sites; however, most of their ecological aspects during the non-breeding season remain largely unknown. We performed on-board census and stable isotope analysis in blood and feathers from 30 adult petrels during the non-breeding season in waters off Argentina. Cape petrels attending ice trawlers showed a higher abundance during winter compared to other seasons. The lack of correlation of isotopic values between tissues suggests changes in the diet and foraging locations between the moulting period and the non-breeding season. Isotopic mixing models revealed low-trophic-level sources as the main contributors to this petrel’s diet while pelagic-demersal fish species targeted by fishing vessels showed the lower contribution. The discrepancies between methods could be a result of partial use of offal/discards or low use of these due to size/mechanic limitations. These findings reinforce the idea of zooplankton and small-pelagic fish facilitation during the hauling operatory and the relevance of combining different methods for a thorough understanding of prey-predator relationships.




Similar content being viewed by others
Data availability
The data that support the findings of this study are not openly available due to reasons of sensitivity and are available from the corresponding author upon reasonable request. Data are located in controlled access data storage at Instituto de Investigaciones Marinas y Costeras, IIMyC (UNMdP-CONICET).
References
Acha EM, Mianzan HW, Guerrero RA, Favero M, Bava J (2004) Marine fronts at the continental shelves of austral South America: physical and ecological processes. J Mar Syst 44:83–105. https://doi.org/10.1016/j.jmarsys.2003.09.005
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA+ for primer: guide to software and statistical methods. PRIMER-E Ltd, Plymouth
Bartumeus F, Giuggioli L, Louzao M, Bretagnolle V, Oro D, Levin SA (2010) Fishery discards impact on seabird movement patterns at regional scales. Curr Biol 20:215–222. https://doi.org/10.1016/j.cub.2009.11.073
Bearhop S, Phillips RA, McGill R, Cherel Y, Dawson DA, Croxall JP (2006) Stable isotopes indicate sex-specific and longer-term individual foraging specialisation in diving seabirds. Mar Ecol Prog Ser 311:157–164. https://doi.org/10.3354/meps311157
Beck JR (1969) Food, moult and age of first breeding in the Cape pigeon, Daption capensis Linnaeus. Brit Antarct Surv Bull 21:33–44
Bertellotti M, Yorio P (2000) Utilisation of fishery waste by Kelp Gulls attending coastal trawl and longline vessels in northern Patagonia, Argentina. Ornis Fenn 77:105–115
BirdLife International (2018) Daption capense. The IUCN red list of threatened species 2018: e.T22697879A132610612. https://doi.org/10.2305/IUCN.UK.2018-2.RLTS.T22697879A132610612.en. Accessed 22 Dec 2020
Bond AL, Jardine TD, Hobson KA (2016) Multi-tissue stable-isotope analyses can identify dietary specialization. Methods Ecol Evol 7:1428–1437. https://doi.org/10.1111/2041-210X.12620
Bovcon ND, Góngora ME, Marinao C, González-Zevallos D (2013) Catches composition and discards generated by hake Merluccius hubbsi and shrimp Pleoticus muelleri fisheries: a case of study in the high-sea ice trawlers of San Jorge Gulf, Chubut, Argentina. Rev Biol Mar Oceanogr 48:303–319. https://doi.org/10.4067/S0718-19572013000200010
Bugoni L, McGill RAR, Furness RW (2008) Effects of preservation methods on stable isotope signatures in bird tissues. Rapid Commun Mass Spectrom 22:2457–2462. https://doi.org/10.1002/rcm
Bugoni L, McGill RAR, Furness RW (2010) The importance of pelagic longline fishery discards for a seabird community determined through stable isotope analysis. J Exp Mar Biol Ecol 391:190–200. https://doi.org/10.1016/j.jembe.2010.06.027
Bugoni L, Griffiths K, Furness RW (2011) Sex-biased incidental mortality of albatrosses and petrels in longline fisheries: differential distributions at sea or differential access to baits mediated by sexual size dimorphism? J Ornithol 152:261–268. https://doi.org/10.1007/s10336-010-0577-x
CCAMLR (2000) Commission for the Conservation of Antarctic Marine Living Resources. Report of the Ad Hoc Working Group on Incidental Mortality Associated With Fishing. Hobart, Australia. https://www.ccamlr.org/en/ad%20hoc%20wg-imaf-00. Accessed 30 May 2022
CCAMLR (2008) Commission for the Conservation of Antarctic Marine Living Resources. Report of the Working Group on Incidental Mortality Associated With Fishing. Hobart, Australia. https://www.ccamlr.org/en/wg-imaf-08. Accessed 30 May 2022
CCAMLR (2009) Commission for the Conservation of Antarctic Marine Living Resources. Report of the Working Group on Incidental Mortality Associated With Fishing. Hobart, Australia. https://www.ccamlr.org/en/wg-imaf-09. Accessed 30 May 2022
CCAMLR (2011) Commission for the Conservation of Antarctic Marine Living Resources. Report of the Working Group on Incidental Mortality Associated With Fishing. Hobart, Australia. https://www.ccamlr.org/en/wg-imaf-11. Accessed 30 May 2022
Ceia FR, Cherel Y, Seco J, Barbosa A, Chipev N, Xavier JC (2021) Variability in tissue-specific trophic discrimination factors (Δ13C and Δ15N) between Antarctic krill Euphausia superba and free-ranging Pygoscelis penguins. Polar Biol 44:1541–1551
Cherel Y (2008) Isotopic niches of emperor and Adélie penguins in Adélie Land, Antarctica. Mar Biol 154:813–821. https://doi.org/10.1007/s00227-008-0974-3
Cherel Y, Hobson KA (2007) Geographical variation in carbon stable isotope signatures of marine predators: a tool to investigate their foraging areas in the Southern Ocean. Mar Ecol Prog Ser 329:281–287. https://doi.org/10.3354/meps329281
Cherel Y, Hobson KA, Hassani S (2005) Isotopic discrimination between food and blood and feathers of captive penguins: implications for dietary studies in the wild. Physiol Biochem Zool 78:106–115. https://doi.org/10.1086/425202
Cherel Y, Phillips RA, Hobson KA, McGill R (2006) Stable isotope evidence of diverse species-specific and individual wintering strategies in seabirds. Biol Lett 2:301–303. https://doi.org/10.1098/rsbl.2006.0445
Cherel Y, Hobson KA, Guinet C, Vanpe C (2007) Stable isotopes document seasonal changes in trophic niches and winter foraging individual specialization in diving predators from the Southern Ocean. J Anim Ecol 76:826–836. https://doi.org/10.1111/j.1365-2656.2007.01238.x
Cherel Y, Jaeger A, Alderman R, Jaquemet S, Richard P, Wanless RM et al (2013) A comprehensive isotopic investigation of habitat preferences in nonbreeding albatrosses from the Southern Ocean. Ecography 36:277–286. https://doi.org/10.1111/j.1600-0587.2012.07466.x
Cherel Y, Jaquement S, Maglio A, Jaeger A (2014a) Differences in δ13C and δ15N values between feathers and blood of seabird chicks: implications for non-invasive isotopic investigations. Mar Biol 161:229–237. https://doi.org/10.1007/s00227-013-2314-5
Cherel Y, Connan M, Jaeger A, Richard P (2014b) Seabird year-round and historical feeding ecology: blood and feather δ13C and δ15N values document foraging plasticity of small sympatric petrels. Mar Ecol Prog Ser 505:267–280. https://doi.org/10.3354/meps10795
Cherel Y, Quillfeldt P, Delord K, Weimerskirch H (2016) Combination of at-sea activity, geolocation and feathers isotopes documents when and where seabirds molt. Front Ecol Evol 4:3. https://doi.org/10.3389/fevo.2016.00003
Ciancio JE, Pascual MA, Botto F, Frere E, Iribarne O (2008) Trophic relationships of exotic anadromous salmonids in the southern Patagonian Shelf as inferred from stable isotopes. Limnol Oceanogr 53:788–798. https://doi.org/10.4319/lo.2008.53.2.0788
Clarke KR, Gorley RN (2006) PRIMER v6: user manual/tutorial. PRIMER-E Ltd, Plymouth
Copello S, Quintana F, Pérez F (2008) Diet of the southern giant petrel in Patagonia: fishery-related items and natural prey. Endanger Species Res 6:15–23. https://doi.org/10.3354/esr00118
Copello S, Seco Pon JP, Favero M (2013) Use of marine space by Black-browed albatrosses during the non-breeding season in the Southwest Atlantic Ocean. Estuar Coast Shelf Sci 123:34–38. https://doi.org/10.1016/j.ecss.2013.02.016
Coria N, Soave G, Montalti D (1997) Diet of Cape petrel Daption capense during the post-hatching period at Laurie Island, South Orkney Islands, Antarctica. Polar Biol 18:236–239
Creet S, Van Franeker JA, Van Spanje TM, Wolff WJ (1994) Diet of the pintado petrel Daption capense at king George Island, Antactica, 1990/1991. Mar Ornithol 22:221–229
Croxall JP, Wood AG (2002) The importance of the Patagonian Shelf for top predator species breeding at South Georgia. Aquat Conserv Mar Freshw Ecosyst 12:101–118. https://doi.org/10.1002/aqc.480
Dehnhard N, Achurch H, Clarke J et al (2020) High inter-and intraspecific niche overlap among three sympatrically breeding, closely related seabird species: generalist foraging as an adaptation to a highly variable environment? J Anim Ecol 89:104–119. https://doi.org/10.1111/1365-2656.13078
del Hoyo J, Elliot A, Sargatal J (1992) Handbook of the birds of the world. Vol. 1. Lynx editions. Barcelona, Spain
Delord K, Pinet P, Pinaud D et al (2016) Species-specific foraging strategies and segregation mechanisms of sympatric Antarctic fulmarine petrels throughout the annual cycle. Ibis 158:569–586. https://doi.org/10.1111/ibi.12365
Dias MP, Martin R, Pearmain EJ et al (2019) Threats to seabirds: a global assessment. Biol Conserv 237:525–537. https://doi.org/10.1016/j.biocon.2019.06.033
Eder EB, Lewis MN, Campagna C, Koch PL (2010) Evidence of demersal foraging from stable isotope analysis of juvenile elephant seals from Patagonia. Mar Mamm Sci 26:430–442. https://doi.org/10.1111/j.1748-7692.2009.00347.x
Favero M, Silva Rodriguez MP (2005) Estado actual y conservación de aves pelágicas que utilizan la plataforma continental argentina como área de alimentación. El Hornero, 20:95110. Retrieved from http://www.scielo.org.ar/scielo.php?pid=S0073-34072005000100007&script=sci_arttext
Favero M, Blanco G, García G et al (2011) Seabird mortality associated with ice trawlers in the Patagonian shelf: effect of discards on the occurrence of interactions with fishing gear. Anim Conserv 14:131–139. https://doi.org/10.1111/j.1469-1795.2010.00405.x
Favero M, Blanco G, Copello S, Seco Pon JP, Paterlini C, Mariano-Jelicich R, García G, Berón MP (2013) Seabird bycatch in the Argentinean demersal longline fishery, 2001–2010. Endanger Species Res 19:187–199. https://doi.org/10.3354/esr00478
Fijn RC, Van Franeker JA, Trathan PN (2012) Dietary variation in chick-feeding and self-provisioning Cape Petrel Daption capense and Snow Petrel Pagodroma nivea at Signy Island, South Orkney Islands, Antarctica. Mar Ornithol 40:81–87
Finkelstein M, Bakker V, Doak DF, Sullivan B, Lewison R, Satterthwaite WH, McIntyre PB, Wolf S, Priddel D, Arnold JM, Henry RW, Sievert P, Croxall J (2008) Evaluating the potential effectiveness of compensatory mitigation strategies for marine bycatch. PLoS ONE 3:e2480. https://doi.org/10.1371/journal.pone.0002480
Forero MG, Bortolotti GR, Hobson KA, Donazar JA, Bertelloti M, Blanco G (2004) High trophic overlap within the seabird community of Argentinean Patagonia: a multiscale approach. J Anim Ecol 74:789–801
Forero MG, González-Solís J, Hobson KA, Donázar JA, Bertellotti M, Blanco G, Bortolotti GR (2005) Stable isotopes reveal trophic segregation by sex and age in the southern giant petrel in two different food webs. Mar Ecol Prog Ser 296:107–113
Fox J, Weisberg S (2011) An R companion to applied regression, 2nd edn. Sage, Thousand Oaks
Fridolfsson AK, Ellegren H (1999) A simple and universal method for molecular sexing non-ratite birds. J Avian Biol 30:116–121. https://doi.org/10.2307/3677252
Gandini P, Seco Pon JP (2007) Seabird assemblages attending longline vessels in the Argentinean Economic Exclusive Zone. Ornitol Neotrop 18:553–561
Garthe S, Camphuysen KCJ, Furness RW (1996) Amounts of discards by commercial fisheries and their significance as food for seabirds in the North Sea. Mar Ecol Prog Ser 136:1–11
Giménez J, Arneill GE, Bennison A, Pirotta E, Gerritsen HD, Bodey TW, Bearhop S, Hamer KC, Votier S, Mark Jessopp M (2021) Sexual mismatch between vessel-associated foraging and discard consumption in a marine top predator. Front Mar Sci 8:636468. https://doi.org/10.3389/fmars.2021.636468
Gonzalez-Zevallos D, Yorio P (2011) Consumption of discards and interactions between Black-browed Albatrosses (Thalassarche melanophrys) and Kelp Gulls (Larus dominicanus) at trawl fisheries in Golfo San Jorge, Argentina. J Ornithol 152:827–838. https://doi.org/10.1007/s10336-011-0657-6
Granadeiro JP, Brickle P, Catry P (2014) Do individual seabirds specialize in fisheries’ waste? The case of black-browed albatrosses foraging over the Patagonian Shelf. Anim Conserv 17:19–26. https://doi.org/10.1111/acv.12050
Grémillet AJ, Wilkinson S, Crawford RJM, Ryan PG (2008) A junk food hypothesis for gannets feeding on fishery waste. Proc R Soc B 275:1149–1156. https://doi.org/10.1098/rspb.2007.1763
Harrison P (1983) Seabirds: an identification guide. Houghton Mifflin, Boston
Heinemann D (1981) A range-finder for pelagic bird censusing. J Wildl Manag 45:489–493
Hobson KA, Gibbs HL, Gloutney ML (1997) Preservation of blood and tissue samples for stable-carbon and stable-nitrogen isotope analysis. Can J Zool 75:1720–1723
Hodum PJ, Hobson KA (2000) Trophic relationships among Antarctic fulmarine petrels: insights into dietary overlap and chick provisioning strategies inferred from stable-isotope (δ15N and δ13C) analyses. Mar Ecol Prog Ser 198:273–281. https://doi.org/10.3354/meps198273
Inger R, Bearhop R (2008) Applications of stable isotope analyses to avian ecology. Ibis 150:447–461. https://doi.org/10.1111/j.1474-919X.2008.00839.x
Jaeger A, Connan M, Richard P, Cherel Y (2010a) Use of stable isotopes to quantify seasonal changes of trophic niche and levels of population and individual specialisation in seabirds. Mar Ecol Prog Ser 401:269–277. https://doi.org/10.3354/meps08380
Jaeger A, Lecomte VJ, Weimerskirch H, Richard P, Cherel Y (2010b) Seabird satellite tracking validates the use of latitudinal isoscapes to depict predators’ foraging areas in the Southern Ocean. Rapid Commun Mass Spectrom 24:3456–3460. https://doi.org/10.1002/rcm.4792
Jiménez S, Domingo A, Abreu M, Brazeiro A (2011) Structure of the seabird assemblage associated with pelagic longline vessels in the Southwestern Atlantic: implications for bycatch. Endanger Species Res 15:241–254. https://doi.org/10.3354/esr00378
Juares MA, Santos M, Mennucci JA, Coria NR, Mariano-Jelicich R (2016) Diet composition and foraging habitats of Adélie and gentoo penguins in three different stages of their annual cycle. Mar Biol 163:105. https://doi.org/10.1007/s00227-016-2886-y
Kelly B, Dempson JB, Power M (2006) The effects of preservation on fish tissue stable isotope signatures. J Fish Biol 69:1595–1611. https://doi.org/10.1111/j.1095-8649.2006.01226.x
Lourey MJ, Trull TW, Sigman DM (2003) Sensitivity of d15N of nitrate, surface suspended and deep sinking particulate nitrogen to seasonal nitrate depletion in the Southern Ocean. Global Biogeochem Cycles 17:1081. https://doi.org/10.1029/2002GB001973
Magozzi S, Yool A, Vander Zanden HB, Wunder MB, Trueman CN (2017) Using ocean models to predict spatial and temporal variation in marine carbon isotopes. Ecosphere 8:e01763. https://doi.org/10.1002/ecs2.1763
Mariano-Jelicich R, Copello S, Seco Pon JP, Favero M (2014) Contributionof fishery discards to the diet of the Black-browed albatross (Thalassarche melanophris) during the non-breeding season: an assessment through stable isotope analysis. Mar Biol 161:119–129. https://doi.org/10.1007/s00227-013-2320-7
Michalik A, McGill RAR, Furness RW, Eggers T, van Noordwijk HJ, Quillfeldt P (2010) Black and white—does melanin change the bulk carbon and nitrogen isotope values of feathers? Rapid Commun Mass Spectrom 24:875–878. https://doi.org/10.1002/rcm.4462
Moreno R, Jover L, Velando A, Munilla I, Sanpera C (2011) Influence of trophic ecology and spatial variation on the isotopic fingerprints of seabirds. Mar Ecol Prog Ser 442:229–239. https://doi.org/10.3354/meps09420
Onley DJ, Scofield RP (2007) Albatrosses, petrels and shearwaters of the world. Helm Publisher Ltd., London
Parnell AC, Inger R, Bearhop S, Jackson AL (2010) Source partitioning using stable isotopes: coping with too much variation. PLoS ONE 5:e9672. https://doi.org/10.1371/journal.pone.0009672
Phillips RA, Bearhop S, McGill RAR, Dawson DA (2009) Stable isotopes reveal individual variation in migration strategies and habitat preferences in a suite of seabirds during the nonbreeding period. Oecologia 160:795–806. https://doi.org/10.1007/s00442-009-1342-9
Polito MJ, Trivelpiece WZ, Karnovsky NJ, Ng E, Patterson WP, Emslie SE (2011) Integrating stomach content and stable isotope analyses to quantify the diets of Pygoscelid Penguins. PLoS ONE 6:e26642. https://doi.org/10.1371/journal.pone.0026642
Pollet IL, Hedd A, Taylor PD, Montevecchi WA, Shutler D (2014) Migratory movements and wintering areas of Leach’s Storm-Petrels tracked using geolocators. J Field Ornithol 85:321–328. https://doi.org/10.1111/jofo.12071
Post DM (2002) Using stable isotopes to estimate trophic position: models. Methods and assumptions. Ecology 83:703–718. https://doi.org/10.1890/0012-9658(2002)083[0703:USITET]2.0.CO;2
Quillfeldt P, McGill RAR, Furness RW (2005) Diet and foraging areas of Southern Ocean seabirds and their prey inferred from stable isotopes: review and case study of Wilson’s storm-petrel. Mar Ecol Prog Ser 295:295–304. https://doi.org/10.3354/meps295295
Quillfeldt P, McGill RAR, Masello JF, Weiss F, Strange IJ, Brickle P, Furness RW (2008) Stable isotope analysis reveals sexual and environmental variability and individual consistency in foraging of thin-billed prions. Mar Ecol Prog Ser 373:137–148. https://doi.org/10.3354/meps07751
Quillfeldt P, Masello JF, McGill RAR, Adams M, Furness RW (2010) Moving polewards in winter: a recent change in the migratory strategy of a pelagic seabird? Front Zool 7:15. https://doi.org/10.1186/1742-9994-7-15
Quillfeldt P, Moodley Y, Weimerskirch H, Cherel Y, Delord K, Phillips RA, Navarro J, Calderón L, Masello JF (2017) Does genetic structure reflect differences in non-breeding movements? A case study in small, highly mobile seabirds. BMC Evol Biol 17:160. https://doi.org/10.1186/s12862-017-1008-x
Quintana F, Dell’Arciprete OP, Copello S (2010) Foraging behavior and habitat use by the Southern Giant Petrel on the Patagonian Shelf. Mar Biol 157:515–525. https://doi.org/10.1007/s00227-009-1337-4
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Ramos R, Ramirez I, Paiva VH, Militao T, Biscoito M, Menezes D, Phillips RA, Zino F, Gonzalez-Solis J (2016) Global spatial ecology of three closely-related gadfly petrels. Sci Rep 6:23447. https://doi.org/10.1038/srep23447
Ramos R, Carlile N, Madeiros J, Ramírez I, Paiva VH, Dinis HA, Zino F, Biscoito M, Leal GR, Bugoni L, Jodice PGR, Ryan PG, Gonzalez-Solís J (2017) It is the time for oceanic seabirds: tracking year-round distribution of gadfly petrels across the Atlantic Ocean. Divers Distrib 23:794–805. https://doi.org/10.1111/ddi.12569
Raya Rey A, Polito M, Archuby D, Coria N (2012) Stable isotopes identify age- and sex-specific dietary partitioning and foraging habitat segregation in southern giant petrels breeding in Antarctica and southern Patagonia. Mar Biol 159:1317–1326. https://doi.org/10.1007/s00227-012-1912-y
Riccialdelli L, Newsome SD, Fogel ML, Goodall RNP (2010) Isotopic assessment of prey and habitat preferences of a cetacean community in the southwestern South Atlantic Ocean. Mar Ecol Prog Ser 418:235–248. https://doi.org/10.3354/meps08826
Rodriguez A, Arcos JM, Bretagnolle V, Dias MP, Holmes ND, Louzao M et al (2019) Future directions in conservation research on petrels and shearwaters. Front Mar Sci 6:94. https://doi.org/10.3389/fmars.2019.00094
Schreiber EA, Burger J (2002) Biology of marine birds. CRC Press
Seco Pon JP (2014) Asociación de aves marinas pelágicas a la flota argentina de arrastre de altura: caracterización integral de las interacciones y desarrollo de una estrategia de conservación para especies con estado de conservación amenazado. Tesis Doctoral Dissertation. Universidad Nacional de Mar del Plata.
Seco Pon JP, Copello S, Tamini L, Mariano-Jelicich R, Paz J, Blanco G, Favero M (2015) Seabird conservation in fisheries: current state of knowledge and conservation needs for Argentine high seas fleets. In: Mahala G (ed) Seabirds and songbirds. Habitat preferences, conservation and migratory behaviors. Nova publishers, New York, pp 45–88
Shirihai H (2008) The complete guide to Antarctic wildlife, vol 2. Princeton University Press, Princeton
Smith JA, Mazumder D, Suthers IM, Taylor MD (2013) To fit or not to fit: evaluating stable isotope mixing models using simulated mixing polygons. Methods Ecol Evol 4:612–618. https://doi.org/10.1111/2041-210X.12048
Soave GE, Coria N, Silva P, Montalti D, Favero M (2000) Diet of cape petrel daption capense chicks on South Shetland Islands, Antarctica. Acta Ornithol 35:191–195
Stock BC, Jackson AL, Ward EJ, Parnell AC, Phillips DL, Semmens BX (2018) Analyzing mixing systems using a new generation of Bayesian tracer mixing models. PeerJ 6:e5096. https://doi.org/10.7717/peerj.5096
Stowasser G, Atkinson A, McGill RAR, Phillips RA, Collins MA, Pond DW (2012) Food web dynamics in the Scotia Sea in summer: a stable isotope study. Deep-Sea Res II: Top Stud Oceanogr 59–60:208–221. https://doi.org/10.1016/j.dsr2.2011.08.004
Tamini LL, Chavez LN, Góngora ME, Yates O, Rabuffetti FL, Sullivan B (2015) Estimating mortality of black-browed albatross (Thalassarche melanophris, Temminck, 1828) and other seabirds in the Argentinean factory trawl fleet and the use of bird-scaring lines as a mitigation measure. Pol Biol 38:1867–1879. https://doi.org/10.1007/s00300-015-1747-3
Tringali LS (2012) Biología y pesca de la merluza del Mar Argentino. Mar del Plata: Instituto Nacional de Investigación y Desarrollo Pesquero INIDEP, Argentina
Vander Zander MJ, Clayton MK, Moody EK, Solomon CT, Weidel BC (2015) Stable isotope turnover and half-life in animal tissues: a literature synthesis. PLoS ONE 10:e0116182. https://doi.org/10.1371/journal.pone.0116182
Weidinger K (1996) Patterns of colony attendance in the Cape Petrel Daption capense at Nelson Island, South Shetland Islands, Antarctica. Ibis 138:243–249. https://doi.org/10.1111/j.1474-919X.1996.tb04335.x
Yorio P, Caille G (1999) Seabird interactions with coastal fisheries in Northern Patagonia: use of discards and incidental captures in nets. Waterbirds 22:207–216. https://doi.org/10.2307/1522209
Zar JH (2010) Biostatistical analysis, 5th edn. Pearson Prentice-Hall, Upper Saddle River
Acknowledgements
We gratefully acknowledge the captain and the crew of the fishing vessel ‘Ur-ertza’ and Lic. Gabriel Blanco from the National Observers Program (INIDEP) for facilitating the vessel access. P. Lértora, J.P. Romanelli, and observers of the National Observers Program (INIDEP) for helping during sampling potential prey. We appreciate four anonymous reviewers and the editor for their suggestions and comments that hugely improved the manuscript.
Funding
This study was funded by the National Agency for the Promotion of Science and Technology (Agencia Nacional de Promoción Científica y Tecnológica, PICT 2008-0590 and PICT 2017-1761), the National Research Council (PIP CONICET 00070), and the National University of Mar del Plata (Argentina).
Author information
Authors and Affiliations
Contributions
RMJ and MF designed the study. SC and JPSP participated in the field studies. RMJ and JPSP analysed the data. RMJ produced the first draft of the paper, to which all other authors contributed.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This study was conducted under approved animal use protocols and permits provided by the Argentinean Government.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
300_2023_3144_MOESM1_ESM.eps
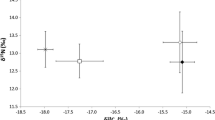
ESM 1 Simulated mixing regions for the biplots in Fig 2. (a) Antarctic subset, (b) Patagonic subset. Probability contours are at the 5% level (outermost contour) and at every 10% level. The positions of individual cape petrels (solid circles) and the average source values (crosses) are shown (EPS 1051 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mariano-Jelicich, R., Seco Pon, J.P., Copello, S. et al. Distribution and diet of cape petrels (Daption capense) attending fishing vessels off the Patagonian Continental Shelf during the non-breeding season in austral winter: insights from on-board censuses and stable isotope analysis. Polar Biol 46, 559–571 (2023). https://doi.org/10.1007/s00300-023-03144-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-023-03144-6