Abstract
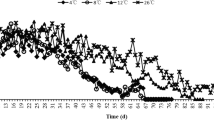
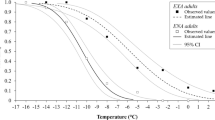
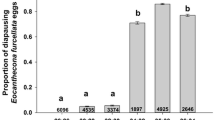
Temperatures in Arctic and sub-Arctic regions are highly variable, with long, and cold winters, and short summers. Studies on thermal biology of ectotherms from these latitudes have mostly focused on how organisms cope with cold temperatures. However, temperatures in microhabitats such as south-facing slopes can occasionally reach high and potentially stressful temperatures. Such microclimatic conditions are challenging for organisms, since they need to time their development to coincide with the short summer, while coping with thermal extremes during summer and winter. In the present study, we investigate upper and lower thermal limits of adults and eggs in Nysius groenlandicus, a univoltine seed bug widely distributed in Greenland and alpine areas of Scandinavia and with preference for dry and sunny habitats. Secondly, we investigate the survival strategy of the overwintering egg stage during cold exposure. We found a clear association between the microhabitat temperatures experienced by each specific life stage and their heat and cold tolerance. Thus, adults and eggs, which are both exposed to high summer temperatures, showed a high heat tolerance. Adults which do not overwinter had very poor cold tolerance, whereas eggs produced in autumn were very cold hardy (with some survival at − 21 °C). Cold tolerance of the eggs of N. groenlandicus was based on freeze avoidance through extensive supercooling and by conserving water. These results suggest life stage-specific adaptation to heat and cold stress in Arctic and sub-Arctic insects, and highlight new research avenues addressing adaptation to higher and more variable temperatures in Arctic and sub-Arctic regions.


Similar content being viewed by others
Availability of data and materials
Data are available from authors on request.
References
Alemu T, Alemneh T, Pertoldi C et al (2017) Costs and benefits of heat and cold hardening in a soil arthropod. Biol J Linn Soc 122:765–773. https://doi.org/10.1093/biolinnean/blx092/4243554
Allen JL, Chown SL, Janion-scheepers C, Clusella-trullas S (2016) Interactions between rates of temperature change and acclimation affect latitudinal patterns of warming tolerance. Conserv Physiol 4:1–14
Anthony SE, Buddle CM, Høye TT, Sinclair BJ (2019) Thermal limits of summer-collected Pardosa wolf spiders (Araneae: Lycosidae) from the Yukon Territory (Canada) and Greenland. Polar Biol 42:2055–2064. https://doi.org/10.1007/s00300-019-02580-7
Ávila-Jiménez ML, Coulson SJ, Solhøy T, Sjöblom A (2010) Overwintering of terrestrial Arctic arthropods: the fauna of Svalbard now and in the future. Polar Res 29:127–137. https://doi.org/10.1111/j.1751-8369.2009.00141.x
Bahrndorff S, Gertsen S, Pertoldi C, Kristensen TN (2016) Investigating thermal acclimation effects before and after a cold shock in Drosophila melanogaster using behavioural assays. Biol J Linn Soc 117:241–251
Block W (1994) Differential scanning calorimetry in ecophysiological research. Acta Oecol 15:13–22
Böcher J (1972) Feeding biology of Nysius groenlandicus (Zett.) (Heteroptera: Lygaeidae) in Greenland, with a note on oviposition in relation to food source and dispersal of the species. Meddelelser om Grønl 191:1–41
Böcher J (1975) Notes on the reproductive biology and egg-diapause in Nysius groenlandicus (Heteroptera: Lygaeidae). Vidensk Meddelelser fra Dansk Naturhistorisk Foren 138:21–38
Böcher J (1976) Population studies on Nysius groenlandicus (Zett.) (Heteroptera: Lygaeidae) in Greenland, with particular reference to climatic factors, especially the snow cover. Vidensk Meddelelser fra Dansk naturhistorisk Foren 139:61–89
Böcher J, Nachman G (2001) Temperature and humidity responses of the arctic-alpine seed bug Nysius groenlandicus. Entomol Exp Appl 99:319–330
Böcher J, Nachman G (2011) Coexistence of bisexual and unisexual populations of Nysius groenlandicus in the Zackenberg Valley, Northeast Greenland. Entomol Exp Appl 140:196–206. https://doi.org/10.1111/j.1570-7458.2011.01153.x
Bowler K, Terblanche JS (2008) Insect thermal tolerance: what is the role of ontogeny, ageing and senescence? Biol Rev 83:339–355. https://doi.org/10.1111/j.1469-185X.2008.00046.x
Convey P, Coulson SJ, Worland MR, Sjöblom A (2018) The importance of understanding annual and shorter-term temperature patterns and variation in the surface levels of polar soils for terrestrial biota. Polar Biol 41:1587–1605. https://doi.org/10.1007/s00300-018-2299-0
Costanzo JP, Lee RE (2013) Avoidance and tolerance of freezing in ectothermic vertebrates. J Exp Biol 216:1961–1967. https://doi.org/10.1242/jeb.070268
Coulson SJ, Hodkinson ID, Block W et al (1995) Low summer temperatures: a potential mortality factor for high arctic soil microarthropods? J Insect Physiol 41:783–792. https://doi.org/10.1016/0022-1910(95)00027-R
Danks HV (2004) Seasonal adaptations in Arctic insects. Integr Comp Biol 44:85–94. https://doi.org/10.1093/icb/44.2.85
Danks HV, Kukal O, Ring RA (1994) Insect cold-hardiness: insights from the Arctic. Arctic 47:391–404. https://doi.org/10.1116/1.591323
Denlinger DL, Lee RE (eds) (2010) Low temperature biology of insects. Cambridge University Press, Cambridge
Deutsch CA, Tewksbury JJ, Huey RB et al (2008) Impacts of climate warming on terrestrial ectotherms across latitude. Proc Natl Acad Sci 105:6668–6672. https://doi.org/10.1073/pnas.0709472105
Everatt MJ, Bale JS, Convey P et al (2013) The effect of acclimation temperature on thermal activity thresholds in polar terrestrial invertebrates. J Insect Physiol 59:1057–1064. https://doi.org/10.1016/j.jinsphys.2013.08.003
Goto SG, Yoshida KM, Kimura MT (1998) Accumulation of Hsp70 mrna under environmental stresses in diapausing and nondiapausing adults of Drosophila triauraria. J Insect Physiol 44:1009–1015. https://doi.org/10.1016/S0022-1910(97)00143-1
Graham E, Rundel P, Kaiser W et al (2012) Fine-scale patterns of soil and plant surface temperatures in an alpine fellfield habitat, white mountains, California. Arctic Antarct Alp Res 44:288–295. https://doi.org/10.1657/1938-4246-44.3.288
Hao S, Kang L (2014) Supercooling capacity and cold hardiness of the eggs of the grasshopper Chorthippus fallax (Orthoptera: Acrididae). J Insect Sci 14:231–236. https://doi.org/10.1093/jisesa/ieu151
Hodkinson ID, Coulson SJ, Webb NR, Block W (1996) Can high Arctic soil microarthropods survive eleveated summer temperatures? Funct Ecol 10:314–321
Holmstrup M (2014) The ins and outs of water dynamics in cold tolerant soil invertebrates. J Therm Biol 45:117–123. https://doi.org/10.1016/j.jtherbio.2014.09.001
Holmstrup M (2018) The springtail Megaphorura arctica survives extremely high osmolality of body fluids during drought. J Comp Physiol B Biochem Syst Environ Physiol 188:939–945. https://doi.org/10.1007/s00360-018-1180-3
Holmstrup M, Bayley M, Ramløv H (2002) Supercool or dehydrate? An experimental analysis of overwintering strategies in small permeable arctic invertebrates. Proc Natl Acad Sci USA 99:5716–5720. https://doi.org/10.1073/pnas.082580699
Høye TT, Culler LE (2018) Tundra arthropods provide key insights into ecological responses to environmental change. Polar Biol 41:1523–1529. https://doi.org/10.1007/s00300-018-2370-x
IPCC (2014) Climate Change 2013
Jaco KC, Chown SL (1997) Critical thermal limits, temperature tolerance and water balance of a sub-Antarctic caterpillar, Pringleophaga marioni (Lepidoptera: Tineidae). J Insect Physiol 43:685–694
Jensen A, Alemu T, Alemneh T et al (2019) Thermal acclimation and adaptation across populations in a broadly distributed soil arthropod. Funct Ecol 33:833–845. https://doi.org/10.1111/1365-2435.13291
Kostal V (2006) Eco-physiological phases of insect diapause. J Insect Physiol 52:113–127
Loeschcke V, Hoffmann AA (2007) Consequences of heat hardening on a field fitness component in Drosophila depend on environmental temperature. Am Nat 169:175–183. https://doi.org/10.1086/510632
MacLean HJ, Kristensen TN, Overgaard J et al (2017) Acclimation responses to short-term temperature treatments during early life stages causes long lasting changes in spontaneous activity of adult Drosophila melanogaster. Physiol Entomol 42:404–411. https://doi.org/10.1111/phen.12212
Marshall KE, Sinclair BJ (2010) Repeated stress exposure results in a survival-reproduction trade-off in Drosophila melanogaster. Proc R Soc B Biol Sci 277:963–969. https://doi.org/10.1098/rspb.2009.1807
Noer NK, Pagter M, Bahrndorff S et al (2020) Impacts of thermal fluctuations on heat tolerance and its metabolomic basis in Arabidopsis thaliana, Drosophila melanogaster, and Orchesella cincta. PLoS ONE 15:1–20. https://doi.org/10.1371/journal.pone.0237201
Overgaard J, Kristensen TN, Sørensen JG (2012) Validity of thermal ramping assays used to assess thermal tolerance in arthropods. PLoS ONE 7:1–7. https://doi.org/10.1371/journal.pone.0032758
Rinehart JP, Yocum GD, Denlinger DL (2000) Developmental upregulation of inducible hsp70 transcripts, but not the cognate form, during pupal diapause in the flesh fly, Sarcophaga crassipalpis. Insect Biochem Mol Biol 30:515–521. https://doi.org/10.1016/S0965-1748(00)00021-7
Sinclair BJ, Addo-Bediako A, Chown SL (2003) Climatic variability and the evolution of insect freeze tolerance. Biol Rev 78:181–195
Sømme L (1982) Supercooling and winter survival in terrestrial arthropods. Comp Biochem Physiol Part A Physiol 73:519–543. https://doi.org/10.1016/0300-9629(82)90260-2
Sørensen JG, Loeschcke V, Kristensen TN (2013) Cellular damage as induced by high temperature is dependent on rate of temperature change—investigating consequences of ramping rates on molecular and organismal phenotypes in Drosophila melanogaster. J Exp Biol 216:809–814. https://doi.org/10.1242/jeb.076356
Sørensen MH, Kristensen TN, Lauritzen JMS et al (2019) Rapid induction of the heat hardening response in an Arctic insect. Biol Lett 15:20190613. https://doi.org/10.1098/rsbl.2019.0613
Strathdee AT, Howling GG, Bale JS (1995) Cold hardiness of overwintering aphid eggs. J Insect Physiol 41:653–657. https://doi.org/10.1016/0022-1910(95)00029-T
Teets NM, Denlinger DL (2013) Physiological mechanisms of seasonal and rapid cold-hardening in insects. Physiol Entomol 38:105–116. https://doi.org/10.1111/phen.12019
Terblanche JS, Hoffmann AA, Mitchell KA et al (2011) Ecologically relevant measures of tolerance to potentially lethal temperatures. J Exp Biol 214:3713–3725. https://doi.org/10.1242/jeb.061283
Yocum GD, Joplin KH, Denlinger DL (1998) Upregulation of a 23 kDa small heat shock protein transcript during pupal diapause in the flesh fly, Sarcophaga crassipalpis. Insect Biochem Mol Biol 28:677–682. https://doi.org/10.1016/S0965-1748(98)00046-0
Acknowledgements
We would like to thank Helle Blendstrup for taking care of the animals during the experiments.
Funding
We thank the Danish Council for Independent Research (Grant No. DFF-8021-00014B) (T.N.K.), the Carlsberg Foundation (CF17-0415) (S.B.), and North2North (S.B.; T.N.K.). for funding.
Author information
Authors and Affiliations
Contributions
S.B., T.N.K., A.T., M.H.S., and M.H. designed the experiments. S.B., T.N.K., M.H., A.T., T.T.H., and M.H.S. performed the experiments. S.B. and M.H. analyzed the data and interpreted the results. S.B. wrote the first version of the manuscript. All authors contributed to revision of the manuscript and approved the final version. All authors agree to be held accountable for the content of this manuscript. We thank Michael Elnitsky and two anonymous reviewers for invaluable suggestions and help in improving the review.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bahrndorff, S., Alemu, T., Kristensen, T.N. et al. Thermal adaptations of adults and eggs in the Arctic seed bug Nysius groenlandicus (Insecta: Hemiptera) from South Greenland. Polar Biol 44, 491–498 (2021). https://doi.org/10.1007/s00300-021-02807-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-021-02807-6