Abstract
During the last few decades, the Arctic has experienced large-scale vegetation changes. Understanding the mechanisms behind this vegetation change is crucial for our ability to predict future changes. This study tested the hypothesis that decreased cryogenic disturbances cause vegetation change in patterned ground study fields (non-sorted circles) in Abisko, Sweden during the last few decades. The hypothesis was tested by surveying the composition of plant communities across a gradient in cryogenic disturbance and by reinvestigating plant communities previously surveyed in the 1980s to scrutinise how these communities changed in response to reduced cryogenic disturbance. Whereas the historical changes in species occurrence associated with decreased cryogenic disturbances were relatively consistent with the changes along the contemporary gradient of cryogenic disturbances, the species abundance revealed important transient changes highly dependent on the initial plant community composition. Our results suggest that altered cryogenic disturbances cause temporal changes in vegetation dynamics, but the net effects on vegetation communities depend on the composition of initial plant species.
Similar content being viewed by others
Introduction
Periglacial environments cover around 25% of the Earth’s land surface (French 2007). The intensity of disturbance generated in these regions due to repeated freezing and thawing processes in the soil is predicted to change due to the ongoing climate warming (Aalto et al. 2014). In addition, climate warming is expected to influence the vegetation abundance and composition in arctic and subarctic areas (Callaghan et al. 2013; Larsen et al. 2014), but to what extent temporal changes in cryogenic disturbance have contributed to temporal changes in vegetation is not well known. Recent vegetation change in the Arctic includes increased primary productivity recorded as increased Normalized Difference Vegetation Index in a large part of the Arctic (Xu et al. 2013). This increased greenness is often attributed to increased abundance of shrubs, a phenomenon observed using modern and historical aerial photographs (Sturm et al. 2001; Tape et al. 2006; Lantz et al. 2012; Becher et al. 2013; Fraser et al. 2014). However, whereas approximately a third of the Arctic is becoming greener, large portions remain unchanged and a small percentage is becoming browner, with presumably lower primary production; this variation cannot be explained by temperature effects alone (Xu et al. 2013). Wildfires, herbivores, plant pests, and extreme weather events may contribute to the observed spatial variability in vegetation dynamics patterns (Bjerke et al. 2014).
Altered cryogenic disturbance processes may constitute an additional mechanism behind the spatial variability in vegetation change in the Arctic, as this disturbance can both promote and hamper vegetation communities (Jonasson 1986). As cryogenic disturbance processes are important factors influencing the distribution of plants at a micro-scale (le Roux et al. 2013; le Roux and Luoto 2014), it has been hypothesised that decreased cryogenic activities in response to warmer climates could contribute to past and future vegetation changes in the Arctic (Hjort and Luoto 2009; Makoto and Klaminder 2012; Becher et al. 2013). In line with this hypothesis, an increased abundance of shrubs has been observed in geomorphological features where intense soil frost actions possibly hampered during the last decades due to recent warming (Becher et al. 2013; Frost et al. 2013). By exposing mineral soil, cryogenic disturbance processes can create gaps in the existing vegetation where seeds can germinate and species can establish (Sutton et al. 2006; Virtanen et al. 2010; Frost et al. 2013). However, geomorphological disturbance can function as an ecological filter, hindering disturbance intolerant plants (e.g., trees) from taking advantage of a warmer climate (Macias-Fauria and Johnson 2013).
Cryogenic disturbance processes might physically damage plants through several processes. First, segregation ice (pipkrake or needle-ice) can damage plant tissues both aboveground and belowground (Hopkins and Sigafoos 1951; Gartner et al. 1986; Jonasson and Callaghan 1992; Kade et al. 2005; Kade and Walker 2008). Second, the differential heave resulting from the heterogenic displacement of soil induced by soil frost and ice-lenses can disrupt plant roots (Jonasson 1986; Kade and Walker 2008). Third, the disruption of the soil surface caused by freeze–thaw cycles or the injection of soil to the surface by cryogenic overpressures in sub-soil layers can also damage plants (French 2007; Makoto and Klaminder 2012). Plant species such as Tofieldia pusilla, Juncus biglumis, Salix reticulata, Saxifraga oppositifolia, and Carex capillaris are more frequently found in cryogenically disturbed sites (Jonasson 1986; Kade and Walker 2008; Walker et al. 2004, 2011), whereas shrubs with woody root systems—e.g., Vaccinium uliginosum, Vaccinium vitis-idaea, Salix myrsinifolia, Empetrum nigrum, Andromeda polifolia, and Betula nana—grow in soils less affected by cryogenic disturbances (Walker et al. 2011; Klaus et al. 2013). However, the effect of temporal changes in cryogenic disturbances on these Arctic plant communities remains largely illusive.
To test the hypothesis that decreased cryogenic disturbances cause changes in the plant community composition, we used a cryogenic disturbance gradient developed on a patterned ground feature referred to as non-sorted circles to study the relationship between the cryogenic disturbance (differential heave) and plant community composition and we quantified vegetation changes that occurred over almost three decades in response to reduced cryogenic disturbances, as the vegetation developing on parts of this gradient was previously surveyed in detail (Jonasson 1986).
Materials and methods
Study area
The study was conducted at a patterned ground system, referred to as non-sorted circles or frost-boils (Washburn 1979). The study system is situated in northern Sweden on a shallow tilting slope of subarctic heath close to the Abisko Scientific Research Station (68°21′N, 18°48′E) (Fig. 1). Here, we refer to these non-sorted circles as “circles”. During the plant survey (2012), the mean annual air temperature (MAAT) was considerably lower (−0.86 °C) and the climate was wetter (367 mm) than the average for the last 100 years (1914–2013, −0.47 °C and 307 mm, respectively, data provided by the Abisko Scientific Research Station), and the MAAT has increased over the last few decades (Callaghan et al. 2010). Circles were characterised by sparsely vegetated circle-like areas (here called centre) 1.5 to 25 m2 in diameter surrounded by a rim of more densely vegetated areas (here called margin) with a thick layer of organic horizon (13.2 ± 6.6 cm) (Becher et al. 2015). Within these circles, several documented cryogenic activities can affect plants: (i) differential frost heave of the centre domain of the circle because of poor insulation in comparison to the surrounding areas during the winter (Peterson 2011); (ii) lateral and vertical soil motion induced both by gravity gradients and cryogenic pressure gradients arising during freezing and thawing (Becher et al. 2013); and (iii) surface ice (pipkrake or needle ice) generated at the poorly insulated areas in the circle (Kade and Walker 2008). In the study area, all these cryogenic disturbance processes have been observed, but inactive circles lacking apparent signs of recent soil motion do occur frequently in the terrain (Klaus et al. 2013).
a Map over Scandinavia with Abisko highlighted with a black dot. b Close-up of Abisko area with the lower two maps highlighted (c, d) and Abisko Scientific Research Station (ANS) highlighted with the text ANS. c Close-up of study site 1–10. d Close-up of study site 11–15. LiDAR-derived hillshade with 350° illumination angle from Lantmäteriet as background in b–d. Maps are projected on SWEREF99TM. Grid on map a: Latitude/Longitude. Grid on map b–d: SWEREF99TM, linear unit: meter. e Image of non-sorted circle 2, 10, and 11 from left to right
We selected three separated patterned ground fields to allow sampling of sites exposed to variable cryogenic activities (Fig. 1). Within each field, the plant composition and cryogenic activities were measured in five circles. Two of the fields were situated on a gentle north-facing slope (Fig. 1c), whereas the other fields were situated in a flat area adjacent to a mire (Fig. 1d). None of the studied circles was underlain by permafrost within the upper meter of the soil, but (<0.5 m) permafrost near the surface was evident in adjacent soils (distances ranging from 7 to 200 m), where circles located closer to permafrost were more exposed to cryogenic disturbance processes (Becher et al. 2015).
Plant surveys
Vegetation composition along a cryogenic disturbance gradient
The between circle difference in differential heave (generated as the centre and the margin of the circles heave to various extent between summer and winter) was used to measure of cryogenic disturbance. At the study site, differential heave of each circle was measured during the seasons of 2012/2013 (Becher et al. 2015) using GPS (Trimble R7 GNSS, Trimble Navigation Limited, 935 Stewart Drive Sunnyvale, California 94085 USA) with a real-time kinematic survey technique. The estimated vertical reproducibility of the method is 0.5 cm (Klaus et al. 2013). Measures were chosen to represent the highest possible differential heave—i.e., the most elevated point of the circle in the winter was used as the surface level of the circle. This approach was used to be able to compare our measurements with previous measurements on the site done in 1985–1986 (Jonasson 1986), knowing that the values on differential heave were more conservative in the 1980s as they represented average values for the whole circle.
The composition of the plant community was investigated using the quadrat method (50 × 50 cm frame with 25 10 × 10 cm squares) (Bullock 2006) in the centre of each circle. The abundance of each species was estimated as the number of squares in which each species was found. Plants shorter than 5 mm and dead material were not counted. Carex capillaris and Carex dioica were treated as one group because these species are hard to separate using vegetative parts only.
Vegetation changes over three decades
The composition of the plant community in two circles from one of the circle fields (Fig. 1c, field including non-sorted circle 6–11) was investigated in 1984–1985 by Jonasson (1986). In 2012, we repeated this investigation in five circles in the same field using a point intercept method (100 points/m2) (Jonasson 1988). The number of hits (on plant or flower) for the respective vascular plant was noted, and the relative hit frequency was calculated as follows:
Plants less than 5 mm in height and dead material were not counted and C. capillaris and C. dioica were treated collectively. In the 2012 survey, we investigated more circles (n = 5) than the survey conducted in 1986 (n = 2), as the location of the circles surveyed in 1986 was too uncertain to allow identification of specific circles used by Jonasson (1986).
Additional measurements
Environmental variables were measured for each circle—e.g., soil pH, soil moisture, soil temperature, and organic horizon thickness—to allow the composition of the plant community to be linked to environmental correlates. The soil moisture and temperature were measured with an instant gauge in the upper 4 cm (ML3-ThetaProbe soil moisture sensor connected to a HH2 moisture metre (Delta-T devices Ltd)) every second week during the growing season in 2012. The pH was measured in 10 g of moist soil that was sampled with a steel auger down to a depth of 5 cm and diluted in MQ-water (soil:water ratio of 5:1) at one time during the summer 2012 [For further details, see Becher et al. (2015)]. Snow depth was measured at the centre of each non-sorted circle in March 2013.
Climatic variables
Because the studied circles are exposed to strong winds and accumulate limited snow cover (average 4 ± 4 cm), the studied circles have a temperature regime strongly influenced by air temperatures. The daily mean air temperature measured at the Abisko Scientific Research Station (ANS), located about 600–1500 m from the study sites (Fig. 1b), was used to calculate the total of the negative mean daily air temperatures for a year (freeze indices) and the total of the positive mean daily air temperature for a year (thaw indices). Although the freezing and thawing conditions of the soil are highly dependent on snow conditions, the calculated freeze and thaw indices have previously been used to predict the activity of non-sorted circles without considering between-site differences in snow depth (Harris 1981). The average freeze and thaw indices were calculated for the two periods before measuring the differential heave (1976–1985 and 2003–2012) to highlight temporal trends in cryogenic conditions ongoing over a decadal time-scale.
Statistics
A two-dimensional non-metric multidimensional scaling (NMDS) ordination using the Bray–Curtis similarity measure was used to investigate the differences in the plant community composition within and among circles and indicate the direction and strength of environmental correlates such as the O-horizon thickness, pH in mineral soil, soil temperature, soil moisture, and differential heave (Oksanen et al. 2015). Differences in vegetation cover between 1984–1985 and 2012 was tested by examining the confidence intervals for each species in 2012 overlap with the average abundance reported in 1984–1985. This method was used because raw data for each circle from 1984 to 1985 were not available. A time-series analysis was performed to detect trend in freeze and thaw indices from 1980 to 2013. This long-term perspective might be important, as previous studies have claimed that the vegetation in the non-sorted circles has experienced changes since the beginning of the 20th century (Becher et al. 2013). Furthermore, ANOVA was performed to detect differences in freeze and thaw indices between the two periods around the time of investigations (1976–1985 and 2003–2012). All statistical analyses were performed using the software R (R Core Team 2015).
Results
The study site in context of the current and past cryogenic activities
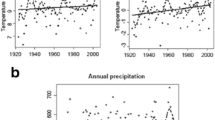
The differential heave ranged between 2 and 20 cm in all studied non-sorted circles, with an average of 11 cm (n = 15, 95th confidence interval = ±3 cm). In the re-visited field, the differential heave was 16 cm in 2013 (n = 5, 95th confidence interval = ±6 cm), which should be compared to measures in 1984–1985 that displayed a mean of 26 cm (n = 2, 95th confidence interval = ±5 cm). These differences indicated that the circles experienced a decrease in differential heave during the last three decades, as the calculated freeze and thaw indices suggested a significant trend in warming of the climate both during the summer (p < 0.001, r 2 = 0.28) and winter (p = 0.002, r 2 = 0.21) from the 1980s until today (Fig. 2). Furthermore, the ten-year average of the freeze and thaw indices show that the climatic conditions for cryogenic disturbances have been significantly weakened between the two surveys (Fig. 3) (freeze indices: F 1,18 = 14.32, p = 0.001, thaw indices: F 1,18 = 11.08, p = 0.003).
Ten-year average values for the freeze and thaw indices (°C days/year) for the years of 1976–1985 (open circle) and 2003–2012 (open triangle) and for the single year 1913 (open square). Whiskers standard deviations. Also shown are the climatic windows in which the cryogenically active non-sorted circles are expected to develop (Harris 1981). Continuous permafrost (dot-dash line), discontinuous permafrost (long-dash line), and sporadic permafrost (dashed line) limits and mean annual isotherms (solid lines) are also shown
Plant communities along the cryogenic gradient
In total, 27 species were found within the studied gradient of the non-sorted circles (Online Resource 1). The multidimensional scaling of the environmental factors along the gradient and the plant communities is shown in Fig. 4a and b. The NMDS axis 1 was most strongly correlated with the soil temperature and pH, a finding that explains 73 and 71% of the variance, respectively. Axis 2 was most strongly correlated with the thickness of the O-horizon and differential heave, a finding that explains 64 and 51% of the variance, respectively (Table 1; Fig. 4a). Most species with woody roots (e.g., E. nigrum, V. uliginosum, A. polifolia, and V. vitis-idaea) were associated with less cryogenically active sites with thicker O-horizon covering the mineral soil. Bartsia alpina and Loiseleuria procumbens were also found around less cryogenically active sites with thick O-horizon cover. Betula nana and S. myrsinifolia, both with woody roots, were found at locations with higher soil moistures. Grasses and herbs were found in sites with high cryogenic disturbances and a thinner O-horizon (Fig. 4b).
Non-metric multidimensional scaling (NMDS) ordination based on the abundance of vascular plant species for each individual non-sorted circle with a ordination indicating the strength and direction of environmental variables (Temp = Soil temperature, Moist = Soil moisture, pH = pH in mineral soil, DH = Differential heave, O horizon = depth of O-horizon) and b 95th confidence ellipsoids with the standard error for groups (low, intermediate, and high disturbance) based on the differential heave (0–9, 9–18, and 18–22 cm). AP = Andromeda polifolia, ArA = Arctostaphylos alpinus, AsA = Astragalus alpinus, BA = Bartsia alpina, BN = Betula nana, BP = Betula pubescens ssp. Czerepanovii, BV = Bistorta vivipar, CCD = Carex capillaris/dioica, CV = Carex vaginata, DO = Dryas octopetala, EN = Empetrum nigrum, EA = Equisetum arvense, ES = Equisetum scirpoides, EF = Euphrasia frigida, FO = Festuca ovina, JB = Juncus biglumis, JT = Juncus triglumis, LP = Loiseleuria procumbens, P = Pinguicula sp., RL = Rhododendron lapponicum, SM = Salix myrsinifolia, SR = Salix reticulata, Sa A = Saxifraga aizoides, SO = Saxifraga oppositifolia, TP = Tofieldia pusilla, VU = Vaccinium uliginosum, and VV = Vaccinium vitis-idaea)
Changes in vegetation communities between 1984–1985 and 2012
All species from the 1984–1985-year inventory that had a cover falling outside of the 95th confidence interval from the 2012-year inventory were considered to be significantly different between the two inventories (Fig. 5, Online Resource 2). Saxifraga oppositifolia, Equisetum scirpoides, C. capillaris, and Bistorta vivipara displayed a lower cover in 2012, and T. pusilla, Pinguicula sp., Equisetum arvense, and Dryas octopetala increased in cover since the 1984–1985 survey. Saxifraga aizoides, S. reticulata, J. biglumis/C. capillaris, and Arctostaphylos alpinus did not change between the two investigated years. One species (Minuartia stricta) that appeared in 1984–1985 was not present in 2012. However, eleven new species were found in the latter survey (V. uliginosum, S. myrsinifolia, Rhododendron lapponicum, Pedicularis lapponica, Juncus triglumis, Festuca ovina, E. nigrum, B. nana, B. alpina, Astragalus alpinus, and A. polifolia). This difference could partly be attributed to inventorying more circles, but since eight of these species were found in the surrounding area in the 1984–1985 inventory (V. uliginosum, R. lapponicum, P. lapponica, F. ovina, E. nigrum, B. nana, B. alpina, and A. polifolia), it is mainly species striving in a less disturbed environment that are found.
Discussion
Plant species and their response to differential heave
Clearly, conditions for freeze–thaw processes believed to drive cryogenic disturbance processes at the study site have decreased since the 1980s as indicated by our climatic proxies (Fig. 2). Current air temperatures and thaw indices are less ideal for the formation of active non-sorted circles than in the beginning of the twentieth century (Fig. 3) (Harris 1981). Our proxies suggest decreased cryogenic disturbance in circles within the study area during the last decades. These proxies do not consider the snow depth and its isolating capacity. Snow depth increased during the twentieth century at the Abisko Scientific Research Station, but since the 1980s there has been a decrease (Callaghan et al. 2013). However, average snow depth is only one aspect of the role of snow as an isolator that does not include temporal and spatial variation or the physical properties of the snow. The role of snow for the detected changes of cryogenic activity is still unknown. Despite indications of reduced cryogenic disturbance processes during the last decades, the studied circle system still represents a strong gradient in cryogenic disturbance processes as indicated by the measured differential heave values ranging from 2 to 20 cm. This substantial heave demonstrates that local site factors, such as water content, the soil permeability, soil surface thermal properties, and organic layer thickness (Peterson and Krantz 2003; Walker et al. 2004; Peterson 2011; Hjort 2014) stimulate cryogenic processes at our study site.
Based on the community composition of plants along the gradient in differential heave, it is clear that the variation in cryogenic processes affects some plant species. For example, in non-sorted circles experiencing high differential heave in the winter, plants characterised as being favoured by disturbances and/or high pH (e.g., A. alpinus, B. vivipara, S. aizoides, J. triglumis, D. octopetala, and S. reticulata) are more common (Fig. 4a, b). That S. reticulata and B. vivipara grow in soil affected by cryogenic processes is also supported by previous studies of these plants in active non-sorted circles both in Scandinavia and North America (Jonasson 1986; Walker et al. 2011). Salix reticulata has a woody root system on top of the soil and fibrous roots penetrating into the soil, adaptations that facilitate growth in cryogenic active soil (Jonasson 1986; Walker et al. 2011). B. vivipara, a plant with a small condensed root system, is a pioneer species in environments affected by freezing and thawing (Dormann et al. 2002). This plant, together with C. capillaris, has been found to withstand disturbances (Jonasson and Callaghan 1992). A small root system can also explain why S. oppositifolia appears to be among the most resistant plants to differential heave in the studied gradient. This species is also found in more cryogenically active sites in Alaska (Walker et al. 2004; Kade and Walker 2008), indicating that this plant can grow in soils with even stronger disturbances than in our study gradient. Juncus biglumis, also found on active patterned ground features (Anderson and Bliss 1998), is restricted to non-sorted circles with intermediate disturbances, but is closer to the high disturbance group. The group of plants that are mainly found at sites subjected to low differential heave included species with woody roots (e.g., V. uliginosum and E. nigrum), species with long fibrous roots (e.g., E. scirpoides), and species with fibrous roots (e.g., E. frigida, a low herb). Fibrous roots are fragile and easily disturbed, which may explain why the latter two plants are found in soils with low differential heave. The differential heave constitutes a strong disturbance for roots (Kade and Walker 2008), and the properties that make plants successful in cryoactive sites along the gradient can be traced back to root traits (Jonasson and Callaghan 1992). Notably, the results of the ordination indicate that direct effects from differential heave and disturbance from cryogenic processes are not the main factors controlling distribution of plant species, although these explain 51% of the variability in the studied gradient (Table 1). Soil temperature and pH are more powerful predictors of the composition of a plant community at sites with low to intermediate disturbances, indicating that multiple environmental drivers have to be considered simultaneously to understand responses of plant communities to cryogenic disturbance (Mod et al. 2014).
Vegetation changes in non-sorted circles during the last three decades
The lower differential heave in 2012–2013 than in 1984–1985 indicates that the modelled reductions in conditions for freeze and thaw processes (Fig. 3) have influenced the local scale studied here. Findings from the differential heave measurements should be treated with caution given the small number of circles, but reduced cryogenic disturbance implied by these data agrees with the main regional trend that suggests that the activity of cryogenic disturbance processes has been reduced in many circles since the 1950s (Becher et al. 2013) and that permafrost has been rapidly lost during the last decades (Akerman and Johansson 2008). During the last decades of decreased influence of cryogenic disturbance processes, the total vegetation cover increased, mainly because species already present in the surrounding in 1984–1985 (e.g., V. uliginosum, R. lapponicum, P. lapponicum, F. ovina, E. Nigrum, B. nana, B. alpina, and A. polifolia) colonised the centre of the non-sorted circles. The increased vegetation cover could in principal further reduce the differential heave, but we think that the vegetation cover and organic horizon depth, which has been observed to influence activity in non-sorted circles from the area (Klaus et al. 2013), are still too sparse for this to be realised.
Based on previous studies (Virtanen et al. 2010; le Roux et al. 2013; le Roux and Luoto 2014), we expected that cryogenic disturbance could also facilitate the establishment of species and cause vegetation changes. Specifically, circles have been shown to be favourable sites for seed germination (Sutton et al. 2006; Frost et al. 2013) and could thus facilitate the spread of species. This mechanism can explain why numerous species have also invaded the plots since the 1980s, although none of those species succeeded in becoming highly abundant (Fig. 5). Importantly, we find no support for the previous observation that cryogenic disturbance processes facilitate the spread of species at larger spatial scales, since most of the plants species that invaded non-sorted circles were found in the surrounding area during 1984–1985 and are today most common in the circles with the low (e.g., V. uliginosum, E. nigrum, B. alpina, and A. polifolia) or intermediate (e.g., S. myrsinifolia, R. lapponicum, F. ovina, and B. nana) differential heave (Fig. 4b).
In the study area, around 16% of Arctic heathlands are covered by non-sorted circles (Becher et al. 2013) and even larger regions can be influenced by cryogenic disturbances (Aalto et al. 2014; Hugelius et al. 2014). Although we observed that the shrub species that characterised the outer rim of the circles have started to invade the centre during the last decades and may in the end dominate the vegetation as the differential heave continues to decrease, the sparse vegetation in the centre is presently in a transient stage dominated by disturbance-adapted species.
Both our comparison of vegetation changes occurring since the 1980s and our assessment of vegetation along the cryogenic disturbance gradient indicate that vegetation changes occur when cryogenic disturbances decrease, findings that support our main hypothesis. By combining these two methods, we overcome many of the limitations associated with each of the individual methods and therefore obtain a more complete understanding of how the vegetation will change at different temporal scales (Dunne et al. 2004). When only the presence–absence of species is considered, the changes in the vegetation in the circles during the last two decades are relatively consistent with the changes along the spatial gradient in cryogenic disturbance processes. However, the vegetation changes are more complex when the species abundance is also considered because the species that increased the most during the period of low cryogenic disturbance were species associated with a high cryogenic disturbance (high differential heave). This result indicates that the re-vegetation of non-sorted circles following a decreased cryogenic disturbance will likely follow a transient trajectory strongly influenced by the species initially present for decades in the non-sorted circles. These results further indicate that generally decreased cryogenic disturbances (especially the re-vegetation of patterned ground) may contribute to the observed greening of the Arctic. However, the changes in cryogenic activity depend heavily on local geomorphological and environmental conditions, and the colonisation of circles following reduced cryogenic activity depends on the local pools of plant species. Therefore, more data from numerous locations around the Arctic are needed to develop a general understanding for the role of changes in cryogenic activities for current vegetation changes observed in the Arctic.
References
Aalto J, Venäläinen A, Heikkinen RK, Luoto M (2014) Potential for extreme loss in high-latitude earth surface processes due to climate change. Geophys Res Lett 41:3914–3924. doi:10.1002/2014gl060095
Akerman HJ, Johansson M (2008) Thawing permafrost and thicker active layers in sub-Arctic Sweden. Permafr Periglac Process 19:279–292. doi:10.1002/ppp.626
Anderson DG, Bliss LC (1998) Association of plant distribution patterns and microenvironments on patterned ground in a polar desert, Devon Island, N.W.T, Canada. Arctic Alpine Res 30:97–107
Becher M, Olid C, Klaminder J (2013) Buried soil organic inclusions in non-sorted circles fields in northern Sweden: age and paleoclimatic context. J Geophys Res 118:104–111. doi:10.1002/jgrg.20016
Becher M, Olofsson J, Klaminder J (2015) Cryogenic disturbance and its impact on carbon fluxes in a subarctic heathland. Environ Res Lett 10:114006. doi:10.1088/1748-9326/10/11/114006
Bjerke JW, Karlsen SR, Hogda KA, Malnes E, Jepsen JU, Lovibond S, Vikhamar-Schuler D, Tommervik H (2014) Record-low primary productivity and high plant damage in the Nordic Arctic Region in 2012 caused by multiple weather events and pest outbreaks. Environ Res Lett 9:084006. doi:10.1088/1748-9326/9/8/084006
Bullock J (2006) Plants. In: Sutherland WJ (ed) Ecological census techniques: a handbook. Cambridge University Press, Cambridge
Callaghan TV, Bergholm F, Christensen TR, Jonasson C, Kokfelt U, Johansson M (2010) A new climate era in the sub-Arctic: accelerating climate changes and multiple impacts. Geophys Res Lett 37:L14705. doi:10.1029/2009gl042064
Callaghan TV, Jonasson C, Thierfelder T, Yang Z, Hedenås H, Johansson J, Molau U, Van Bogaert R, Michelsen A, Olofsson J, Gwynn-Jones D, Bokhorst S, Phoenix G, Bjerke JW, Tømmervik H, Christensen TR, Hanna E, Koller EK, Sloan V (2013) Ecosystem change and stability over multiple decades in the Swedish subarctic: complex processes and multiple drivers. Phil Trans R Soc B 368:20120488. doi:10.1098/rstb.2012.0488
Dormann CF, Albon SD, Woodin SJ (2002) No evidence for adaptation of two Polygonum viviparum morphotypes of different bulbil characteristics to length of growing season: abundance, biomass and germination. Polar Biol 25:884–890. doi:10.1007/s00300-002-0417-4
Dunne JA, Saleska SR, Fischer ML, Harte J (2004) Intergrating experimental and gradient methods in ecological climate change research. Ecology 85:904–916
Fraser RH, Lantz TC, Olthof I, Kokelj SV, Sims RA (2014) Warming-induced shrub expansion and lichen decline in the western Canadian Arctic. Ecosystems 17:1151–1168. doi:10.1007/s10021-014-9783-3
French HM (2007) The periglacial environment, 3rd edn. Wiley, Chichester
Frost GV, Epstein HE, Walker DA, Matyshak G, Ermokhina K (2013) Patterned-ground facilitates shrub expansion in Low Arctic tundra. Environ Res Lett 8:015035. doi:10.1088/1748-9326/8/1/015035
Gartner BL, Chapin Iii FS, Shaver GR (1986) Reproduction of eriophorum vaginatum by seed in alaskan tussock tundra. J Eco 74:1–18
Harris SA (1981) Distribution of zonal permafrost landforms with freezing and thawing indices. Erdkunde 35:81–90
Hjort J (2014) Which environmental factors determine recent cryoturbation and solifluction activity in a subarctic landscape? A comparison between active and anactive features. Permafr Periglac Process 25:136–143. doi:10.1002/ppp.1808
Hjort J, Luoto M (2009) Interaction of geomorphic and ecologic features across altitudinal zones in a subarctic landscape. Geomorphology 112:324–333. doi:10.1016/j.geomorph.2009.06.019
Hopkins DM, Sigafoos RS (1951) Frost action and vegetation patterns on seward peninsula alaska. Geo Surv Bull 974-C:51–100
Hugelius G, Strauss J, Zubrzycki S, Harden JW, Schuur AG, Ping CL, Schirrmeister L, Grosse G, Michaelson GJ, Koven CD, O´Donnell JA, Elberling B, Mishra U, Camill P, Yu Z, Palmtag J, Kuhry P (2014) Estimated stocks of circumpolar permafrost carbon with quantified uncertainty ranges and identified data gaps. Biogeosciences 11:6573–6593
Jonasson S (1986) Influence of frost heaving on soil chemistry and on the distribution of plant growth forms. Geogr Ann A 68:185–195
Jonasson S (1988) Evaluation of the point intercept method for the estimation of plant biomass. Oikos 52:101–106
Jonasson S, Callaghan TV (1992) Root mechanical properties related to disturbed and stressed habitats in the Arctic. New Phytol 122:179–186. doi:10.1111/j.1469-8137.1992.tb00064.x
Kade A, Walker DA (2008) Experimental alteration of vegetation on nonsorted circles: effects on cryogenic activity and implications for climate change in the Arctic. Arct Antarct Alp Res 40:96–103
Kade A, Walker DA, Raynolds MK (2005) Plant communities and soils in cryoturbated tundra along a bioclimate gradient in the Low Arctic, Alaska. Phytocoenologia 35:761–820. doi:10.1127/0340-269X/2005/0035-0761
Klaus M, Becher M, Klaminder J (2013) Cryogenic soil activity along bioclimatic gradients in Northern Sweden: insights from eight different proxies. Permafr periglac process 24:210–223. doi:10.1002/ppp.1778
Lantz TC, Marsh P, Kokelj SV (2012) Recent shrub proliferation in the Mackenzie delta uplands and microclimatic implications. Ecosystems 16:47–59. doi:10.1007/s10021-012-9595-2
Larsen JN, Anisimov OA, Constable A, Hollowed AB, Maynard N, Prestrud P, Prowse TD, Stone JMR (2014) Polar regions. In: Barros VR et al. (eds) Climate change 2014: impacts, adaptation, and vulnerability. Part B: regional aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, pp 1567–1612
le Roux PC, Luoto M (2014) Earth surface processes drive the richness, composition and occurrence of plant species in an arctic-alpine environment. J Veg Sci 25:45–54. doi:10.1111/jvs.12059
le Roux PC, Virtanen R, Luoto M (2013) Geomorphological disturbance is necessary for predicting fine-scale species distributions. Ecography 36:800–808. doi:10.1111/j.1600-0587.2012.07922.x
Macias-Fauria M, Johnson EA (2013) Warming-induced upslope advance of subalpine forest is severely limited by geomorphic processes. Proc Natl Acad Sci USA 110:8117–8122. doi:10.1073/pnas.1221278110
Makoto K, Klaminder J (2012) The influence of non-sorted circles on species diversity of vascular plants, bryophytes and lichens in Sub-Arctic Tundra. Polar Biol 35:1659–1667. doi:10.1007/s00300-012-1206-3
Mod HK, le Roux PC, Luoto M (2014) Outcomes of biotic interactions are dependent on multiple environmental variables. J Veg Sci 25:1024–1032. doi:10.1111/jvs.12148
Oksanen J et al. (2015) vegan: Community Ecology Package. R package version 1.153. http://cran.r-project.org/, http://vegan.r-forge.r-project.org/
Peterson RA (2011) Assessing the role of differential frost heave in the origin of non-sorted circles. Quat Res 75:325–333. doi:10.1016/j.yqres.2010.08.003
Peterson RA, Krantz WB (2003) A mechanism for differential frost heave and its implications for patterned-ground formation. J Glaciol 49:69–80
R Development Core Team (2015) R: A language and environment for statistical computing. R Foundation for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, URL http://www.R-project.org
Sturm M, Racine C, Tape K (2001) Climate change—Increasing shrub abundance in the Arctic. Nature 411:546–547. doi:10.1038/35079180
Sutton JT, Hermanutz L, Jacobs JD (2006) Are frost boils importnat for the recruitment of Arctic-Alpine plants? Arct Antarct Alp Res 38:273–275
Tape K, Sturm M, Racine C (2006) The evidence for shrub expansion in Northern Alaska and the Pan-Arctic. Glob Chang Biol 12:686–702. doi:10.1111/j.1365-2486.2006.01128.x
Virtanen R, Luoto M, Rämä T, Mikkola K, Hjort J, Grytnes J, Birks H (2010) Recent vegetation changes at the high latitude tree line ecotone are controlled by goemorphological disturbance, productivity and diversity. Glob Ecol Biogeogr 19:810–821
Walker DA, Epstein HE, Gould WA, Kelley AM, Kade AN, Knudson JA, Krantz WB, Michaelson G, Peterson RA, Ping CL, Raynolds MK, Romanovsky VE, Shur Y (2004) Frost-boil ecosystems: complex interactions between landforms, soils, vegetation and climate. Permafr Periglac Process 15:171–188. doi:10.1002/ppp.487
Walker DA, Kuss P, Epstein HE, Kade AN, Vonlanthen CM, Raynolds MK, Daniëls FJA (2011) Vegetation of zonal patterned-ground ecosystems along the North America Arctic bioclimate gradient. Appl Veg Sci 14:440–463
Washburn AL (1979) Geocryology: a survey of periglacial processes and environments. E. Arnold Ltd., London
Xu L, Myneni RB, Chapin FS III, Callaghan TV, Pinzon JE, Tucker CJ, Zhu Z, Bi J, Ciais P, Tommervik H, Euskirchen ES, Forbes BC, Piao SL, Anderson BT, Ganguly S, Nemani RR, Goetz SJ, Beck PSA, Bunn AG, Cao C, Stroeve JC (2013) Temperature and vegetation seasonality diminishment over northern lands. Nat Clim Change 3:581–586. doi:10.1038/nclimate1836
Acknowledgements
We would like to thank the Abisko Scientific Research Station for providing climatic data and accommodations. We would also like to thank Professor Stuart Harris for valuable discussions about the study and three anonymous reviewers whose comments greatly help us improve the manuscript. Funding for this study was provided by the Swedish Research Council Project No. 2009-3282.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Becher, M., Olofsson, J., Berglund, L. et al. Decreased cryogenic disturbance: one of the potential mechanisms behind the vegetation change in the Arctic. Polar Biol 41, 101–110 (2018). https://doi.org/10.1007/s00300-017-2173-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-017-2173-5