Abstract
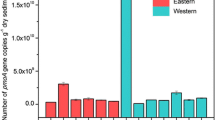
Archaeal communities represent a significant fraction of the Antarctic marine microbial plankton and surely play a relevant role in the proper functioning of the ecosystem. We studied the archaeal community structure in surface water samples from Potter Cove, Antarctica. Temporal and spatial variability was investigated along a whole year cycle using DGGE and 16S rRNA gene sequencing from clone libraries. Additionally, photosynthetic pigments, suspended particulate matter (SPM), salinity and temperature were measured. The multivariate analysis performed using diversity, dominance and richness indexes, and environmental data evidenced a seasonal pattern in the archaeal community and revealed that spring–summer samples clustered separately from autumn to winter ones. High salinity and high values of diversity and richness were related to autumn–winter samples, whereas the spring–summer samples were associated mainly with higher values of temperature, SPM, Chl-a, carotenoids and archaeal dominance. The phylogenetic analysis of five independent clone libraries (467 sequences) showed that 448 sequences fell into a clade containing Nitrosopumilus maritimus and other sequences of ammonia-oxidizing archaea which belong to the Thaumarchaeota phylum. A high fraction of these sequences (62 %) constituted a single cluster containing only highly similar Potter Cove representatives, which probably belong to the same species. Fifteen sequences were affiliated to a group closely related to the order Thermoplasmatales (Euryarchaeota). This work represents a first step towards obtaining a deep understanding of the structure of archaeal communities from Antarctic coastal marine environments and contributes to cover the current gap in knowledge of the dynamics of the archaeoplankton in the Antarctic seas.







Similar content being viewed by others
References
Amano-Sato C, Akiyama S, Uchida M, Shimada K, Utsumi M (2013) Archaeal distribution and abundance in water masses of the Arctic Ocean, Pacific sector. Aquat Microb Ecol 69:101–112
Bano N, Ruffin S, Ransom B, Hollibaugh JT (2004) Phylogenetic composition of Arctic Ocean archaeal assemblages and comparison with Antarctic assemblages. Appl Environ Microbiol 70:781–789
Brochier-Armanet C, Boussau B, Gribaldo S, Forterre P (2008) Mesophilic crenarchaeota: proposal for a third archaeal phylum, the Thaumarchaeota. Nat Rev Microbiol 6:245–252
Brochier-Armanet C, Gribaldo S, Forterre P (2012) Spotlight on the Thaumarchaeota. ISME J 6:227–230
Church MJ, DeLong EF, Ducklow HW, Karner MB, Preston CM, Karl DM (2003) Abundance and distribution of planktonic Archaea and Bacteria in the waters west of the Antarctic Peninsula. Limnol Oceanogr 48:1893–1902
Collins RE, Rocap G, Deming JW (2010) Persistence of bacterial and archaeal communities in sea ice through an Arctic winter. Environ Microbiol 12:1828–1841
Cowie ROM, Masa EW, Ryan KG (2010) Archaeal diversity revealed in Antarctic sea ice. Antarct Sci 23:531–536
Curtosi A, Pelletier E, Vodopivez CL, Mac Cormack WP (2007) Distribution pattern of PAHs in soil and surface marine sediments near Jubany Station (Antarctica). Possible role of permafrost as a low-permeability barrier. Sci Total Environ 383:193–204
DeLong EF (1992) Archaea in coastal marine environments. Proc Natl Acad Sci USA 89:5685–5689
DeLong EF, Wu KY, Prezelin BB, Jovine RVM (1994) High abundance of Archaea in Antarctic marine picoplankton. Nature 371:695–697
Di Rienzo JA, Casanoves F, Balzarini MG, Gonzalez L, Tablada M, Robledo CW (2001) InfoStat versión 2011. Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. http://www.infostat.com.ar
Ferrari VC, Hollibaugh T (1999) Distribution of microbial assemblages in the central Arctic Ocean basin studied by PCR/DGGE: analysis of a large data set. Hydrobiologia 401:55–68
Fuenets VL, Schnack-Schiel SB, Schloss IR, Esnal GG (2008) Mesozooplankton of Potter Cove: community composition and seasonal distribution in 2002–2003. Rep Pol Mar Res 571:75–84
Fuhrman J, Hagström Å (2008) Bacterial and archaeal community structure and its patterns. In: Kirchman DL (ed) Microbial ecology of the oceans. Wiley, Hoboken, pp 45–90
Galand PE, Lovejoy C, Pouliot J, Vincent WF (2008) Heterogeneous archaeal communities in the particle-rich environment of an arctic shelf ecosystem. J Mar Syst 74:774–782
Hallam SJ, Mincer TJ, Schleper C, Preston CM, Roberts K, Richardson PM, DeLong EF (2006) Pathways of carbon assim-ilation and ammonia oxidation suggested by environmental genomic analyses of marine Crenarchaeota. PLoS Biol 4:520–536, E95. doi:10.1371/journal.pbio.0040095
Herfort L, Schouten S, Abbas B, Veldhui MJW, Coolen MJL, Wuchter C (2007) Variations in spatial and temporal distribution of Archaea in the North Sea in relation to environmental variables. FEMS Microbiol Ecol 62:242–257
Holland SM (2003) Analytic rarefaction. See http://www.uga.edu/strata/software/
Huber T, Faulkner G, Hugenholtz P (2004) Bellerophon, a program to detect chimeric sequences in multiple sequence alignments. Bioinformatics 20:2317–2319
Iino T, Mori K, Suzuki K (2010) Methanospirillum lacunae sp. Nov., a methane-producing archaeon isolated from a puddly soil, and emended descriptions of the genus Methanospirillum and Methanospirillum hungatei. Int J Syst Evol Microbiol 60:2563–2566
Ingalls AE, Shah SR, Hansman RL, Aluwihare LI, Santos GM, Druffel ER, Pearson A (2006) Quantifying archaeal community autotrophy in the mesopelagic ocean using natural radiocarbon. Proc Nat Acad Sci USA 103:6442–6447
JGI (Joint Genome Institute) (2013) https://img.jgi.doe.gov/cgi-bin/mer/main.cgi?section=TaxonDetail&page=taxonDetail&taxon_oid=3300000136
Kalanetra KM, Bano N, Hollibaugh JT (2009) Ammonia-oxidizing Archaea in the Arctic Ocean and Antarctic coastal waters. Environ Microbiol 11:2434–2445
Karner MB, DeLong EF, Karl DM (2001) Archaeal dominance in the mesopelagic zone of the Pacific Ocean. Nature 409:507–510
Lin X, Kennedy D, Fredrickson J, Bjornstad B, Konopka A (2012) Vertical stratification of subsurface microbial community composition across geological formations at the Hanford Site. Environ Microbiol 14:414–425
Lorenzen CJ (1967) Determination of chlorophyll and phaeopigments: spectrophotometric equations. Limnol Oceanogr 12:343–346
Massana R, Taylor LT, Murray AE, Wu KY, Jeffrey WH, DeLong EF (1998) Vertical distribution and temporal variation of marine planktonic archaea in the Gerlache Strait, Antarctica, during early spring. Limnol Oceanogr 43:607–617
Murray AE, Preston CM, Massana R, Taylor LT, Blakis A, Wu K, DeLong EF (1998) Seasonal and spatial variability of Bacterial and Archaeal assemblages in the coastal waters near Anvers Island, Antarctica. Appl Environ Microbiol 64:2585–2595
Muyzer G, de Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rDNA. Appl Environ Microb 59:695–700
Muyzer G, Brinkhoff T, Nübel U, Santegoeds C, Schäfer H, Wawer C (2004) Denaturing gradient gel electrophoresis (DGGE) in microbial ecology. In: Kowalchuk GA, de Bruin FJ, Head IM, Akkermans DL, Van Elsas JD (eds) Molecular microbial ecology manual. Kluwer, Dordrecht, pp 743–770
Pester M, Schleper C, Wagner M (2011) The Thaumarchaeota: an emerging view of their phylogeny and ecophysiology. Curr Opin Microbiol 14:300–306
Piquet AM-T, Bolhuis H, Davidson AT, Thomson PG, Buma AGJ (2008) Diversity and dynamics of Antarctic marine microbial eukaryotes under manipulated environmental UV radiation. FEMS Microbiol Ecol 66:352–366
Piquet AM-T, Bolhuis H, Davidson AT, Buma AG (2010) Seasonal succession and UV sensitivity of marine bacterioplankton at an Antarctic coastal site. FEMS Microbiol Ecol 73:68–82
Quartino ML, Boraso de Zaixso AL (2008) Summer macroalgal biomass in Potter Cove, South Shetland Islands, Antarctica: its production and flux to the ecosystem. Pol Biol 31:281–294
Raskin L, Stromley JM, Rittmann BE, Stahl DA (1994) Group-specific 16S rRNA hybridization probes to describe natural communities of methanogens. Appl Environ Microbiol 60:1232–1240
Schleper C, Jurgens G, Jonuscheit M (2005) Genomic studies of uncultivated Archaea. Nat Rev Microbiol 3:479–488
Schloss IR, Ferreyra GA (2002) Primary production, Light and vertical mixing in Potter Cove, a shallow bay in the maritime Antractica. Pol Biol 25:41–48
Schloss IR, Ferreyra GA, Ruiz-Pino D (2002) Phytoplankton biomass in Antarctic shelf zones: a conceptual model based on Potter Cove, King George Island. J Mar Syst 36:129–143
Schloss IR, Abele D, Demers S, Bers V, González O, Ferreyra GA (2012) Response of phytoplankton dynamics to 19-year (1991–2009) climate trends in PotterCove (Antarctica). J Mar Syst 92:53–66
Strickland JDH, Parsons TR (1968) A practical handbook of seawater analysis. Pigment analysis, Bull Fish Res Bd, Otawa, Canada, 167, 311 pp
Strickland JDH, Parsons DR (1972) A practical handbook of seawater analysis. J Fish Res Board Can Bull 167:1–310
Wuchter C, Abbas B, Coolen MJL, Herfort L, van Bleijswijk J, Timmers P (2006) Archaeal nitrification in the ocean. Proc Natl Acad Sci USA 103:12317–12322
Zehr J, Ward BB (2002) Nitrogen Cycling in the Ocean: new perspectives on processes and paradigms. Appl Environ Microbiol 79:1015–1024
Acknowledgments
This research was carried out under an agreement between the Instituto Antártico Argentino and the Facultad de Farmacia y Bioquímica of the Universidad de Buenos Aires. This work was supported by grants PICTO 2010-0124 from the Agencia Nacional de Promoción Científica y Tecnológica (ANPCyT) and UBA 20020100100378 from Universidad de Buenos Aires. Also we had the financial support from the European Commission through the Marie Curie Action IRSES, project no 318718, IMCONet (Interdisciplinary Modelling of climate change in coastal Western Antarctica—Network for staff Exchange and Training). We thank Gustavo Latorre, Gastón Aguirre and Oscar Gonzalez for their technical assistance and Cecilia Ferreiro for the correction of the English manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
300_2014_1569_MOESM1_ESM.tif
Annual variation of chlorophyll a in Potter Cove. E1 (inner cove), E2 (outer cove), E3 (mouth of Potter Creek) (TIFF 44 kb)
300_2014_1569_MOESM2_ESM.tif
Annual variation of carotenoids in Potter Cove. E1 (inner cove), E2 (outer cove), E3 (mouth of Potter Creek) (TIFF 42 kb)
300_2014_1569_MOESM3_ESM.tif
Annual variation of phaeopigments in Potter Cove. E1 (inner cove), E2 (outer cove), E3 (mouth of Potter Creek) (TIFF 42 kb)
300_2014_1569_MOESM4_ESM.tif
Annual variation of suspended particulate matter (SPM) in Potter Cove. E1 (inner cove), E2 (outer cove), E3 (mouth of Potter Creek) (TIFF 38 kb)
300_2014_1569_MOESM5_ESM.tif
Annual variation of temperature in Potter Cove. E1 (inner cove), E2 (outer cove), E3 (mouth of Potter Creek) (TIFF 35 kb)
300_2014_1569_MOESM7_ESM.tif
Rarefaction curves of the archaeal 16S rRNA sequences from Potter Cove sea waters at 97 % identity. a) Rarefaction curve from 5 combined libraries, b) rarefaction curves from each library. The total number of sequenced clones is plotted against the number of OTUs observed in the same library (TIFF 194 kb)
Rights and permissions
About this article
Cite this article
Hernández, E.A., Piquet, A.MT., Lopez, J.L. et al. Marine archaeal community structure from Potter Cove, Antarctica: high temporal and spatial dominance of the phylum Thaumarchaeota. Polar Biol 38, 117–130 (2015). https://doi.org/10.1007/s00300-014-1569-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-014-1569-8