Abstract
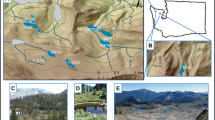
We investigated the role of autochthonous and terrestrial carbon in supporting aquatic food webs in the Canadian High Arctic by determining the diet of the dominant primary consumer, aquatic chironomids. These organisms were studied in fresh waters on 3 islands of the Arctic Archipelago (~74–76°N) including barren polar desert watersheds and a polar oasis with lush meadows. Stomach content analysis of 578 larvae indicated that chironomids primarily ingested diatoms and sediment detritus with little variation among most genera. Carbon and nitrogen stable isotope mixing models applied to 2 lakes indicated that benthic algae contributed 68–95% to chironomid diet at a polar desert site and 70–78% at a polar oasis site. Detritus, originating from either phytoplankton or terrestrial sources, also contributed minor amounts to chironomid diet (0–32%). Radiocarbon measurements for the 2 lakes showed that old terrestrial carbon did not support chironomid production. Carbon stable isotope ratios of chironomids in other High Arctic lakes provided further dietary evidence that was consistent with mixing model results. These findings indicate that, in the Canadian High Arctic, chironomids (and fish that consume them) are supported primarily by benthic algae in both polar desert and oasis lakes. In contrast, our review of carbon flow studies for lakes in other Arctic regions of North America shows that terrestrial carbon and phytoplankton can be important energy sources for consumers. This study provides a baseline to detect future climate-related impacts on carbon pathways in High Arctic lakes.



Similar content being viewed by others
References
Abbot MB, Stafford TW (1996) Radiocarbon geochemistry of modern and ancient Arctic lake systems, Baffin Island, Canada. Quaternary Res 45:300–311
ACIA (2005) Arctic climate impact assessment (ACIA). Cambridge University Press, New York, USA
Barr W (1971) Postglacial isostatic movement in northeastern Devon Island: a reappraisal. Arctic 24:249–268
Berg MB (1995) Larval food and feeding behavior. In: Armitage PD, Cranston PS, Pinder LCV (eds) The chironomidae: the biology and ecology of non-biting midges. Chapman and Hall, London, UK
Blake W (1991) Ratios of stable carbon isotopes in some High Arctic plants and lake sediments. J Paleolimnol 6:157–166
Bliss LC (1977) Introduction. In: Bliss LC (ed) Truelove Lowland, Devon Island, Canada: a High Arctic ecosystem. University of Alberta Press, Edmonton, CA
Bunn SE, Barton DR, Hynes HBN, Power G, Pope MA (1989) Stable isotope analysis of carbon flow in a tundra river system. Can J Fish Aquat Sci 46:1769–1775
Chapin DM (1996) Nitrogen mineralization, nitritification, and denitrification in a high arctic lowland ecosystem, Devon Island, N.W.T., Canada. Arctic Alpine Res 28:85–92
Chételat J, Amyot M, Cloutier L, Poulain A (2008) Metamorphosis in chironomids, more than mercury supply, controls methylmercury transfer to fish in High Arctic lakes. Environ Sci Technol 42:9110–9115
Doi H, Kikuchi E, Takagi S, Shikano S (2006) Selective assimilation by deposit feeders: experimental evidence using stable isotope ratios. Basic Appl Ecol 7:159–166
Gantner N, Power M, Iqaluk D, Meili M, Borg H, Sundbom M, Solomon KR, Lawson G, Muir DC (2010) Mercury concentrations in landlocked Arctic char (Salvelinus alpinus) from the Canadian Arctic: Part I: Insights from trophic relationships in 18 lakes. Environ Toxicol Chem 29:621–632
Givelet N, Roos-Barraclough F, Goodsite ME, Cheburkin AK, Shotyk W (2004) Atmospheric mercury accumulation rates between 5900 and 800 calibrated years BP in the High Arctic of Canada recorded by peat hummocks. Environ Sci Technol 38:4964–4972
Goedkoop W, Åkerblom N, Demandt MH (2006) Trophic fractionation of carbon and nitrogen stable isotopes in Chironomus riparius reared on food of aquatic and terrestrial origin. Freshwat Biol 51:878–886
Guiguer KRRA, Reist JD, Power M, Babaluk JA (2002) Using stable isotopes to confirm the trophic ecology of Arctic charr morphotypes from Lake Hazen, Nunavut, Canada. J Fish Biol 60:348–362
Hecky RE, Hesslein RH (1995) Contributions of benthic algae to lake food webs as revealed by stable isotope analysis. J N Am Benthol Soc 14:631–653
Hershey AE, Fortino K, Beaty S, Kelly S, Keyse M, Luecke C, O’Brien WJ (2005) δ13C signatures of chironomids in arctic lakes: Role and direction of benthic-pelagic coupling. Verh Internat Verein Limnol 29:92–96
Hershey AE, Beaty S, Fortino K, Kelly S, Keyse M, Luecke C, O’Brien WJ, Walen SC (2006) Stable isotope signatures of benthic invertebrates in arctic lakes indicate limited coupling to pelagic production. Limnol Oceanogr 51:177–188
Ingvason HR, Ólafsson JS, Gardarsson A (2004) Food selection of Tanytarsus gracilentus larvae (Diptera: Chironomidae): an analysis of instars and cohorts. Aquat Ecol 38:231–237
Johnson L (1980) The arctic charr, Salvelinus alpinus. In: Balon EK (ed) Charrs, salmonid fishes of the genus Salvelinus. Dr. W. Junk Publishers, The Hague, NE
Karlsson J, Byström P (2005) Littoral energy mobilization dominates energy supply for top consumers in subarctic lakes. Limnol Oceanogr 50:538–543
Karlsson J, Jonsson A, Jansson M (2001) Bacterioplankton production in lakes along an altitude gradient in the subarctic north of Sweden. Microb Ecol 42:372–382
Karlsson J, Ask J, Jansson M (2008) Winter respiration of allochthonous and autochthonous organic carbon in a subarctic clear-water lake. Limnol Oceanogr 53:948–954
Karlsson J, Byström P, Ask J, Ask P, Persson L, Jansson M (2009) Light limitation of nutrient-poor lake ecosystems. Nature 460:506–509
Kidd KA, Hesslein RH, Ross BJ, Koczanski K, Stephens GR, Muir DCG (1998) Bioaccumulation of organochlorines through a remote freshwater food web in the Canadian Arctic. Environ Pollut 102:91–103
Markager S, Vincent WF, Tang EPY (1999) Carbon fixation by phytoplankton in high Arctic lakes: Implications of low temperature for photosynthesis. Limnol Oceanogr 44:597–607
Merritt RW, Cummins KW, Berg MB (2008) An introduction to the aquatic insects of North America, 4th edn. Kendall/Hunt Publishing Company, Dubuque
Minns CK (1977) Limnology of some lakes in Truelove Lowland. In: Bliss LC (ed) Truelove Lowland, Devon Island, Canada: a high Arctic ecosystem. University of Alberta Press, Edmonton, CA
Oliver DR, Roussel ME (1983) The insects and arachnids of Canada, Part 11. The genera of larval midges of Canada. Diptera: Chironomidae. Agriculture Canada Publication 1746
Phillips DL, Gregg JW (2001) Uncertainty in source partitioning using stable isotopes. Oecologia 127:171–179
Phillips DL, Gregg JW (2003) Source partitioning using stable isotopes: coping with too many sources. Oecologia 136:261–269
Post DM (2002) Using stable isotopes to estimate trophic position: models, methods, and assumptions. Ecology 83:703–718
Power M, Klein GM, Guiger KRRA, Kwan MKH (2002) Mercury accumulation in the fish community of sub-Arctic lake in relation to trophic position and carbon sources. J Appl Ecol 39:819–830
Rautio M, Vincent WF (2007) Isotopic analysis of the sources of organic carbon for zooplankton in shallow subarctic and arctic waters. Ecography 30:77–87
Riber HH, Wetzel RG (1987) Boundary-layer and internal diffusion effects on phosphorus fluxes in lake periphyton. Limnol Oceanogr 32:1181–1194
Rigler FH (1978) Limnology in the high arctic: a case study of Char Lake. Verhan Internat Verein Limnol 20:127–140
Schell DM (1983) Carbon-13 and carbon-14 abundances in Alaskan aquatic organisms: delayed production from peat in arctic food webs. Science 219:1068–1071
Semkin RG, Mierle G, Neureuther RJ (2005) Hydrochemistry and mercury cycling in a High Arctic watershed. Sci Tot Environ 342:199–221
Sierszen ME, McDonald ME, Jensen DA (2003) Benthos as the basis for arctic lake food webs. Aquat Ecol 37:437–445
Solomon CT, Carpenter SR, Cole JJ, Pace ML (2008) Support of benthic invertebrates by detrital resources and current autochthonous primary production: results from a whole-lake 13C addition. Freshwat Biol 53:42–54
Squires MM, Lesack LFW (2002) Water transparency and nutrients as controls on phytoplankton along a flood-frequency gradient among lakes of the Mackenzie Delta, western Canadian Arctic. Can J Fish Aquat Sci 59:1339–1349
Trudeau V, Rasmussen JB (2003) The effect of water velocity on stable carbon and nitrogen isotope signatures of periphyton. Limnol Oceanogr 48:2194–2199
Vadeboncoeur Y, Jeppesen E, Vander Zanden MJ, Schierup HH, Christoffersen K, Lodge DM (2003) From Greenland to green lakes: cultural eutrophication and the loss of benthic pathways in lakes. Limnol Oceanogr 48:1408–1418
Vadeboncoeur Y, Peterson G, Vander Zanden J, Kalff J (2008) Benthic algal production across lake size gradients: interactions among morphometry, nutrients, and light. Ecology 89:2542–2552
Welch HE (1976) Ecology of Chironomidae (Diptera) in a polar lake. J Fish Res Board Can 33:227–247
Welch HE, Kalff J (1974) Benthic photosynthesis and respiration in Char Lake. J Fish Res Board Can 31:609–620
Wiederholm T (1983) Chironomidae of the Holarctic region. Keys and diagnoses. Part 1. Larvae. Entomologica Scandinavica Supplement 19
Wolfe AP, King RH (1999) A paleolimnological constraint to the extent of the last glaciation on northern Devon Island, Canadian High Arctic. Quaternary Sci Rev 18:1563–1568
Acknowledgments
This work was funded by NSERC and FQRNT postgraduate scholarships to JC and NSERC Discovery, FQRNT-Équipe, Science Horizon, and Northern Supplement grants to MA. Logistical support from the Polar Continental Shelf Project (PCSP) to MA is gratefully acknowledged. JC and MA received NSTP funds from INAC. Lake and drainage basin areas were determined by Jean Daoust. We thank Mélanie Bouffard for ancillary water quality analyses, Edenise Garcia, Alexandre Poulain and Pilipoosie Iqaluk for assistance in the field, and Dominic Bélanger for laboratory assistance. Two anonymous reviewers provided valuable comments on the manuscript. This paper is PCSP contribution 001-10.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chételat, J., Cloutier, L. & Amyot, M. Carbon sources for lake food webs in the Canadian High Arctic and other regions of Arctic North America. Polar Biol 33, 1111–1123 (2010). https://doi.org/10.1007/s00300-010-0797-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-010-0797-9