Abstract
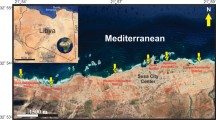
Activities related to Antarctic research stations have caused significant local impacts on the marine environment, potentially affecting the recruitment of benthic invertebrates. Herein, we report the community structure of recruiting marine eukaryotes onto artificial substrata using molecular techniques. Slides were deployed at three sites adjacent to McMurdo Station, Scott Base, and Cape Armitage in McMurdo Sound. Denaturing gradient gel electrophoresis (DGGE) analysis revealed complex and diverse eukaryotic communities had established on artificial surfaces deployed at a range of site and depth regimes after 12 months. Analysis of similarity results detected significantly greater variability in community profiles among sites than within sites. The nonmetric multidimensional scaling plot constructed from DGGE banding patterns revealed different benthic communities had established at 12 and 18 m depths. Despite this, the variation in community composition was greater among sites than between depths, especially at Cape Armitage and Scott Base. Sequence analysis of excised DGGE bands revealed a predominance of arthropod and dinoflagellate sequences at Cape Armitage. In contrast, a wide diversity of phyla including cnidaria, bryozoa, protozoa, dinoflagellates, arthropods, platyhelminths, and annelids were present adjacent to the two research stations. The abundance of diatoms detected in Cape Armitage benthic assemblages exceeded the abundance of diatoms from McMurdo Station and Scott Base by almost two orders of magnitude. The discovery that distinct eukaryotic communities recruit at different sites and depths is probably due to complex interactions between multiple factors including water quality, larval supply, and light. The detection of sessile phyla on slides at each of the sites indicates that the pollution profiles present at each site is not an impediment to successful recruitment of these species.






Similar content being viewed by others
References
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucl Acids Res 25:3389–3402
Anderson B, Chague-Goff C (1996) Benthic foraminifera and trace metals in sediments off the Scott Base sewer outfall, Antarctica. Antarctic Research Centre Report No. 18. Research School of Earth Sciences, Victoria University
Battershill C (1989) Distribution and abundance of benthic marine species at Cape Armitage, Ross Island, Antarctica—initial results. NZ Ant Rec 9:35–51
Battershill C (1990) Temporal changes in Antarctic marine benthic community structure. NZ Ant Rec 10:23–27
Clarke KR (1993) Non-parametric multivariate analysis of changes in community structure. Aust J Ecol 18:117–143
Conlan KE, Kim SL, Lenihan HS, Oliver JS (2004) Benthic changes during 10 years of organic enrichment by McMurdo Station, Antarctica. Mar Pollut Bull 49:43–60
Cox TF, Cox MA (1994) Multidimensional scaling. Chapman & Hall, London
Crockett AB, White GJ (2003) Mapping sediment contamination and toxicity in Winter Quarters Bay, McMurdo Station, Antarctica. Environ Monit Assess 85:257–275
Cunningham L, Snape I, Stark JS, Riddle MJ (2005) Benthic diatom community response to environmental variables and metal concentrations in a contaminated bay adjacent to Casey Station, Antarctica. Mar Pollut Bull 50:264–275
Dayton PK, Oliver JS (1977a) Antarctic soft bottom benthos in oligotrophic and eutrophic environments. Science 197:55–58
Dayton PK, Oliver JS (1977b) Processes organising contrasting benthic communities of McMurdo Sound. Ant J USA 12:20–21
Dayton PK, Robilliard GA, Paine RT (1970) Benthic faunal zonation as a result of anchor ice at McMurdo Station, Antarctica. Academic, London
Dayton PK, Robilliard GA, Paine T, Dayton LB (1974) Biological accommodation in the benthic community at McMurdo Sound, Antarctica. Ecol Monogr 44:105–128
Fent K (1996) Ecotoxicology of organotin compounds. Crit Rev Toxicol 26:1–117
Ferrari VC, Hollibaugh JT (1999) Distribution of microbial assemblages in the Central Arctic Ocean Basin studied by PCR/DGGE: analysis of a large data set. Hydrobiology 401:55–68
Ferris MJ, Muyzer G, Ward DM (1996) Denaturing gradient gel electrophoresis profiles of 16S rRNA-defined populations inhabiting a hot spring microbial mat community. Appl Environ Microbiol 62:340–346
Fromin N, Hamelin J, Tarnawski S, Roesti D, Jourdain-Miserez K, Forestier N, Teyssier-Cuvelle S, Gillet F, Aragno M, Rossi P (2002) Statistical analysis of denaturing gel electrophoresis (DGE) fingerprinting patterns. Environ Microbiol 4:634–643
Gillan DC (2004) The effect of an acute copper exposure of the diversity of a microbial community in North Sea sediments as revealed by DGGE analysis—the importance of the protocol. Mar Pollut Bull 49:504–513
Harris C, Given D, Bassett J, Patrick M, Wood S (2001) Key pressures on the Ross Sea region environment. New Zealand Antarctic Institute, Christchurch
Jackson CR, Churchill PF (1999) Analysis of microbial communities by denaturing gradient gel electrophoresis: applications and limitations. Recent Res Dev Microbiol 3:81–91
Jukes TH, Cantor CR (1969) Evolution of protein molecules. In: Munro HN (ed) Mammalian protein metabolism. Academic, New York, pp 21–132
Kennicutt MC, McDonald SJ, Sericano JL, Boothe P, Oliver J, Safe S, Presley BJ, Liu H, Wolfe D, Wade TL, Crockett A, Bockus D (1995) Human contamination of the marine environment—Arthur Harbor and Mcmurdo Sound, Antarctica. Environ Sci Technol 29:1279–1287
King CK, Riddle MJ (2001) Effects of metal contaminants on the embryonic and larval development of the common Antarctic sea urchin Sterechinus neumayeri (Meissner). Mar Ecol Prog Ser 215:143–154
Leitz T, Wagner T (1993) The marine bacterium Alteromonmas espejiana induces metamorphosis of the hydroid Hydractinia echinate. Mar Biol 115:173–178
Lenihan HS (1992) Benthic marine pollution around McMurdo Station, Antarctica: a summary of findings. Mar Pollut Bull 25:318–323
Lenihan HS, Oliver JS (1995) Anthropogenic and natural disturbances to marine benthic communities in Antarctica. Ecol Appl 5:311–326
Lenihan HO, Oliver JS, Oakden JM, Stephenson MD (1990) Intense and localized benthic marine pollution around McMurdo Station, Antarctica. Mar Pollut Bull 21:422–430
Lenihan HS, Peterson CH, Kim SL, Conlan KE, Fairey R, McDonald C, Grabowski JH, Oliver JS (2003) Variation in marine benthic community composition allows discrimination of multiple stressors. Mar Ecol Prog Ser 261:63–73
Ludwig W, Strunk O, Klugbauer S, Klugbauer N, Weizenegger M, Neumaier J, Bachleitner M, Schleifer K-H (1998) Bacterial phylogeny based on comparative sequence analysis. Electrophoresis 19:554–568
Maidak BL, Cole JR, Parker CT Jr, Garrity GM, Larsen N, Li B, Lilburn TG, McCaughey MJ, Olsen GJ, Overbeek R, Pramanik S, Schmidt TM, Tiedje JM, Woese CR (1999) A new version of the RDP (ribosomal database project). Nucl Acids Res 27:171–173
Mcminn A, Runcie JW, Riddle MJ (2004) Effect of seasonal sea ice breakout on the photosynthesis of benthic diatom mats at Casey, Antarctica. J Phycol 40:62–69
Mihm JW, Banta WC, Loeb GI (1981) Effects of adsorbed organic and primary fouling films on bryozoan settlement. J Exp Mar Biol Ecol 54:167–169
Muyzer G, de Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Negri AP, Hales LT, Battershill C, Wolff C, Webster NS (2004) TBT contamination identified in Antarctic marine sediments. Mar Pollut Bull 48:1142–1144
Negri A, Boyle S, Brinkman D, Burns K, Webster N (2006) Contamination in sediments, bivalves and sponges of McMurdo Sound, Antarctica. Environ Pollut (in press)
Nübel U, Garcia-Pichel F, Muyzer G (1997) PCR primers to amplify 16S rRNA genes from cyanobacteria. Appl Environ Microbiol 63:3327–3332
Pearson TH, Rosenberg R (1978) Macrobenthic succession in relation to organic enrichment and pollution of the marine environment. Oceanogr Mar Biol Annu Rev 16:229–311
Powell SM, Bowman JP, Snape I, Stark JS (2003) Microbial community variation in pristine and polluted nearshore Antarctic sediments. FEMS Microbiol Ecol 45:135–145
Powell SM, Riddle MJ, Snape I, Stark JS (2005) Location and DGGE methodology can influence interpretation of field experimental studies on the response of hydrocarbons by Antarctic microbial community. Ant Sci 17:353–360
Risebrough RW, De Lappe BW, Younghans-Haug (1990) PCB and PCT contamination in Winter Quarters Bay, Antarctica. Mar Pollut Bull 21:523–529
Stark JS, Riddle MJ, Snape I, Scouller RC (2003a) Human impacts in Antarctic marine soft-sediment assemblages: correlations between multivariate biological patterns and environmental variables at Casey Station. Est Coast Shelf Sci 56:717–734
Stark JS, Snape I, Riddle MJ (2003b) The effects of petroleum hydrocarbon and heavy metal contamination of marine sediments on recruitment of Antarctic soft-sediment assemblages: a field experimental investigation. J Exp Mar Biol Ecol 28:21–50
Stark JS, Riddle MJ, Smith SDA (2004) Influence of an Antarctic waste dump on recruitment to nearshore marine soft-sediment assemblages. Mar Ecol Prog Ser 276:53–70
Suzuki MT, Giovannoni SJ (1996) Bias caused by template annealing in the amplification of mixtures of 16S rRNA genes by PCR. Appl Environ Microbiol 62:625–630
Van Hilst CM, Smith WOJ (2002) Photosynthesis/irradiance relationships in the Ross Sea, Antarctica and their control by phytoplankton assemblage composition and environmental factors. Mar Ecol Prog Ser 226:1–12
Villafañe VE, Helbling EW, Holm-Hansen O (1995) Spatial and temporal variability of phytoplankton biomass and taxonomic composition around Elephant Island, Antarctica, during the summers of 1990–1993. Mar Biol 123:677–686
Watnick P, Kolter R (2000) Biofilm, city of microbes. J Bacteriol 182:2675–2679
Webster NS, Negri AP, Munro MM, Battershill CN (2004a) Diverse microbial communities inhabit Antarctic sponges. Environ Microbiol 6:288–300
Webster NS, Smith LD, Heyward AJ, Watts JE, Webb RI, Blackall LL, Negri AP (2004b) Metamorphosis of a scleractinian coral in response to microbial biofilms. Appl Environ Microbiol 70:1213–1221
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic, New York, pp 315–322
Wieczorek SK, Todd CD (1998) Inhibition and facilitation of settlement of epifaunal marine invertebrate larvae by microbial biofilm cues. Biofouling 12:81–118
Acknowledgments
We thank S. Delean for assistance with statistical analysis and W. Mclean, C. Wolff, and J. Monkivitch for field assistance. This research was supported by Antarctica New Zealand, University of Canterbury, and a NZ Foundation for Research Science and Technology Postdoctoral Scholarship. We are also indebted to all of the scientific support staff at Scott Base and McMurdo Station during 2001/2002. All experiments comply with the current laws of the country in which they were performed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Webster, N.S., Battershill, C.N. & Negri, A.P. Recruitment of Antarctic marine eukaryotes onto artificial surfaces. Polar Biol 30, 1–10 (2006). https://doi.org/10.1007/s00300-006-0153-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-006-0153-2