Abstract
Key message
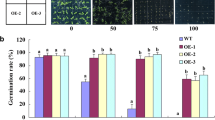
A novel function of plasma membrane-localized H+-ATPase, OsAHA3, was identified in rice, which is involved in saline–alkaline tolerance and specifically responds to high pH during saline–alkaline stress.
Abstract
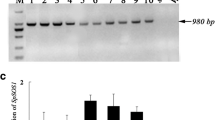
Saline–alkaline stress causes serious damage to crop production on irrigated land. Plants suffer more severe damage under saline–alkaline stress than under salinity stress alone. Plasma membrane-localized proton (H+) pump (H+-ATPase) is an important enzyme that controls plant growth and development by catalyzing H+ efflux and enabling effective charge balance. Many studies about the role of plasma membrane H+-ATPases in saline–alkaline stress tolerance have been reported in Arabidopsis, especially on the AtAHA2 (Arabidopsis thaliana H+-ATPase 2) gene; however, whether and how plasma membrane H+-ATPases play a role in saline–alkaline stress tolerance in rice remain unknown. Here, using the activation-tagged rice mutant pool, we found that the plasma membrane-localized H+-ATPase OsAHA3 (Oryza sativa autoinhibited H+-ATPase 3) is involved in saline–alkaline stress tolerance. Activation-tagged line 29 (AC29) was identified as a loss-of-function mutant of OsAHA3 and showed more severe growth retardation under saline–alkaline stress with high pH than under salinity stress. Moreover, osaha3 loss-of-function mutants generated by CRISPR/Cas9 system exhibited saline–alkaline stress sensitive phenotypes; staining of leaves with nitrotetrazolium blue chloride (NBT) and diaminobenzidine (DAB) revealed more reactive oxygen species (ROS) accumulation in osaha3 mutants. OsAHA3-overexpressing plants showed increased saline–alkaline stress tolerance than wild-type plants. Tissue-specific expression analysis revealed high expression level of OsAHA3 in leaf, sheath, glume, and panicle. Overall, our results revealed a novel function of plasma membrane-localized H+-ATPase, OsAHA3, which is involved in saline–alkaline stress tolerance and specifically responds to high pH.





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abdel LAA, Tran L-SP (2016) Impacts of priming with silicon on the growth and tolerance of maize plants to alkaline stress. Front Plant Sci 7:243. https://doi.org/10.3389/fpls.2016.00243
Adcock D, McNeill AM, McDonald GK, Armstrong RD (2007) Subsoil constraints to crop production on neutral and alkaline soils in south-eastern Australia: a review of current knowledge and management strategies. Aust J Exp Agric 47(11):1245–1261. https://doi.org/10.1071/ea06250
Ali M, Afzal S, Parveen A, Kamran M, Javed MR, Abbasi GH, Malik Z, Riaz M, Ahmad S, Chattha MS, Ali M, Ali Q, Uddin MZ, Rizwan M, Ali S (2021) Silicon mediated improvement in the growth and ion homeostasis by decreasing Na+ uptake in maize (Zea mays L.) cultivars exposed to salinity stress. Plant Physiol Biochem 158:208–218. https://doi.org/10.1016/j.plaphy.2020.10.040
Ariz I, Asensio AC, Zamarreño AM, García-Mina JM, Aparicio-Tejo PM, Moran JF (2013) Changes in the C/N balance caused by increasing external ammonium concentrations are driven by carbon and energy availabilities during ammonium nutrition in pea plants: the key roles of asparagine synthetase and anaplerotic enzymes. Physiol Plant 148(4):522–537. https://doi.org/10.1111/j.1399-3054.2012.01712.x
Aung TT, Shi F, Zhai Y, Xue J, Wang S, Ren X, Zhang X (2022) Acidic and alkaline conditions affect the growth of tree peony plants via altering photosynthetic characteristics, limiting nutrient assimilation, and impairing ROS balance. Int J Mol Sci 23(9):5094. https://doi.org/10.3390/ijms23095094
Axelsen KB, Venema K, Jahn T, Baunsgaard L, Palmgren MG (1999) Molecular dissection of the C-terminal regulatory domain of the plant plasma membrane H+-ATPase AHA2: mapping of residues that when altered give rise to an activated enzyme. Biochemistry 38(22):7227–7234. https://doi.org/10.1021/bi982482l
Baxter I, Tchieu J, Sussman MR, Boutry M, Palmgren MG, Gribskov M, Harper JF, Axelsen KB (2003) Genomic comparison of P-type ATPase ion pumps in Arabidopsis and rice. Plant Physiol 132(2):618–628. https://doi.org/10.1104/pp.103.021923
Baxter IR, Young JC, Armstrong G, Foster N, Bogenschutz N, Cordova T, Peer WA, Hazen SP, Murphy AS, Harper JF (2005) A plasma membrane H+-ATPase is required for the formation of proanthocyanidins in the seed coat endothelium of Arabidopsis thaliana. Proc Natl Acad Sci USA 102(7):2649–2654. https://doi.org/10.1073/pnas.0406377102
Chang C, Hu Y, Sun S, Zhu Y, Ma G, Xu G (2009) Proton pump OsA8 is linked to phosphorus uptake and translocation in rice. J Exp Bot 60(2):557–565. https://doi.org/10.1093/jxb/ern298
Falhof J, Pedersen JT, Fuglsang AT, Palmgren M (2016) Plasma membrane H+-ATPase regulation in the center of plant physiology. Mol Plant 9(3):323–337. https://doi.org/10.1016/j.molp.2015.11.002
Fang S, Hou X, Liang X (2021) Response mechanisms of plants under saline-alkali stress. Front Plant Sci 12:667458. https://doi.org/10.3389/fpls.2021.667458
Fuglsang AT, Guo Y, Cuin TA, Qiu Q, Song C, Kristiansen KA, Bych K, Schulz A, Shabala S, Schumaker KS, Palmgren MG, Zhu JK (2007) Arabidopsis protein kinase PKS5 inhibits the plasma membrane H+ -ATPase by preventing interaction with 14–3-3 protein. Plant Cell 19(5):1617–1634. https://doi.org/10.1105/tpc.105.035626
Geilfus CM (2017) The pH of the apoplast: dynamic factor with functional impact under stress. Mol Plant 10(11):1371–1386. https://doi.org/10.1016/j.molp.2017.09.018
Hager A (2003) Role of the plasma membrane H+-ATPase in auxin-induced elongation growth: historical and new aspects. J Plant Res 116(6):483–505. https://doi.org/10.1007/s10265-003-0110-x
Havshøi NW, Fuglsang AT (2022) A critical review on natural compounds interacting with the plant plasma membrane H+-ATPase and their potential as biologicals in agriculture. J Integr Plant Biol 64(2):268–286. https://doi.org/10.1111/jipb.13221
Hoffmann RD, Olsen LI, Ezike CV, Pedersen JT, Manstretta R, López-Marqués RL, Palmgren M (2019) Roles of plasma membrane proton ATPases AHA2 and AHA7 in normal growth of roots and root hairs in Arabidopsis thaliana. Physiol Plant 166(3):848–861. https://doi.org/10.1111/ppl.12842
Hoffmann RD, Portes MT, Olsen LI, Damineli DSC, Hayashi M, Nunes CO, Pedersen JT, Lima PT, Campos C, Feijó JA, Palmgren M (2020) Plasma membrane H+-ATPases sustain pollen tube growth and fertilization. Nat Commun 11(1):2395. https://doi.org/10.1038/s41467-020-16253-1
Houlné G, Boutry M (1994) Identification of an Arabidopsis thaliana gene encoding a plasma membrane H+-ATPase whose expression is restricted to anther tissue. Plant J 5(3):311–317. https://doi.org/10.1111/j.1365-313x.1994.00311.x
Inoue SI, Kinoshita T (2017) Blue light regulation of stomatal opening and the plasma membrane H+-ATPase. Plant Physiol 174(2):531–538. https://doi.org/10.1104/pp.17.00166
Inoue SI, Takahashi K, Okumura-Noda H, Kinoshita T (2016) Auxin influx carrier AUX1 confers acid resistance for Arabidopsis root elongation through the regulation of plasma membrane H+-ATPase. Plant Cell Physiol 57(10):2194–2201. https://doi.org/10.1093/pcp/pcw136
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. Embo J 6(13):3901–3907. https://doi.org/10.1002/j.1460-2075.1987.tb02730.x
Li Q, Yang A, Zhang WH (2016) Efficient acquisition of iron confers greater tolerance to saline-alkaline stress in rice (Oryza sativa L.). J Exp Bot 67(22):6431–6444. https://doi.org/10.1093/jxb/erw407
Li Y, Zeng H, Xu F, Yan F, Xu W (2022) H+-ATPases in plant growth and stress responses. Annu Rev Plant Biol 73:495–521. https://doi.org/10.1146/annurev-arplant-102820-114551
Liu YG, Mitsukawa N, Oosumi T, Whittier RF (1995) Efficient isolation and mapping of Arabidopsis thaliana T-DNA insert junctions by thermal asymmetric interlaced PCR. Plant J 8(3):457–463. https://doi.org/10.1046/j.1365-313x.1995.08030457.x
Liu Y, Chen X, Xue S, Quan T, Cui D, Han L, Cong W, Li M, Yun DJ, Liu B, Xu ZY (2021) SET DOMAIN GROUP 721 protein functions in saline-alkaline stress tolerance in the model rice variety Kitaake. Plant Biotechnol J 19(12):2576–2588. https://doi.org/10.1111/pbi.13683
Liu Y, Li M, Yu J, Ma A, Wang J, Yun DJ, Xu ZY (2023) Plasma membrane-localized Hsp40/DNAJ chaperone protein facilitates OsSUVH7-OsBAG4-OsMYB106 transcriptional complex formation for OsHKT1;5 activation. J Integr Plant Biol 65(1):265–279. https://doi.org/10.1111/jipb.13403
Ma X, Zhang Q, Zhu Q, Liu W, Chen Y, Qiu R, Wang B, Yang Z, Li H, Lin Y, Xie Y, Shen R, Chen S, Wang Z, Chen Y, Guo J, Chen L, Zhao X, Dong Z, Liu YG (2015) A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants. Mol Plant 8(8):1274–1284. https://doi.org/10.1016/j.molp.2015.04.007
Meng F-h, Wei Y-z, Yang X-e, J-j L, Liu J-x (2004) Effects of bicarbonate and high pH conditions on zinc and other nutrients absorption in rice. Rice Sci 11(5/6):290–296
Merlot S, Leonhardt N, Fenzi F, Valon C, Costa M, Piette L, Vavasseur A, Genty B, Boivin K, Müller A, Giraudat J, Leung J (2007) Constitutive activation of a plasma membrane H+-ATPase prevents abscisic acid-mediated stomatal closure. Embo j 26(13):3216–3226. https://doi.org/10.1038/sj.emboj.7601750
Michelet B, Boutry M (1995) The plasma membrane H+-ATPase (a highly regulated enzyme with multiple physiological functions). Plant Physiol 108(1):1–6. https://doi.org/10.1104/pp.108.1.1
Mikula K, Izydorczyk G, Skrzypczak D, Mironiuk M, Moustakas K, Witek-Krowiak A, Chojnacka K (2020) Controlled release micronutrient fertilizers for precision agriculture: a review. Sci Total Environ 712:136365. https://doi.org/10.1016/j.scitotenv.2019.136365
Monshausen GB, Bibikova TN, Messerli MA, Shi C, Gilroy S (2007) Oscillations in extracellular pH and reactive oxygen species modulate tip growth of Arabidopsis root hairs. Proc Natl Acad Sci USA 104(52):20996–21001. https://doi.org/10.1073/pnas.0708586104
Morsomme P, Boutry M (2000) The plant plasma membrane H+-ATPase: structure, function and regulation. Biochim Biophys Acta 1465(1–2):1–16. https://doi.org/10.1016/s0005-2736(00)00128-0
Msimbira LA, Smith DL (2020) The roles of plant growth promoting microbes in enhancing plant tolerance to acidity and alkalinity stresses. Front Sustain Food Syst 4:106. https://doi.org/10.3389/fsufs.2020.00106
Ni Z, Kim E-D, Ha M, Lackey E, Liu J, Zhang Y, Sun Q, Chen ZJ (2008) Altered circadian rhythms regulate growth vigour in hybrids and allopolyploids. Nature 457(7227):327–331. https://doi.org/10.1038/nature07523
Palmgren MG (2001) Plant plasma membrane H+-ATPases: powerhouses for nutrient uptake. Annu Rev Plant Physiol Plant Mol Biol 52:817–845. https://doi.org/10.1146/annurev.arplant.52.1.817
Palmgren MG, Sommarin M, Serrano R, Larsson C (1991) Identification of an autoinhibitory domain in the C-terminal region of the plant plasma membrane H+-ATPase. J Biol Chem 266(30):20470–20475. https://doi.org/10.1016/S0021-9258(18)54948-6
Paul MJ, Pellny TK (2003) Carbon metabolite feedback regulation of leaf photosynthesis and development. J Exp Bot 54(382):539–547. https://doi.org/10.1093/jxb/erg052
Rao Y, Peng T, Xue S (2023) Mechanisms of plant saline-alkaline tolerance. J Plant Physiol 281:153916. https://doi.org/10.1016/j.jplph.2023.153916
Robertson WR, Clark K, Young JC, Sussman MR (2004) An Arabidopsis thaliana plasma membrane proton pump is essential for pollen development. Genetics 168(3):1677–1687. https://doi.org/10.1534/genetics.104.032326
Serrano R (1984) Purification of the proton pumping ATPase from plant plasma membranes. Biochem Biophys Res Commun 121(2):735–740. https://doi.org/10.1016/0006-291x(84)90243-2
Sperandio MV, Santos LA, Bucher CA, Fernandes MS, de Souza SR (2011) Isoforms of plasma membrane H+-ATPase in rice root and shoot are differentially induced by starvation and resupply of NO3-or NH4+. Plant Sci 180(2):251–258. https://doi.org/10.1016/j.plantsci.2010.08.018
Takahashi K, Hayashi K, Kinoshita T (2012) Auxin activates the plasma membrane H+-ATPase by phosphorylation during hypocotyl elongation in Arabidopsis. Plant Physiol 159(2):632–641. https://doi.org/10.1104/pp.112.196428
Toda Y, Wang Y, Takahashi A, Kawai Y, Tada Y, Yamaji N, Feng Ma J, Ashikari M, Kinoshita T (2016) Oryza sativa H+-ATPase (OSA) is involved in the regulation of dumbbell-shaped guard cells of rice. Plant Cell Physiol 57(6):1220–1230. https://doi.org/10.1093/pcp/pcw070
Vitart V, Baxter I, Doerner P, Harper JF (2001) Evidence for a role in growth and salt resistance of a plasma membrane H+-ATPase in the root endodermis. Plant J 27(3):191–201. https://doi.org/10.1046/j.1365-313x.2001.01081.x
Yang Y, Qin Y, Xie C, Zhao F, Zhao J, Liu D, Chen S, Fuglsang AT, Palmgren MG, Schumaker KS, Deng XW, Guo Y (2010) The Arabidopsis chaperone J3 regulates the plasma membrane H+-ATPase through interaction with the PKS5 kinase. Plant Cell 22(4):1313–1332. https://doi.org/10.1105/tpc.109.069609
Yang Y, Wu Y, Ma L, Yang Z, Dong Q, Li Q, Ni X, Kudla J, Song C, Guo Y (2019) The Ca2+ sensor SCaBP3/CBL7 modulates plasma membrane H+-ATPase activity and promotes alkali tolerance in Arabidopsis. Plant Cell 31(6):1367–1384. https://doi.org/10.1105/tpc.18.00568
Yoshida S, Fomo DA, Cock JH, Gomez KA (1976) Routine procedures for growing rice plants in culture solution. Laboratory Manual for Physiological Studies of Rice, pp 61–66
Zhang Y, Su J, Duan S, Ao Y, Dai J, Liu J, Wang P, Li Y, Liu B, Feng D, Wang J, Wang H (2011) A highly efficient rice green tissue protoplast system for transient gene expression and studying light/chloroplast-related processes. Plant Methods 7(1):30. https://doi.org/10.1186/1746-4811-7-30
Zhang M, Wang Y, Chen X, Xu F, Ding M, Ye W, Kawai Y, Toda Y, Hayashi Y, Suzuki T, Zeng H, Xiao L, Xiao X, Xu J, Guo S, Yan F, Shen Q, Xu G, Kinoshita T, Zhu Y (2021) Plasma membrane H+-ATPase overexpression increases rice yield via simultaneous enhancement of nutrient uptake and photosynthesis. Nat Commun 12(1):735. https://doi.org/10.1038/s41467-021-20964-4
Zhao Z, Fan J, Gao YG, Wang Z, Yang P, Liang Y, Opiyo S, Xia Y (2022) Arabidopsis plasma membrane ATPase AHA5 is negatively involved in PAMP-triggered immunity. Int J Mol Sci 23(7):3857. https://doi.org/10.3390/ijms23073857
Zhou Z, Wu Y, Yang Y, Du M, Zhang X, Guo Y, Li C, Zhou JM (2015) An Arabidopsis plasma membrane proton ATPase modulates JA signaling and is exploited by the Pseudomonas syringae effector protein AvrB for stomatal invasion. Plant Cell 27(7):2032–2041. https://doi.org/10.1105/tpc.15.00466
Zhou JY, Hao DL, Yang GZ (2021) Regulation of cytosolic pH: the contributions of plant plasma membrane H+-ATPases and multiple transporters. Int J Mol Sci 22(23):12998. https://doi.org/10.3390/ijms222312998
Funding
This research was supported by the National Natural Science Foundation of China (32272027).
Author information
Authors and Affiliations
Contributions
Z-YX conceptualized and supervised the research. ML performed most of the physiological experiments and plasmid construction. PG performed the screening of activation-tagged lines and some physiological analyses. NN carried out the identification of plant materials. AM established the screening system. WL performed the qPCR analysis. T-JW assisted in caring for the plant materials. D-JY provided helpful suggestions. Z-YX wrote the manuscript. All authors reviewed, revised, and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Byeong-ha Lee.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, M., Guo, P., Nan, N. et al. Plasma membrane-localized H+-ATPase OsAHA3 functions in saline–alkaline stress tolerance in rice. Plant Cell Rep 43, 9 (2024). https://doi.org/10.1007/s00299-023-03103-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00299-023-03103-9